AbstractPurposeWe evaluated the association between changes in social support after cancer treatment and recurrence-free survival (RFS) in such patients using a prospective cohort study.
Materials and MethodsData were obtained from a prospective cohort study (NCT03131089) conducted at Samsung Medical Center (2013-2021). The primary outcome measure was RFS. Social support was measured using the social and family well-being (SFWB) domain of the Functional Assessment of Cancer Therapy-General. We calculated the changes in SFWB scores before and during treatment and the hazard ratio for RFS by comparing such changes.
ResultsThe mean±standard deviation (SD) age of the patients was 35±3.9 years, and 71.5% and 64.8% of the patients were married and had children, respectively. The mean±SD SFWB score at baseline was 20.5±5.0 out of 26. After cancer treatment, 35.9%, 10.3%, and 53.8% of the participants had increasing, unchanged, and decreasing SFWB scores, respectively. The decreasing SFWB score group had a higher risk of mortality or recurrence than the increasing group. Risk factors for the decreasing score were the presence of children during diagnosis.
IntroductionPatients with breast cancer face multifaceted challenges in cancer diagnosis and treatment that cause physical and psychosocial distress, such as anxiety, fear of recurrence, posttraumatic stress disorder, cognitive dysfunction, sexuality, body image, social isolation, and loneliness [1,2]. Especially, young patients with breast cancer report many problems related to social functions and role functions during treatment, as they often play multiple roles as mothers, spouses, friends, and workers [3,4]. Thus, according to a standard set of value-based patient-centered outcomes for breast cancer, social function and social support are recommended as important patient-reported outcomes [5].
Social support is a key factor that can alleviate cancer-related distress and improve the emotional and psychological needs of breast cancer survivors [6,7] by enhancing their mood and sense of identity. More importantly, several studies have reported that social support may be associated with the survival of patients with cancer [8-10]. In a cohort study of 1,004 cancer patients, those living alone at the time of diagnosis had an 11%-80% statistically significant increase in all-cause mortality [9]. In another cohort study of 2,835 breast cancer patients, those who were socially isolated before diagnosis had a 2.14 times higher risk of breast cancer mortality (95% confidence interval [CI], 1.11 to 4.12) compared to women who were socially integrated [8]. Furthermore, in a cohort study of 9,267 women from the After Breast Cancer Pooling Project, socially isolated women, within approximately 2 years following diagnosis, had a 1.43 higher risk of recurrence (95% CI, 1.15 to 1.77) than socially integrated women [11].
Additionally, many survivors need to make lifestyle adjustments after a cancer treatments and it can lead to altered relationships and isolation from friends and family [12]. In fact, according to a longitudinal study, the quantity of social support provided significantly decreased during treatment [13]. However, the impact of social support decline after treatment on clinical outcomes among young patients (≤ 40 years old) with breast cancer is not well understood. Social support may play a larger role for younger patients with breast cancer, as the burden of the disease may be greater for younger women than for their older counterparts [14]. The treatment is often more aggressive, which heightens the need for both emotional and instrumental support. Moreover, younger patients with breast cancer are in the midst of their productive years; family and work demands are already high in addition to their illness, thus, making the need for emotional support even more critical [14,15]. Social support is associated with reduced stress and a higher quality of life (QOL) in cancer patients [16], which were factors that have been associated with disease progression [17]. Thus, we compared the recurrence-free survival (RFS) between patients who experienced a decrease or no change in social support and those who experienced an increase in social support after cancer treatment.
Materials and Methods1. Study design and populationThe data for this study were collected from a prospective cohort study of young patients with breast cancer, known as the Samsung Medical Center–Young Breast Cancer Registry (NCT03131089). The cohort study enrolled patients from Samsung Medical Center, Seoul, South Korea, starting from May 11, 2013. For this particular research, we included patients who were diagnosed with breast cancer between May 2013 and April 2021 (n=1,552), and they had to be 40 years or younger at the time of diagnosis. Patient-reported outcomes, including QOL, function, and symptoms, were measured before treatment, at the 30-month mark, and every 6 months thereafter.
