AbstractPurposeThe purpose of this study was to prospectively validate the Korean Cancer Study Group Geriatric Score (KG)-7, a novel geriatric screening tool, in older patients with advanced cancer planned to undergo first-line palliative chemotherapy.
Materials and MethodsParticipants answered the KG-7 questionnaire before undergoing geriatric assessment (GA) and first-line palliative chemotherapy. The performance of KG-7 was evaluated by calculating the sensitivity (SE), specificity (SP), positive and negative predictive value (PPV and NPV), balanced accuracy (BA), and area under the curve (AUC).
ResultsThe baseline GA and KG-7 results were collected from 301 patients. The median age was 75 years (range, 70 to 93 years). Abnormal GA was documented in 222 patients (73.8%). Based on the ≤ 5 cut-off value of KG-7 for abnormal GA, abnormal KG-7 score was shown in 200 patients (66.4%). KG-7 showed SE, SP, PPV, NPV, and BA of 75.7%, 59.7%, 84.4%, 46.0%, and 67.7%, respectively; AUC was 0.745 (95% confidence interval, 0.687 to 0.803). Furthermore, patients with higher KG-7 scores showed significantly longer survival (p=0.006).
IntroductionGeriatric assessment (GA) can identify various problems necessary to determine the appropriate geriatric intervention and follow-up strategies for older patients, otherwise not possible by conventional physical examination and/or history taking [1,2]. It is considered useful due to its ability to differentiate between older patients with good and diminished physiological reserves. In older patients with cancer, GA has been used to predict tolerability of cancer treatment [3,4], toxicity of chemotherapy [5,6], and survival [7,8]. However, the full GA consists of many domains with items, including medical, functional, neuropsychiatric, nutritional, and social assessments [9], which can be highly time consuming and labor intensive. Therefore, it can be ineffective in routine practice, especially in institutions with limited resources and high patient volume, especially in high-burden oncology clinics. To remedy this, a screening method that differentiates older patients who need the full GA from those who do not need such extensive approach may be highly beneficial [10].
To date, there are several geriatric screening tools available, including Geriatric 8 (G-8), Abbreviated Comprehensive Geriatric Assessment (aCGA), Groningen Frailty Index (GFI), Vulnerable Elders Survey-13 (VES-13), and Triage Risk Screening Tool (TRST) [11-13]. However, these methods provide insufficient information to precisely identify patients that need the full GA [9]. Moreover, they do not represent all domains evenly [9,14]. VES-13 was developed to identify vulnerable older individuals in a community, and TRST was developed to predict repeat emergency department visits [15,16]. Hence, the applicability of these methods in cancer patients may be limiting. In addition, domain of GA could be various based on cultural backgrounds and clinical practice patterns. Given these considerations, we have developed a novel screening tool: the Korean Cancer Study Group Geriatric Score (KG)-7 with objective, easy-to-answer questions that have been selected from a large dataset to represent each domain of GA with high sensitivity (SE) and specificity (SP) in the previous study [17].
In this study, we aimed to prospectively validate the clinical utility of KG-7 from an independent cohort of older patients with advanced cancer planned to undergo first-line palliative chemotherapy. We also assessed the prognostic value of KG-7 in terms of overall survival (OS).
Materials and MethodsThis prospective study to validate KG-7 was conducted as a substudy of another prospective, longitudinal, and multicenter cohort study to develop a prediction tool for adverse events ≥ grade 3 due to chemotherapy [18]. Inclusion criteria of this study were as follows: patient age ≥ 70 years; candidate for first-line palliative chemotherapy; and patients with histologically confirmed solid tumor. The exclusion criteria were hematologic malignancy, such as lymphoma, leukemia, and multiple myeloma; patient with a treatment plan of monotherapy with biologic agent or targeted agent, concurrent chemoradiotherapy, combination chemotherapy with investigational agents, or monotherapy with oral agents; and recurrent cases during adjuvant chemotherapy.
