Introduction
Epithelial ovarian cancer (EOC) is one of the leading causes of death in females with gynecological malignancies [1]. Most patients respond to primary treatment, and 75% of patients reach complete response. However, 40%-60% of all patients with EOC and 75% of those with advanced stage disease will eventually experience recurrence [2,3]. Accurate estimation of survival for patients with EOC is important because prognosis is a determinate of treatment aggressiveness tailored to the individual situation. Patients who experience recurrence after 6 months from the end of primary chemotherapy are classified as platinum sensitive, and currently, platinum sensitivity is considered an important factor for predicting survival outcomes [4]. Predicting platinum sensitivity in patients with EOC may play an important role in establishing treatment plans.
Previous studies demonstrated several biological markers as significant prognostic factors for oncologic outcomes after treatment. Laboratory systemic inflammatory response markers have been studied as prognostic factors in a variety of cancers [5-7]. Paraneoplastic lymphocytopenia, leukocytosis, and thrombocytosis are significant prognostic factors in many solid tumors. However, the level of contribution of each biological marker to oncological outcomes such as platinum sensitivity and survival in EOC is not fully understood.
This study was conducted to evaluate the clinical impact of pre-treatment complete blood cell count (CBC) including white blood cell (WBC) differential components as prognostic factors for platinum-sensitivity on EOC patients with primary debulking surgery (PDS) and to develop nomograms for platinum sensitivity, 3-year progression-free survival (PFS), and 5-year overall survival (OS) with prognostic CBC components and known prognostic clinical parameters.
Materials and Methods
1. Patients
After obtaining Institutional Review Board approval (IRB file No. 2015-06-092), data were collected from Samsung Medical Center for patients with EOC who were treated from January 2002 to December 2012. We identified 757 patients whose primary treatment consisted of PDS and adjuvant chemotherapy. Patients who underwent neo-adjuvant chemotherapy and interval debulking surgery, those who had a transfusion within 2 weeks prior to PDS, and patients with concurrent cancer other than ovarian cancer were excluded from the study. Patients who received intraperitoneal or dose-dense chemotherapy were not included in this study.
2. Treatment and follow-up
Standard primary surgical treatment consisted of hysterectomy, bilateral salpingo-oophorectomy, omentectomy, retroperitoneal (pelvic and para-aortic) lymphadenectomy, and any tumorectomy of metastatic lesions, if applicable. Peritoneal washing was routinely conducted. If any abnormalities were identified, peritoneal biopsies from different sites were performed. Early stage EOC patients who wanted fertility saving received fertility saving surgery with/without chemotherapy as primary treatment.
Within 2 weeks prior to PDS, patients were routinely required to undergo basic preoperative evaluation including complete blood count. After PDS, patients started the first cycle of platinum-based combination chemotherapy, which was repeated every 3 weeks for six cycles. Abdominopelvic computed tomography (CT) scan was routinely performed after first three cycles of chemotherapy and after six cycles of first-line treatment. Following primary treatment, patients were assessed by physical examination, CBC, and chemistry with serum tumor markers, including cancer antigen 125 (CA-125) measurements, every 3 months for the first 2 years and twice per year thereafter. Chest radiography and abdominopelvic CT scan (or alternatively abdominopelvic magnetic resonance imaging) were performed every 6 months for the first 3 years and every 12 months thereafter. Additional diagnostic procedures were performed according to specific clinical suspicions. If recurrence was suspected with symptoms or CA-125 elevation, additional imaging studies were performed. Recurrence may have been detected by imaging studies with or without CA-125 elevation. Response to chemotherapy was assessed and recorded according to the Response Evaluation Criteria in Solid Tumors (RECIST).
OS was defined as the time from diagnosis to the date of the patient’s death or loss to follow-up. PFS was defined as the time from diagnosis to the date of recurrence or loss to follow-up. Disease-free interval (DFI) was defined as the time from the end of primary treatment to the date of recurrence.