Our study aimed to evaluate the association between changes in social well-being before and during treatment and disease progression. Thus, we included patients who had completed the questionnaire before treatment and three months after diagnosis (during treatment) and did not show recurrence during this period, resulting in 610 participants in the final sample.
2. MeasurementsThe primary outcome measure was RFS. To reduce immortal and selection bias, we excluded patients who had a recurrence or died within three months after diagnosis (the date of second visit for the response social and family well-being [SFWB]); then, we followed up with patients from the second date of visit (index date) to the date of recurrence, death, last surveillance without evidence of recurrence, or 5 years since follow-up, whichever came first.
Social support was measured using the SFWB domain in the Korean version of the Functional Assessment of Cancer Therapy-General (FACT-G) [18]. This domain consists of 27 questions, evaluating four primary dimensions of QOL: SFWB (7 items), physical well-being (PWB; 7 items), emotional well-being (EWB; 6 items), and functional well-being (FWB; 7 items). The Likert-type response categories range from 0=“not at all” to 4=“very much.” The maximum scores for SFWB, PWB, EWB, and FWB are 28, 28, 24, and 28, respectively. The total FACT-G score is the sum of the four subscale scores and ranges from 0 to 108. We handled incomplete questionnaires based on the recommendations provided by the developers.
To generate changes in SFWB, we calculated the difference in the score between SFWB before and during treatment (SFWB during treatment–SFWB before treatment). Using the difference in the score, patients whose scores were zero were categorized as “no change,” and those with scores < 0 and > 0 were recognized as the “decreasing” and “increasing” groups, respectively. In terms of individual SFWB items, we calculated the change in the item score using the same procedure as the total score.
Sociodemographic and clinical information, including Eastern Cooperative Oncology Group (ECOG), comorbidity, pathology stage at diagnosis, subtype, pathogenic mutation, and treatment modality, is regularly updated by a trained data manager.
3. Statistical analysisThe primary outcome of this study was RFS. We used Cox proportional hazards regression models to estimate HRs with 95% CI for RFS, comparing the “increasing” SFWB score to the “no change” and “decreasing” SFWB scores. Considering the influence of clinical, demographic, and socioeconomic factors on RFS, we adjusted for age, ECOG performance status, history of birth, year of diagnosis, marital status, job status, social support at diagnosis, stage, and subtype. We observed high correlation between treatment and stage or subtype, resulting in variance inflation factors values exceeding 5. Consequently, we decided not to include the treatment values in the analysis [19]. Furthermore, participants in our analyses had to report patient-reported well-being at the end of active treatment (1 year since diagnosis); therefore, we used inverse probability censoring weights to correct for potential selection bias.
To identify the risk factors at baseline for no change or decreasing SFWB during treatment, we calculated the relative risk (RR). As the dependent variable had more than two categorical types, we used multinomial logistic regression.
Statistical significance was set at p < 0.05, and two-sided tests were used in all calculations. Statistical analyses were performed using STATA ver. 16.0 (StataCorp LP, College Station, TX).
Results1. Characteristics of the study populationThe mean±standard deviation (SD) age of the participants was 35±3.9 years, and 71.5% and 64.8% were married and had children, respectively (Table 1). The mean±SD SFWB score at baseline was 20.5±5.0 out of 26.
Between diagnoses and during treatment, the largest decreasing item was “I get support from my friends” (38.2%) followed by “I feel close to my friends” (36.5%). Additionally, the largest increasing item was “my family has accepted my illness” (29.9%) followed by “I am satisfied with family communication about my illness” (23.7%) (Fig. 1). The mean (range) of change in the total SFWB score was –0.9 (–14 and 17.5). Between diagnoses and during treatment, 35.9%, 10.3%, and 53.8% of the participants had an increasing, no change and decreasing SFWB score, respectively. When we compared characteristics, age, ECOG performance status, comorbidity, stage, and subtype did not differ among the three groups by changes in SFWB, while the decreasing group was more likely to have children than the other groups (57.1% vs. 68.3% vs. 69.2) (Table 1).