1. KG-7 and GAKG-7 questionnaire was answered before full GA. KG-7 consisted of seven questions evenly distributed to represent each domain of GA (Table 1). KG-7 scores ranged from 0 to 7, and higher scores indicated better conditions. In a previous study, the cut-off value of KG-7 was defined as 5.5 [17]. As in our previous studies, GA consisted of an evaluation of medical problems, social support, functional status, cognitive status, emotional status, nutritional status, and mobility [3,17,19]. To measure comorbidity, Charlson comorbidity index was used, which was divided into low (0 points), medium (1-2 points), high (3-4 points), and very high (≥ 5 points) groups in accordance with the original weighting system [20]. The functional status was tested using the activities of daily living (ADL) and Korean instrument activities of daily living (K-IADL) scores [21,22]. At least one item with dependency in ADL or K-IADL was categorized as ADLdependent or IADL-dependent, respectively. Timed get-upand-go test (TGUG) of greater than 20 seconds was defined as impaired mobility [23]. Cognitive function was tested using Mini-Mental Status Examination in the Korean version of the Consortium to Establish a Registry for Alzheimer's disease Assessment Packet (MMSE-KC), which was categorized into severe cognitive impairment (scores ≤ 16) and mild cognitive impairment (scores 17-24) [24]. For depression, shortform geriatric depression scale (SGDS) scores of 5 to 9 and of 10 or more showed mild depression and severe depression, respectively [25]. In terms of nutritional status, the mini nutritional assessment (MNA) score of less than 17.0 and between 17.0 and 23.5 indicated malnutrition and risk of malnutrition, respectively [26]. GA was evaluated by clinical research coordinators who underwent appropriate education – and were certified by the Korean Cancer Study Group (KCSG) for the standardization of GA. Abnormal GA was defined as deficits in at least two out of six domains (ADL, K-IADL, MMSE-KC, SGDS, MNA, and TGUG).
2. Statistical analysisSE, SP, positive and negative predictive value (PPV and NPV), balanced accuracy [BA=(SE+SP)/2], as well as the area under the curve (AUC) were calculated. The performance of KG-7 was numerically compared with that of G-8. The G-8 was obtained from MNA and age. OS was calculated from the date of GA to the last follow-up or any cause of death. The event for OS was defined as any cause of death. The probability of OS was calculated using Kaplan-Meier survival analysis with log-rank significance tests. All analyses were performed using PASW Statistics ver. 18 (SPSS Inc., Chicago, IL).
3. Ethical statementThis study was approved by the institutional review boards and independent ethics committee of each hospital and KCSG (KCSG PC13-09). This study was registered with the Clinical Research Information Service (CRiS, WHO ICTRP number: KCT0001071). All participating patients provided written informed consent.
Results1. Baseline characteristics and GABetween February 2014 and December 2015, 301 patients undergoing palliative chemotherapy were enrolled in 17 hospitals affiliated with KCSG. The baseline patient characteristics and results of GA are shown in Table 2. The median age was 75 years (range, 70 to 93 years). Forty-two patients (14.0%) were 80 years of age or older. Males were more than females (69.1% and 30.9%, respectively). The most common cancer types were colorectal cancer (28.9%), lung cancer (24.6%), and hepato-biliary-pancreatic cancer (22.3%). In terms of GA, Charlson comorbidity index showed low or medium risk of comorbidity (52.2% and 37.9%, respectively) in most patients. Dependency in ADL and IADL was identified in 28.6% and 41.2% of patients, respectively. MMSE indicated mild and severe impairment of cognitive function in 45.5% and 10.0% of patients, respectively. Mild and severe depression were detected in 30.6% and 13.3% of patients, respectively. The risk of malnutrition and having malnutrition by MNA were seen in 56.8% and 19.6% of patients, respectively. There were 47 patients (15.6%) who had impaired mobility by TGUG > 20 seconds or were unable to complete the test. In two patients, abnormality of GA could not be determined in accordance with the definition of abnormality defined above due to missing values of GA. Abnormal GA was identified in 222 patients (73.8%).
2. KG-7The KG-7 score was collected in all patients. The distribution of KG-7 score is shown in Fig. 1. SE and SP using different cutoffs were presented as a S1 Table. Based on the ≤ 5 cut-off value of KG-7 for abnormal GA, as suggested by a previous study [17], abnormal KG-7 score was shown in 200 patients (66.4%). The SE, SP, PPV, NPV, and BA were 75.7%, 59.7%, 84.4%, 46.0%, and 67.7%, respectively. According to the receiver operating characteristic (ROC) curve analysis, AUC was 0.745 (95% confidence interval [CI], 0.687 to 0.803; p < 0.001) (Fig. 2A).