3. Statistical analysis
A multivariate logistic regression model with stepwise variable selection using Akaike’s information criterion (AIC) was employed to identify factors predictive of platinum sensitivity. A multivariate Cox regression model with stepwise variable selection using AIC was used to identify prognostic factors for OS and PFS. The proportional hazards assumption was assessed using the method proposed by Grambsch and Therneau [8] and the linearity assumption was checked using a penalized smoothing spline method. Two assumptions for neutrophil, lymphocyte, monocyte, platelet, and age were satisfied (S1 and S2 Tables). Prognostic factors identified by multivariate analysis were used to create a nomogram to predict platinum sensitivity, 3-year PFS, and 5-year OS. We validated each nomogram using 10-fold cross-validation (CV) [9]. We then constructed a receiver operating characteristic (ROC) curve for platinum-sensitivity and calculated the area under the curve (AUC). The optimal cut-off point for predicted probability was determined by maximizing the Youden index [10]. Time-dependent ROC curves were constructed using the Nearest Neighbor Estimation method for 3-year PFS and 5-year OS [11] and AUCs were calculated. Statistical analyses were performed using R 3.0.3 (Vienna, Austria; http://www.R-project.org).
Prognostic variables including age, histology, stage, tumor grade, residual disease after PDS, and preoperative CBC (hemoglobin, WBC differential [neutrophil count, lymphocyte count, monocyte count], and platelet count) were used in the analysis. Platinum sensitivity was included in analysis of the 5-year OS. Patients with non-serous carcinoma were combined and compared to those with serous carcinoma. Residual disease after PDS was categorized based on size (no residual, ≤ 1 cm, and > 1 cm). Platinum sensitivity was classified as platinum-resistant or platinum-sensitive (DFI ≤ 6 months, DFI > 6 months), or unknown for platinum sensitivity. Patients with insufficient observation time to determine platinum sensitivity, as well as those who did not receive platinum-based chemotherapy as primary treatment were classified as unknown for platinum sensitivity.
Results
Data from the records of a total of 757 patients with EOC who were treated at Samsung Medical Center were analyzed. Patient demographic and clinical characteristics are listed in Table 1. There were 430 cases of cancer recurrence and 276 cases of cancer-specific death with a median follow-up of 51 months (range, 4 to 156 months). The majority of patients had stage III (56.5%), grade 3 disease (70.9%) of serous type (63.8%). There was no residual disease in 277 patients (36.6%) in the whole cohort, nor in 22.2% (115/517) of those in advanced stage. Of all patients, 647 patients (85.5%) were platinum-sensitive.
Multivariate logistic regression analysis for platinum sensitivity and multivariate Cox modeling for PFS and OS were used to evaluate independent prognostic factors and estimate their effects for all patients. Stage and residual disease after PDS were significant predictors of OS, PFS, and platinum sensitivity upon multivariate analysis. In addition, some of the preoperative CBC components were found to be significant prognostic factors.
Analyses for platinum sensitivity are shown in S3 Table. Among the significant variables identified upon univariate analysis, preoperative lymphocyte count and monocyte count, stage, histology, and residual disease after PDS were significant predictors of platinum sensitivity. Platelet count, stage, grade, and residual disease after PDS appeared to be significant predictors of 3-year PFS upon multivariate analysis (S4 Table). Pretreatment neutrophil count, stage, residual disease after PDS, and platinum sensitivity were significant prognostic factors for 5-year OS (S5 Table).