2. Change of social/family well-being and recurrence-free survivalDuring 2,265 person-years of follow-up (median follow-up, 4.4 years), we observed 57 recurrences or deaths, and the weighted incidence rate was 4.1 per 100 person-years. The incident rates in increasing, no change, and decreasing SFWB scores were 2.4, 3.3, and 5.3 per 100 person-years, respectively (Table 2, Fig. 2). The decreasing SFWB score group was associated with a higher risk of mortality or recurrence compared to the increasing group, even after adjusting for age, ECOG performance status, history of birth, year of diagnosis, marital status, job status, total well-being score, social/family well-being at diagnosis, stage, and subtype (adjusted HR comparing increasing group [reference], 2.49; 95% CI, 1.04 to 5.98; p for trends=0.044) (Table 2). In the sensitivity analysis, further adjusted for the treatment modality, the adjusted HR of the decreasing SFWB score group, comparing it to the increasing group [reference], was 2.79 (95% CI, 1.18 to 6.60; p for trend=0.022). In particular, decreasing emotional support from the family (HR, 4.03; 95% CI, 1.16 to 13.96; p for trends=0.041) was associated with an increased risk of mortality or recurrence (Table 2).
3. Risk factors at diagnosis for decreasing social/family well-being between diagnosis and treatmentThe fully adjusted RR for no change and decreasing SFWB scores in patients with children at diagnosis were 1.14 (95% CI, 0.44 to 2.98) and 1.86 (95% CI, 1.01 to 3.40), respectively (Table 3). Although it was not statistically significant, married patients (RR for decreasing SFWB, 1.36; 95% CI, 0.88 to 2.09) and those who were pregnant when diagnosed (RR for decreasing SFWB, 1.22; 95% CI, 0.39 to 3.77) showed similar trends (Table 3).
In terms of clinical factors, although an advanced cancer stage showed a relatively higher risk for a decrease in the SFWB score (neoadjuvant cohort: RR, 1.41; 95% CI, 0.84 to 2.36; palliative cohort: RR, 1.74; 95% CI, 0.29 to 10.45), it was not statistically significant. Regarding the other dimensions of well-being at diagnosis, there was no association with a decrease in SFWB (Table 3).
DiscussionIn a prospective cohort of breast cancer patients aged less than 40 years, 53.8% had decreased SFWB scores after cancer treatment. Decreasing SFWB was associated with poorer RFS. Patients who were married, had children or were pregnant when diagnosed was associated with decreased SFWB in the period between before and during treatment.
Among breast cancer patients aged 40 years or younger, half of the participants had decreasing SFWB scores in the period between before and during treatment. This result is consistent with a previous study, which showed that social support quantity significantly decreased during treatment [13]. Several changes occur in a family when a family member is diagnosed with cancer. According to a meta-analysis, the diagnosis and treatment of cancer affect the patient’s course of life, daily activities, work, relationships, and roles [20]. In this study, we also found that social support from friends decreased, while social support from family increased. According to a national survey in Korea, more than half of the public had negative attitudes, stereotypes, and discrimination toward cancer patients, despite medical advancements and improved survival rates [21]. Regardless of whether survivors experience discrimination from friends and neighbors, they may feel discriminated against because of self-stigmatization [22]. Cultural myths and stigma about cancer discourage cancer patients from sharing personal information, resulting in social isolation [22]. Public campaigns and education are necessary to increase awareness about cancer and provide the right information about cancer patients. Health professionals need to teach cancer patients self-coping and communication skills to enable them to seek the support they would need during treatment.