3. The performance of KG-7 compared with that of G-8The G-8 score was calculated using MNA and age. In one patient, G-8 score could not be calculated due to missing values. There was the positive correlation between KG-7 and G-8 scores (Pearson correlation, 0.517; p < 0.001). With the G-8 cut-off value of ≤ 14 for abnormal GA, as suggested by a reference study [27], abnormal G-8 score was shown in 263 patients (87.4%). SE, SP, PPV, NPV, and BA were 94.1%, 28.6%, 79.0%, 61.1%, and 61.4%, respectively. According to ROC, AUC was 0.772 (95% CI, 0.710 to 0.834; p < 0.001) (Fig. 2B). The SE and NPV of G-8 were higher than those of KG-7. However, SP and PPV of G-8 were lower than those of KG-7. Furthermore, BA was higher in KG-7 than in G-8 (67.7% and 61.4%, respectively). Normal G-8 score (> 14 points) was shown in only 12.3% of all patients. Hence, G-8 showed a lower number of patients without the need of full GA than KG-7 (33.6%).
4. OS according to KG-7 scoreThe KG-7 scores showed a prognostic value for OS. Patients with a higher KG-7 score showed longer OS (p=0.006). The median OS was longer in patients with normal KG-7 scores than those with abnormal KG-7 scores (19 months vs. 10 months, p=0.003) (Fig. 3A). When patients were categorized into four groups according to KG-7 score (0, 1 vs. 2, 3 vs. 4, 5 vs. 6, 7), groups with higher scores also showed longer OS (median OS, 4 vs. 5 vs. 12 vs. 19 months, respectively; p=0.002) (Fig. 3B).
DiscussionIn this study, we prospectively validated the performance of KG-7, which is a novel screening tool suitable for an outpatient oncology clinic setting with low resources and high patient burden to identify patients in need of full GA [17]. KG-7 could identify patients with abnormal GA with comparable performance with G-8. Furthermore, KG-7 showed a prognostic value for OS.
KG-7 consists of seven easy-to-answer questions. In the previous development study, several characteristics of KG-7 have been reported to be noteworthy when compared with the characteristics of other screening tools [17]. First, KG-7 was based on a large GA dataset, which included the standard method in evaluating older patients. Second, the items in each domain of KG-7 were rearranged to encompass the entire GA and distributed evenly across each essential domain. Third, KG-7 was developed with the consideration of its applicability to cancer patients. Finally, KG-7 showed the prognostic value.
In a previous systematic review, SE and SP of each screening tool for predicting frailty on GA was various, ranging from 25% to 92% and 39% to 100%, respectively, depending on the definition of abnormal GA, prevalence of abnormal GA, enrolled population, and applied tools [9]. They did not show enough of a discriminative value to be applicable in routine practice. Recently, it has been reported that modifying or adding relevant items enhanced the SE and SP of the current tools [28,29]. Based on these findings, KG-7 showed affordable accuracy in this validation study (SE, SP, PPV, NPV, BA, and AUC were 75.7%, 59.7%, 84.4%, 46.0%, 67.7%, and 0.745, respectively). This value was somewhat attenuated compared to those reported in development cohort and retrospective cohort receiving first-line palliative chemotherapy [17]. This could be explained by lower incidence of abnormal GA (66.4% vs. 79.5%). However, in comparison with G-8, our study showed similar AUC value, although SE and NPV of KG-7 were slightly lower than those of G-8. SP and PPV were higher than those of G-8. Furthermore, BA was higher in KG-7 than in G-8.
GA should reflect various social and cultural aspects. Body mass index selected in G-8 is not acceptable for older Asian patients with cancer due to relatively smaller body mass indexes compared with older Western patients with cancer. Therefore, a modified or novel tool was necessary to meet the characteristics of Asian clinical setting and population. KG-7 is the first validated screening tool in older Asian patients with cancer.
The percentage of patients with a normal score of KG-7 was higher in this validation study than that in the previous development cohort study (33.6% vs. 20.5%) [17]. The percentage of patients with a normal score of G-8 was also higher in this study population than in the previous study (12.3% vs. 9.2%). This can be attributed to the characteristics of patients enrolled. In this validation study, candidates for first-line palliative chemotherapy were included prospectively, and all patients were fit to receive chemotherapy on physician’s decisions and were able to provide informed consent. This result suggests that KG-7 may be a good, functional screening tool, of which results could be different according to characteristics of patients.