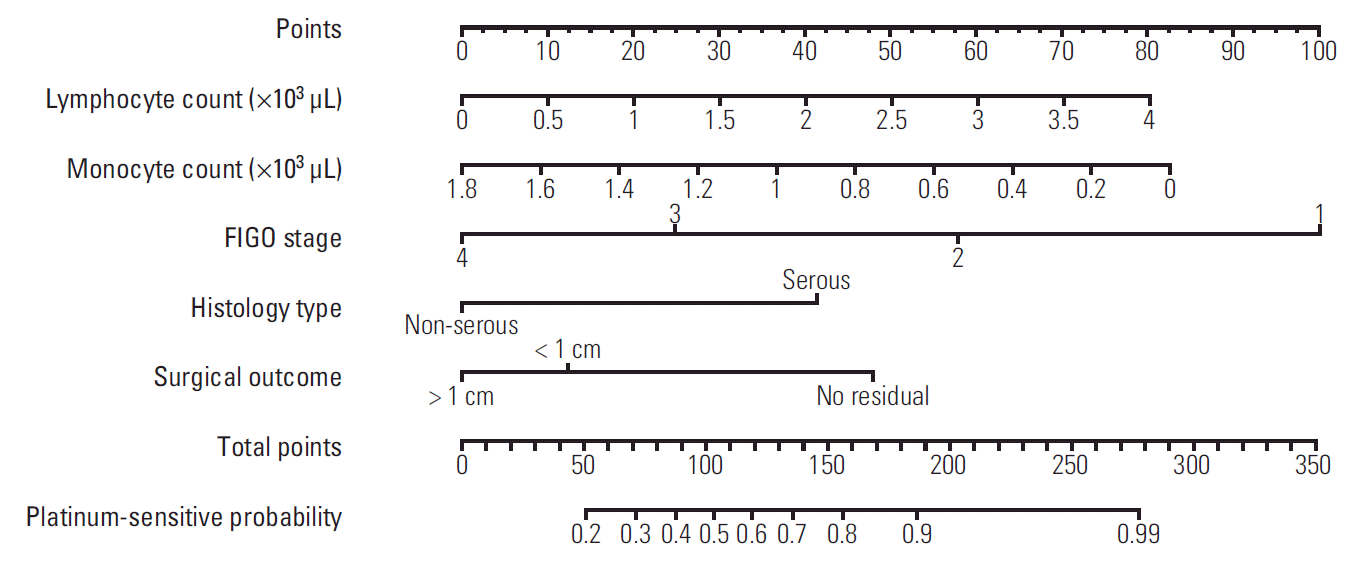
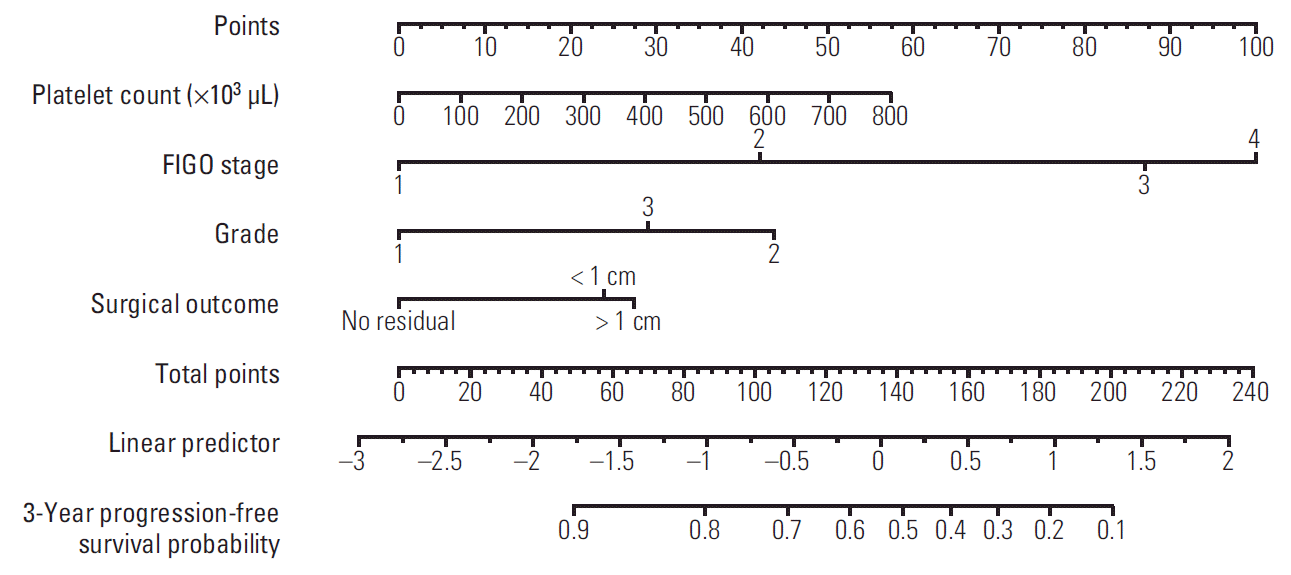
Nomograms were developed to account for the importance of each clinical prognostic variable. Nomograms were created using the multivariate logistic regression model for platinum sensitivity and multivariate Cox regression models for 3-year PFS and 5-year OS (Figs. 1-3). Use of the nomograms is described in the figure captions. For example, in the nomogram for platinum sensitivity, the user should find the patient’s stage on the Stage axis, then draw a straight line upward to the Points axis to determine how many points toward progression the patient receives for stage. This should be done again for the other axes, with the user drawing a straight line upward toward the Points axis each time. The points received for each predictor are then summed and the sum is found on the total points axis. The user should then draw a straight line down to the platinum sensitivity probability axis to find the patient’s probability of platinum sensitivity.
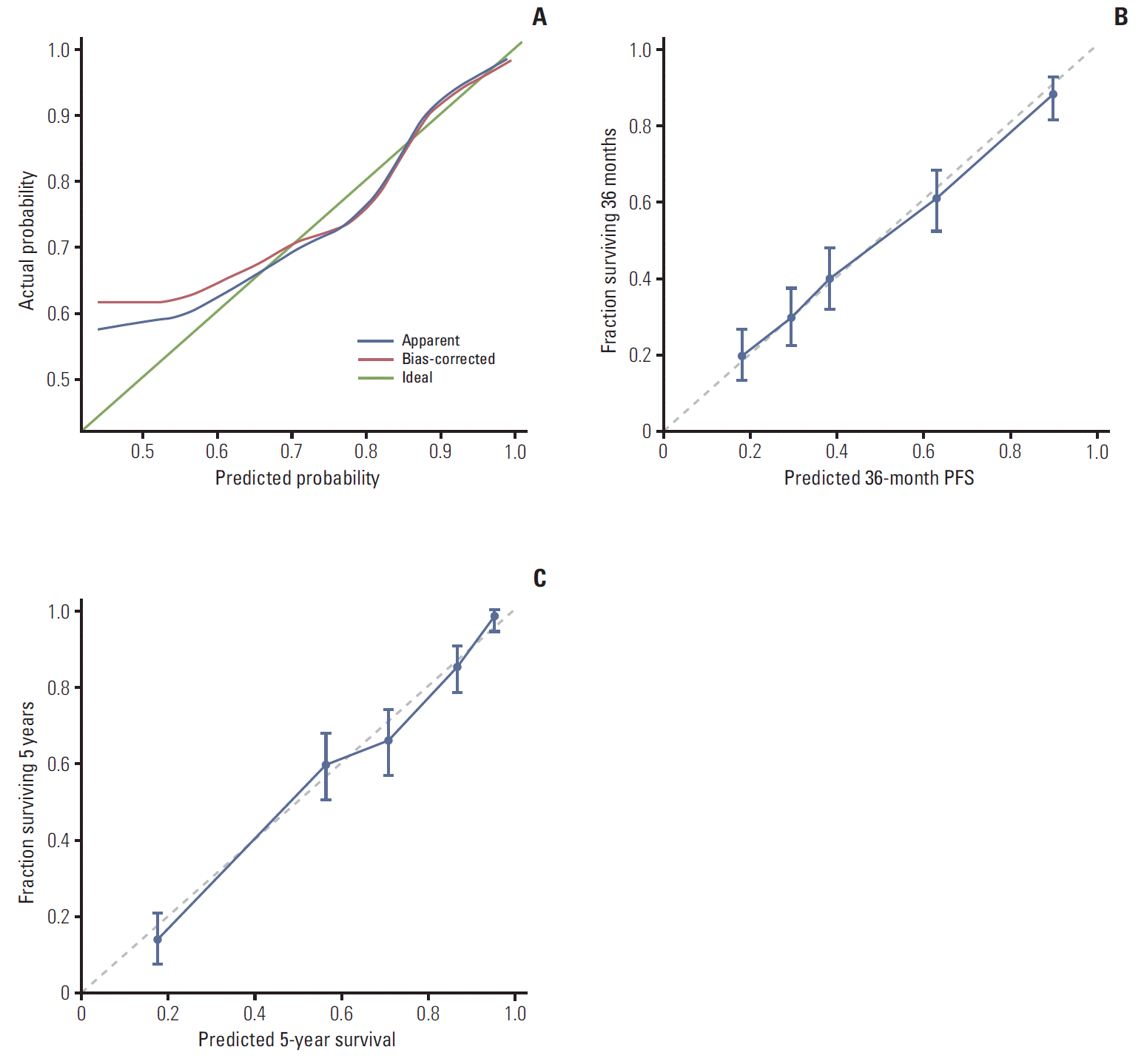
The AUCs of platinum sensitivity, 3-year PFS, and 5-year OS calculated using the 10-fold CV procedures were 0.7405, 0.8159, and 0.815, respectively. The optimal cut-off value of predicted probability for platinum sensitivity was 0.8594, and the sensitivity and specificity were 0.6318 and 0.8073, respectively. Calibration curves for the platinum sensitivity, 3-year PFS and 5-year OS nomograms are shown in Fig. 4. The nomograms for 3-year PFS and 5-year OS were well calibrated.
Discussion