In this study, decreasing SFWB was associated with poorer RFS, compared to increasing SFWB, in the period between before and during treatment. Although the associations between social support and poor RFS have been reported previously [14], this study had strength reporting an association between changes in SFWB and poor RFS in patients aged 40 or less. Since younger than 40 years old is a time point when have the most responsibilities from family and jobs whole their life time, thus, making the need for emotional support even more critical [14,15]. Thus, young patients with breast cancer who experienced decreasing SFWB were more likely to a much more negative impact on behavioral adaptation in the face of stress, and it could affect biological functions and poor RFS by multifactorial mechanisms. First, resilience and psychosocial resources could mediate this positive relationship between social support and QOL of breast cancer survivors [23,24], and it also impacts on RFS. Greater social exchange and emotional support after a cancer diagnosis may benefit women’s survival by enhancing their coping skills, providing additional support, and increasing opportunities to obtain cancer-related information. Being a part of a network of supportive relationships can foster a sense of community necessary for successful coping with the disease and provide opportunities to learn from one another [14]. Second, the impact of social support on RFS could also be supported by the psycho-neuro-endocrinologic viewpoint that breast cancer survivors who are less satisfied with their social relationships have higher levels of inflammatory markers such as C-reactive protein, tumor necrosis factor-α, and interleukin-6 (IL-6) and 18 [25], which might disrupt their general health status and, thus, lower their functional status. In a cohort study, patients with diverse types of cancer who reported higher social support satisfaction presented a lower risk of mortality over time (HR, 0.89; 95% CI, 0.76 to 1.05) and lower levels of C-reactive protein, IL-6, and TNF-α [10]. Furthermore, oxytocin (OT) has remained understudied as a possible mediator of social influences on breast cancer; although it is released during social and physical contact, it serves important biological functions in the mammary gland and modulates both the hypothalamic-pituitary-adrenal axis and sympathetic nervous system. OT plays a role in the mechanisms through which social isolation affects a wide range of disease outcomes, including wound healing, pain responses, atherosclerosis, cerebral ischemia, and depressive-like behavior [26]. Given that OT is modulated by social isolation and interaction, directly affects tumor cell growth, is present with its receptor in tumors, and has the ability to interact with tumor-modifying immune cells, it is a protein that warrants further exploration in young patients with breast cancer [27].
In this study, clinical factors did not have a significant impact on reducing SFWB. This result was similar with previous studies, which found that emotional outcomes were not affected by the stage of cancer [28]. According to previous studies, even when patients were diagnosed with cancer, the impact of the diagnosis have negative impact on the patients regardless of stage [29]. On the other hand, patients who had children or pregnancies when diagnosed were associated with a decrease in SFWB during treatment. This is consistent with the results of a previous study [30]. Among diagnosed mothers under the age of 40, one particular challenge is that their children are usually minors and, thus, more dependent on them [31]. Motherhood remains a primary role, even when women are ill [31]. As mothers facing a breast cancer diagnosis and related treatments must concurrently manage their roles, these circumstances can adversely affect both their psychological well-being [32] and sense of competent mothering [33]. Especially, women are expected to dedicate themselves to being good wives and devoted mothers in the traditional Asian culture [34]. Thus, although frequent hospital visits for treatments can become overwhelming, especially due to the many side effects and symptoms, patients may feel that they are unable to fulfil their roles [34]. Consequently, open and effective communication of thoughts and emotions among family members should be encouraged. Social support dynamics, parental rules, and coping strategies should be explored and improved upon. Therefore, children and fathers should also be included in the psychosocial interventions offered to young patients with breast cancer.
Several limitations need to be considered when interpreting the results. First, most of the data were collected from self-reporting, which could be influenced by the participants’ characteristics or social desirability. However, we used standardized questionnaires to reduce potential bias. Second, we were unable to establish the exact mechanisms underlying the association between decreasing SFWB and a poor RFS. Further biological studies are required to confirm these results. Third, because this was an observational study, there were various confounding factors. However, we adjusted for important clinical factors such as age at diagnosis, stage, and type of cancer. Fourth, one of the primary reasons for the patients’ refusal to participate in our study was because they were feeling overwhelmed by cancer experience. Therefore, our findings may underestimate the change in SFWB in these patients. Finally, all the women in this study were Asian and well educated, which limits the generalizability of our findings.
Despite these limitations, the results of this study may provide valuable information on the relevance of changes in social support in the period between diagnosis and during treatment associated with RFS in young patients with breast cancer. While modern advances in supportive care have led to better symptom management and improved survival rates, there is relatively little preparation for the social environment factors during treatment. Health professionals should develop family interventions to help young patients with breast cancer receive proper social support.
NotesEthical Statement The study protocol was approved by our institutional review board (SMC 2013-04-005), and written informed consent was obtained from all participants. AcknowledgmentsThis study was supported by Future Medicine 20*30 Project of the Samsung Medical Center [grant numbers: SMX1210831].