In this validation study, prognostic factors of enrolled patients were homogeneous. Most patients had good performance status of Eastern Cooperative Oncology Group performance status 0-1 and less comorbidity of low/medium Charlson risk index. All patients were candidates for firstline palliative chemotherapy. Nevertheless, according to the KG-7 score, OS was significantly discriminated, similar to a previous study [17]. The findings of this study indicate that KG-7 is a good tool for routine practice as it provides not only screening but also prognosis.
There are some limitations in this study. First, KG-7 was obtained by face-to-face interview before full GA; however, the scores of G-8 were retrieved using MNA and age. Hence, our study is not a head-to-head comparison between KG-7 and G-8. However, because items of G-8 are identical to corresponding items of MNA and it was collected prospectively, the results of G-8 should be reliable. Although indirect, the comparison performed in this study between KG-7 and G-8 should provide the reader with the relative performance of KG-7. Second, this study was conducted as a substudy of another prospective, longitudinal, and multicenter cohort study. The validation of KG-7 was the secondary outcome of another study. However, GA was the essential data in the primary study and KG-7 was collected in all patients. Timing of obtaining KG-7 before GA was followed with great attention. Further studies are necessary to determine whether a two-step approach using KG-7 screening followed by full GA impacts patient outcome, including toxicity, chemotherapy completion rate, and ultimate survival of older cancer patients. Finally, abnormal GA was defined as two or more deficits of GA domains in our study, which have been used in many studies related with geriatric screening tools. Older patients with cancer who have only one deficit of GA domain should be identified to provide a proper geriatric intervention to correct the deficit.
In this study, we have validated the usefulness of KG-7 as a screening tool in older patients with cancer planned to receive first-line palliative chemotherapy. We showed that KG-7 was comparable to G-8. Moreover, OS was discriminated according to KG-7 scores. KG-7 could be used effectively in countries with high patient burden and low resources to select patients in need of full GA.
Electronic Supplementary MaterialSupplementary materials are available at Cancer Research and Treatment website (http://www.e-crt.org).
AcknowledgmentsAll authors would like to express our deepest respect and gratitude to late Prof. Hong Suk Song, a valued member of Korean Cancer Study Group, who participated as principal investigator and contributed greatly from initial stage of the study.
This study was supported by grants from the National R&D Program for Cancer Control, Ministry for Health and Welfare, Republic of Korea (study no: 1320370). The funding source did not play any role in the study except for funding.