In this study, we evaluated pre-treatment CBC as a prognostic factor for EOC treated with PDS and adjuvant chemotherapy and its impact on prognosis. We found that platelet count, neutrophil count, monocyte count, and lymphocyte count were significantly associated with platinum sensitivity, PFS, and OS. Other common prognostic factors identified in our study included stage and residual disease after PDS. Therefore, we sought to develop nomograms that would include these prognostic factors for platinum sensitivity, PFS, and OS. The nomograms were validated for platinum sensitivity, 3-year PFS, and 5-year OS.
There are number of risk factors associated with survival in EOC. Most of the previous risk analyses only provide information regarding individual risk factors based on univariate and multivariate analyses. Traditional risk stratification strategies assign all risk factors the same weight, which may cause bias. In contrast, a nomogram provides parametric information for end-point prediction by integrating multiple weighted risk factors. After summing all of the points for each factor, results can be translated into information regarding survival. This information can facilitate discussion between the physician and the patient and guide clinical care. Nomograms have been constructed to predict various clinical end points for patients with different types of cancer [12-14]. A nomogram should theoretically be more specific to each individual patient and thus able to predict specific clinical endpoints more accurately.
Paraneoplastic lymphocytopenia, leukocytosis, and thrombocytosis are well-known prognostic factors for many solid tumors. Various studies explaining the prognostic significance of inflammatory markers from peripheral blood including components from pretreatment CBCs have been reported [15-17]. Cancer cells secrete cytokines such as interleukin 6, which directly and indirectly stimulate platelet production in tumor cells, thereby enhancing proliferation and metastasis [17]. Tumors are known to induce neutrophilic differentiation and stimulate angiogenesis and cell proliferation by producing chemokines, cytokines, and prostaglandins [18]. Previous reports demonstrated that a higher neutrophil count and lower lymphocyte count predict poorer survival in EOC [7,16]. Lymphocytes showed greater decreases in patients with higher stage, ascites, and residual cancer after PDS in EOC [19]. Moreover, significantly lower lymphocyte counts and higher neutrophil counts were associated with greater tumor grade, advanced stage, and presence of ascites [20]. These associations between WBC differential counts and tumor aggressiveness may partially explain the correlation with survival outcomes. A recent study demonstrated that elevated peripheral blood monocyte count was associated with worse OS, and that monocytes are associated with increased adrenergic signaling via monocyte chemotactic protein 1, which facilitates tumor progression in EOC [21,22]. Despite evidence that factors from WBC differential counts are predictors of prognosis for different cancers including EOC, larger studies with greater detail on patient profiles, tumor features, and treatment will be necessary to demonstrate that these factors are truly independent predictors.