Fig. 1.(A-F) Probability of change of social/family well-being between diagnosis and during treatment. 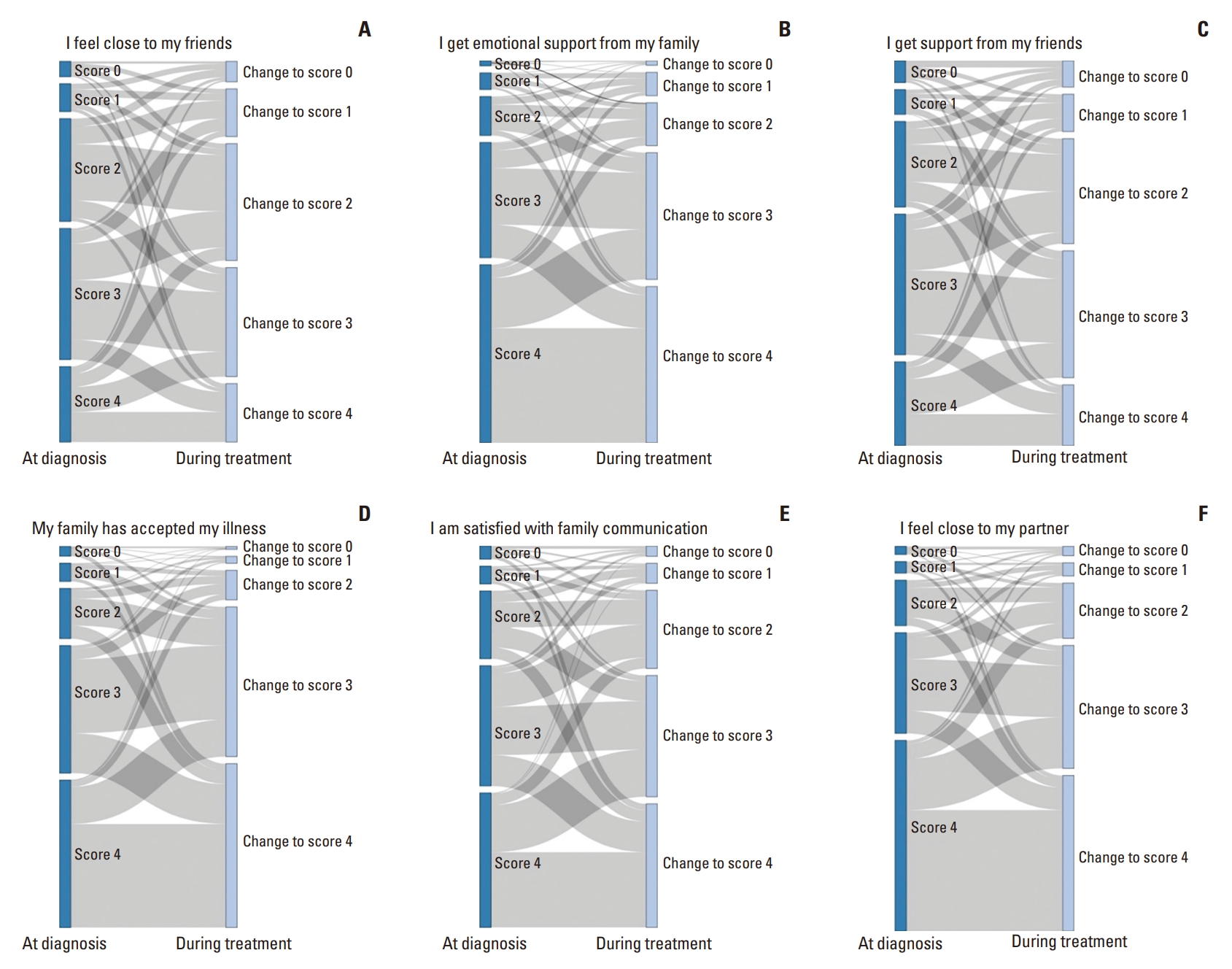
Fig. 2.Kaplan-Meier curve by change of social/family well-being between diagnosis and during treatment. Index date was three months after diagnosis (during active treatment). The curve was considered an inverse probability weight to correct for potential selection bias. 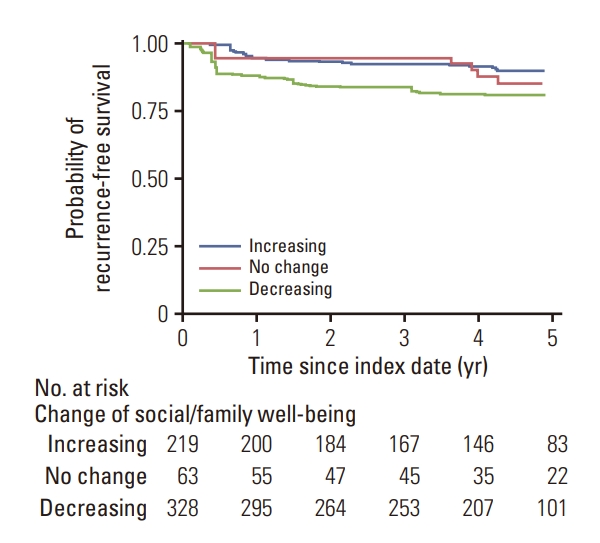
Table 1.Characteristics of study participants Values are presented as mean±SD or number (%). BMI, body mass index; BRCA, breast cancer susceptibility gene; ECOG, European Cooperative Oncology Group; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; SD, standard deviation; TNBC, triple-negative breast cancer. Include neoadjuvant or adjuvant. Table 2.Hazard ratios for incidents of recurrence or death by change of social support (n=610) Table 3.Multinomial logistic regression for baseline factors associated with no change or decreased social well-being from diagnosis to during treatment References1. Kenyon M, Mayer DK, Owens AK. Late and long-term effects of breast cancer treatment and surveillance management for the general practitioner. J Obstet Gynecol Neonatal Nurs. 2014;43:382–98.
2. Choi E, Henneghan AM. Comparing fatigue, loneliness, daytime sleepiness, and stress in younger and older breast cancer survivors: a cross-sectional analysis. Clin J Oncol Nurs. 2022;26:155–64.
3. Keesing S, Rosenwax L, McNamara B. The implications of women’s activity limitations and role disruptions during breast cancer survivorship. Womens Health (Lond). 2018;14:1745505718756381.