Fig. 1.Distribution of Korean Cancer Study Group Geriatric Score (KG)-7 score according to status of geriatric assessment (GA). Values shown in Table are presented as number (%). 
Fig. 2.Receiver operating characteristic (ROC) curve comparing Korean Cancer Study Group Geriatric Score (KG)-7 (A) with Geriatric 8 (G-8) (B). CI, confidence interval. 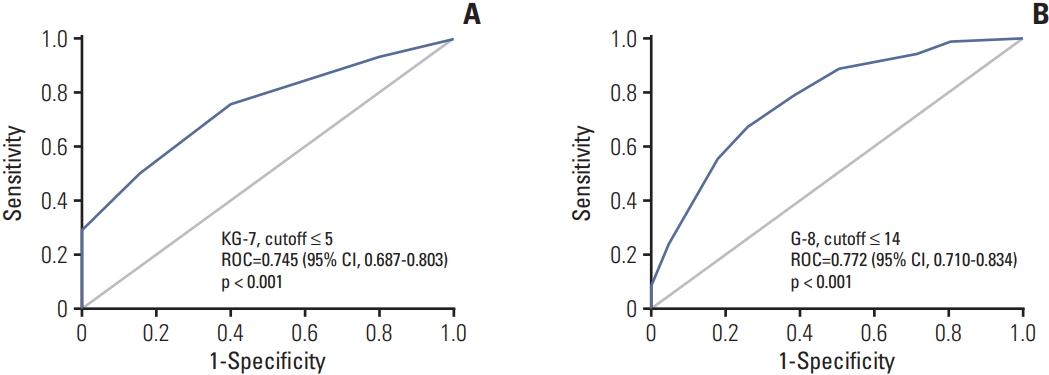
Fig. 3.Overall survival according to normal Korean Cancer Study Group Geriatric Score (KG)-7 vs. abnormal KG-7 score (A), overall survival based on subgroups of KG-7 scores (B). 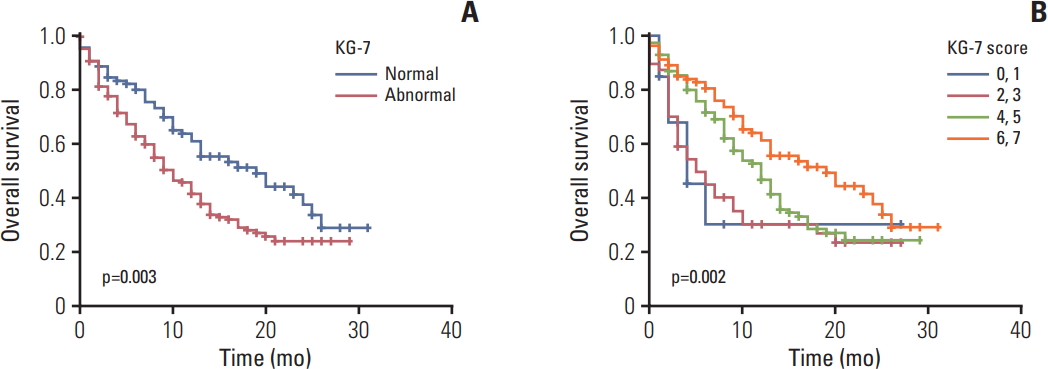
Table 1.The Korean Cancer Study Group Geriatric Score (KG)-7 Table 2.Baseline characteristics including geriatric assessment ECOG PS, Eastern Cooperative Oncology Group Scale of Performance Status; MMSE-KC, Mini-Mental Status Examination in the Korean version of the Consortium to Establish a Registry for Alzheimer's disease Assessment Packet; SGDS, Short-Form Geriatric Depression Scale; MNA, Mini Nutritional Assessment; TGUG, Timed Get Up and Go test. a)Others: breast (4), gynecological (4), esophageal cancer (3), sarcoma (2), melanoma (2), thymoma (1). References1. Kim YJ, Kim JH, Park MS, Lee KW, Kim KI, Bang SM, et al. Comprehensive geriatric assessment in Korean elderly cancer patients receiving chemotherapy. J Cancer Res Clin Oncol. 2011;137:839–47.
2. Extermann M, Hurria A. Comprehensive geriatric assessment for older patients with cancer. J Clin Oncol. 2007;25:1824–31.
3. Kim JW, Kim YJ, Lee KW, Chang H, Lee JO, Kim KI, et al. The early discontinuation of palliative chemotherapy in older patients with cancer. Support Care Cancer. 2014;22:773–81.
4. Extermann M, Aapro M, Bernabei R, Cohen HJ, Droz JP, Lichtman S, et al. Use of comprehensive geriatric assessment in older cancer patients: recommendations from the task force on CGA of the International Society of Geriatric Oncology (SIOG). Crit Rev Oncol Hematol. 2005;55:241–52.
5. Extermann M, Boler I, Reich RR, Lyman GH, Brown RH, DeFelice J, et al. Predicting the risk of chemotherapy toxicity in older patients: the Chemotherapy Risk Assessment Scale for High-Age Patients (CRASH) score. Cancer. 2012;118:3377–86.
6. Hurria A, Togawa K, Mohile SG, Owusu C, Klepin HD, Gross CP, et al. Predicting chemotherapy toxicity in older adults with cancer: a prospective multicenter study. J Clin Oncol. 2011;29:3457–65.
7. Kanesvaran R, Li H, Koo KN, Poon D. Analysis of prognostic factors of comprehensive geriatric assessment and development of a clinical scoring system in elderly Asian patients with cancer. J Clin Oncol. 2011;29:3620–7.
8. Soubeyran P, Fonck M, Blanc-Bisson C, Blanc JF, Ceccaldi J, Mertens C, et al. Predictors of early death risk in older patients treated with first-line chemotherapy for cancer. J Clin Oncol. 2012;30:1829–34.