To the best of our knowledge, this is the first study presenting prognostic nomograms for the endpoint of platinum sensitivity with pre-treatment CBC including WBC differential counts. Previous reports of survival models in EOC have been based on variables such as stage, residual disease, grade, histology, age, and performance status [23-25]. Furthermore, a number of studies demonstrated laboratory markers from routine testing, such as CBC, as prognostic variables [13,26-28]. Our study demonstrated that, in addition to the previously published prognostic variables for EOC, WBC differential counts (lymphocyte, monocyte, and neutrophil counts) of pre-treatment CBC may be considered prognostic factors for platinum sensitivity and survival in EOC. We expected minimal bias associated with using pretreatment CBC since it is commonly performed in a majority of institutions and gives objective values. Based on patient clinicopathologic information and components of pretreatment CBC, the nomogram could be used to estimate platinum sensitivity, 3-year PFS, and 5-year OS rates. With nomograms applied to clinical management, clinicians will be able to apply different strategies for subsequent systemic anti-tumor therapy and follow-up interval based on the platinum sensitivity in EOC patients with PDS.
It should be noted that our study had several limitations. Specifically, its retrospective, single-center nature may have resulted in unmeasured confounding factors. When analyzing variables, not all known prognostic factors identified in other prognostic models, such as the volume of ascites and performance status [13,23,25,29], were included because of a substantial lack of data in the medical records. We also did not have sufficient data describing specific circumstances for each patient included in the unknown platinum sensitivity category to explain the reason for the lower hazard ratio. Moreover, the fact that systemic conditions other than cancer (e.g., inflammatory disease or infection status) may have affected the pretreatment CBC status should not be overlooked. Finally, preoperative comorbidities were not assessed in this analysis.
Although previous studies have shown that factors from WBC differential counts are predictors of prognosis for EOC, the mechanism of results cannot be clearly explained. In addition, our finding that grade reversely affected the survival outcome is difficult to explain. In multivariate analysis for 3-year PFS, grade 3 showed a lower hazard ratio than grade 2, contrary to univariate analysis. In a previous study, no difference was found in clinical outcomes between grade 2 and 3 of serous ovarian cancer, and in serous ovarian cancer, a 2-tier grade system (high grade vs. low grade) was reportedly more accurate for predicting clinical outcomes than a International Federation of Gynecology and Obstetrics (FIGO) 3-tier (grade 1, 2, and 3) system [30]. In our study, we used a FIGO 3-tier system because we included not only the results of serous type, but also other histologic types. It is possible that the lack of a survival difference between grade 2 and 3 serous types might have affected the results of the hazard ratio for grade upon multivariate analysis.
In our study, information regarding dose, schedule, and toxicities of the adjuvant chemotherapy or surgical complications, which may be associated with survival, is lacking because of the deficit of associated information owing to this study’s retrospective nature. A larger number of patient profiles and detailed information will be needed for definite results in the future. Although internally validated, the nomogram needs to be externally validated before it can be generally accepted for clinical application.
Conclusion
In summary, our multivariate analysis identified WBC differential counts on pre-treatment CBC (neutrophil, monocyte, and lymphocyte counts) as prognostic factors in addition to stage and residual disease after PDS in patients with EOC that was primarily treated with PDS and adjuvant chemotherapy. These prognostic factors allowed development of nomograms predicting platinum sensitivity, 3-year PFS, and 5-year OS. Such nomograms could be used to better estimate outcomes for individual patients.