4. Mackenzie CR. ‘It is hard for mums to put themselves first’: how mothers diagnosed with breast cancer manage the sociological boundaries between paid work, family and caring for the self. Soc Sci Med. 2014;117:96–106.
5. Ong WL, Schouwenburg MG, van Bommel ACM, Stowell C, Allison KH, Benn KE, et al. A standard set of value-based patient-centered outcomes for breast cancer: the International Consortium for Health Outcomes Measurement (ICHOM) Initiative. JAMA Oncol. 2017;3:677–85.
6. Simpson JS, Carlson LE, Beck CA, Patten S. Effects of a brief intervention on social support and psychiatric morbidity in breast cancer patients. Psychooncology. 2002;11:282–94.
7. Mirrielees JA, Breckheimer KR, White TA, Denure DA, Schroeder MM, Gaines ME, et al. Breast cancer survivor advocacy at a university hospital: development of a peer support program with evaluation by patients, advocates, and clinicians. J Cancer Educ. 2017;32:97–104.
8. Kroenke CH, Kubzansky LD, Schernhammer ES, Holmes MD, Kawachi I. Social networks, social support, and survival after breast cancer diagnosis. J Clin Oncol. 2006;24:1105–11.
9. Elovainio M, Lumme S, Arffman M, Manderbacka K, Pukkala E, Hakulinen C. Living alone as a risk factor for cancer incidence, case-fatality and all-cause mortality: a nationwide registry study. SSM Popul Health. 2021;15:100826.
10. Boen CE, Barrow DA, Bensen JT, Farnan L, Gerstel A, Hendrix LH, et al. Social relationships, inflammation, and cancer survival. Cancer Epidemiol Biomarkers Prev. 2018;27:541–9.
11. Kroenke CH, Michael YL, Poole EM, Kwan ML, Nechuta S, Leas E, et al. Postdiagnosis social networks and breast cancer mortality in the After Breast Cancer Pooling Project. Cancer. 2017;123:1228–37.