9. Hamaker ME, Jonker JM, de Rooij SE, Vos AG, Smorenburg CH, van Munster BC. Frailty screening methods for predicting outcome of a comprehensive geriatric assessment in elderly patients with cancer: a systematic review. Lancet Oncol. 2012;13:e437
10. Decoster L, Van Puyvelde K, Mohile S, Wedding U, Basso U, Colloca G, et al. Screening tools for multidimensional health problems warranting a geriatric assessment in older cancer patients: an update on SIOG recommendationsdagger. Ann Oncol. 2015;26:288–300.
11. Kellen E, Bulens P, Deckx L, Schouten H, Van Dijk M, Verdonck I, et al. Identifying an accurate pre-screening tool in geriatric oncology. Crit Rev Oncol Hematol. 2010;75:243–8.
12. Kenis C, Decoster L, Van Puyvelde K, De Greve J, Conings G, Milisen K, et al. Performance of two geriatric screening tools in older patients with cancer. J Clin Oncol. 2014;32:19–26.
13. Baitar A, Van Fraeyenhove F, Vandebroek A, De Droogh E, Galdermans D, Mebis J, et al. Evaluation of the Groningen Frailty Indicator and the G8 questionnaire as screening tools for frailty in older patients with cancer. J Geriatr Oncol. 2013;4:32–8.
14. Overcash JA, Beckstead J, Extermann M, Cobb S. The abbreviated comprehensive geriatric assessment (aCGA): a retrospective analysis. Crit Rev Oncol Hematol. 2005;54:129–36.
15. Saliba D, Elliott M, Rubenstein LZ, Solomon DH, Young RT, Kamberg CJ, et al. The Vulnerable Elders Survey: a tool for identifying vulnerable older people in the community. J Am Geriatr Soc. 2001;49:1691–9.
16. Meldon SW, Mion LC, Palmer RM, Drew BL, Connor JT, Lewicki LJ, et al. A brief risk-stratification tool to predict repeat emergency department visits and hospitalizations in older patients discharged from the emergency department. Acad Emerg Med. 2003;10:224–32.
17. Kim JW, Kim SH, Kim YJ, Lee KW, Kim KI, Lee JS, et al. A novel geriatric screening tool in older patients with cancer: the Korean Cancer Study Group Geriatric Score (KG)-7. PLoS One. 2015;10:e0138304
18. Kim JW, Lee YG, Hwang IG, Song HS, Koh SJ, Ko YH, et al. Predicting cumulative incidence of adverse events in older patients with cancer undergoing first-line palliative chemotherapy: Korean Cancer Study Group (KCSG) multicentre prospective study. Br J Cancer. 2018;118:1169–75.
19. Jung HW, Kim JW, Han JW, Kim K, Kim JH, Kim KI, et al. Multidimensional geriatric prognostic index, based on a geriatric assessment, for long-term survival in older adults in Korea. PLoS One. 2016;11:e0147032
20. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83.
21. Mahoney FI, Barthel DW. Functional Evaluation: The Barthel Index. Md State Med J. 1965;14:61–5.
22. Won CW, Rho YG, SunWoo D, Lee YS. The validity and reliability of Korean Instrumental Activities of Daily Living (K-IADL) Scale. J Korean Geriatr Soc. 2002;6:273–80.
23. Podsiadlo D, Richardson S. The timed "Up & Go": a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39:142–8.
24. Lee DY, Lee JH, Ju YS, Lee KU, Kim KW, Jhoo JH, et al. The prevalence of dementia in older people in an urban population of Korea: the Seoul study. J Am Geriatr Soc. 2002;50:1233–9.
25. Bae JN, Cho MJ. Development of the Korean version of the Geriatric Depression Scale and its short form among elderly psychiatric patients. J Psychosom Res. 2004;57:297–305.
26. Vellas B, Guigoz Y, Garry PJ, Nourhashemi F, Bennahum D, Lauque S, et al. The Mini Nutritional Assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition. 1999;15:116–22.
27. Bellera CA, Rainfray M, Mathoulin-Pelissier S, Mertens C, Delva F, Fonck M, et al. Screening older cancer patients: first evaluation of the G-8 geriatric screening tool. Ann Oncol. 2012;23:2166–72.
|
|
|||||||||||||||||||||||||||||||||||||||||||||