12. Sammarco A, Konecny LM. Quality of life, social support, and uncertainty among Latina breast cancer survivors. Oncol Nurs Forum. 2008;35:844–9.
13. Fong AJ, Scarapicchia TM, McDonough MH, Wrosch C, Sabiston CM. Changes in social support predict emotional well-being in breast cancer survivors. Psychooncology. 2017;26:664–71.
14. Chou AF, Stewart SL, Wild RC, Bloom JR. Social support and survival in young women with breast carcinoma. Psychooncology. 2012;21:125–33.
15. Bloom JR, Stewart SL, Johnston M, Banks P, Fobair P. Sources of support and the physical and mental well-being of young women with breast cancer. Soc Sci Med. 2001;53:1513–24.
16. Ruiz-Rodriguez I, Hombrados-Mendieta I, Melguizo-Garin A, Martos-Mendez MJ. The association of sources of support, types of support and satisfaction with support received on perceived stress and quality of life of cancer patients. Integr Cancer Ther. 2021;20:1534735421994905.
17. Yan J, Chen Y, Luo M, Hu X, Li H, Liu Q, et al. Chronic stress in solid tumor development: from mechanisms to interventions. J Biomed Sci. 2023;30:8.
18. Lee EH, Chun M, Kang S, Lee HJ. Validation of the Functional Assessment of Cancer Therapy-General (FACT-G) scale for measuring the health-related quality of life in Korean women with breast cancer. Jpn J Clin Oncol. 2004;34:393–9.
19. Chatterjee S, Simonoff JS. Handbook of regression analysis. New York: John Wiley & Sons; 2013.
20. Zabalegui A, Sanchez S, Sanchez PD, Juando C. Nursing and cancer support groups. J Adv Nurs. 2005;51:369–81.
21. Cho J, Smith K, Choi EK, Kim IR, Chang YJ, Park HY, et al. Public attitudes toward cancer and cancer patients: a national survey in Korea. Psychooncology. 2013;22:605–13.
22. Ernst J, Mehnert A, Dietz A, Hornemann B, Esser P. Perceived stigmatization and its impact on quality of life: results from a large register-based study including breast, colon, prostate and lung cancer patients. BMC Cancer. 2017;17:741.
23. Harms CA, Cohen L, Pooley JA, Chambers SK, Galvao DA, Newton RU. Quality of life and psychological distress in cancer survivors: the role of psycho-social resources for resilience. Psychooncology. 2019;28:271–7.
24. Zhang H, Zhao Q, Cao P, Ren G. Resilience and quality of life: exploring the mediator role of social support in patients with breast cancer. Med Sci Monit. 2017;23:5969–79.
25. Shrout MR, Renna ME, Madison AA, Alfano CM, Povoski SP, Lipari AM, et al. Relationship satisfaction predicts lower stress and inflammation in breast cancer survivors: a longitudinal study of within-person and between-person effects. Psychoneuroendocrinology. 2020;118:104708.
26. Karelina K, DeVries AC. Modeling social influences on human health. Psychosom Med. 2011;73:67–74.
27. Hinzey A, Gaudier-Diaz MM, Lustberg MB, DeVries AC. Breast cancer and social environment: getting by with a little help from our friends. Breast Cancer Res. 2016;18:54.
28. Kissane DW, Grabsch B, Love A, Clarke DM, Bloch S, Smith GC. Psychiatric disorder in women with early stage and advanced breast cancer: a comparative analysis. Aust N Z J Psychiatry. 2004;38:320–6.
29. Dinapoli L, Colloca G, Di Capua B, Valentini V. Psychological aspects to consider in breast cancer diagnosis and treatment. Curr Oncol Rep. 2021;23:38.
30. Cai T, Huang Q, Yuan C. Emotional, informational and instrumental support needs in patients with breast cancer who have undergone surgery: a cross-sectional study. BMJ Open. 2021;11:e048515
31. Ares I, Lebel S, Bielajew C. The impact of motherhood on perceived stress, illness intrusiveness and fear of cancer recurrence in young breast cancer survivors over time. Psychol Health. 2014;29:651–70.
|
|
|||||||||||||||||||||||||||||||||||||