Introduction
Toxocariasis is the clinical term for infection in the human host by either Toxocara canis or Toxocara cati. Both of these organisms are ascarid nematodes in the order Ascaridida, superfamily Ascaridoidea, family Toxocaridae. Visceral larva migrans (VLM) syndrome, a clinical manifestation of systemic toxocariasis, was first described by Beaver et al. in New Orleans in 1952 [1]. This syndrome is caused by migration of the larvae of T. canis, T. cati, and various organs may be involved; the liver, lung, and eye are most commonly affected [2-4].
VLM syndrome is found throughout the world, even in developed countries. Cats and dogs are the definitive hosts of T. canis and T. cati, and human infection is caused by swallowing the eggs shed by these animals. In the small bowel, the eggs release larvae, which penetrate the wall of the intestine and from there can enter the portal venous circulation and hepatic parenchyma [5]. In the liver, they move through the parenchyma and cause necrosis and interstitial edema [5]. The larvae cause disease not only in the liver but also in the heart and brain [6,7]. Lesions caused by T. canis or T. cati infection can be observed on imaging studies, such as those obtained by computed tomography (CT) or magnetic resonance imaging, as mass-like lesions that may confuse clinicians [8]. We report on a case of VLM with involvement of the bladder and liver.
Case Report
A 62-year-old Korean woman who was a homemaker was referred to Korea University Medical Center from a local hospital for evaluation of abnormal findings of bladder and liver mass lesions on CT images. The patient had experienced urinary urgency and frequency for the past three months. Therefore, she visited the local hospital and underwent abdominal ultrasound and CT. Imaging studies showed thickening of the urinary bladder wall and several nodular lesions on the liver.
The patient had undergone bladder surgery for treatment of stress incontinence four years previously, and she did not suffer from any urinary symptoms after surgery. She had diagnosed with asthma 20 years before and was taking montelukast, a beta-2 agonist (Ventolin, GlaxoSmithKline, Madrid, Spain), and an inhaled combination corticosteroid and beta-2 agonist (Seretide, GlaxoSmithKline, London, UK). She had no other prior medical history. She had not been living with animals and had no history of eating raw meat or any recent travel history.
The patient did not complain of any other symptoms (i.e., visual symptoms or respiratory symptoms, chest pain, nausea, vomiting, melena, or hematochezia) except for urinary frequency and urgency. She denied any history of fever, abdominal pain, or hematuria. Her weight had not changed.
On physical examination, the patient appeared well. Her sclera and skin were anicteric, and heart and lung examination revealed no abnormal findings. Abdominal examination revealed no palpable hepatic or splenic mass, and she did not complain of low abdominal tenderness or costovertebral angle tenderness. There was no edema of either lower extremity. Neurologic examination revealed no focal deficits. There was no cervical, axillary, or inguinal lymphadenopathy.
Initial laboratory studies showed no abnormal urinalysis findings. Her white blood cell count was 5,660/μL, and showed 21% eosinophil granulocytes, with an absolute value of 1,188/μL. Her serum electrolyte concentrations were within the normal ranges. Liver function tests showed that her aspartate aminotransferase, alanine aminotransferase, and bilirubin levels were within normal limits, but her protein level was high (11.2 g/dL), with relative hypoalbuminemia (3.1 g/dL). Serum tests for the presence of viral hepatitis, human immunodeficiency virus, and syphilis were all negative. The circulating levels of various tumor markers (carcinoembryonic antigen, carbohydrate antigen [CA] 19-9, alpha-fetoprotein, CA 125, and CA 15-3) were within normal limits. In urine analysis, there were no red blood cells or white blood cells, and no organism was found in the urine culture.
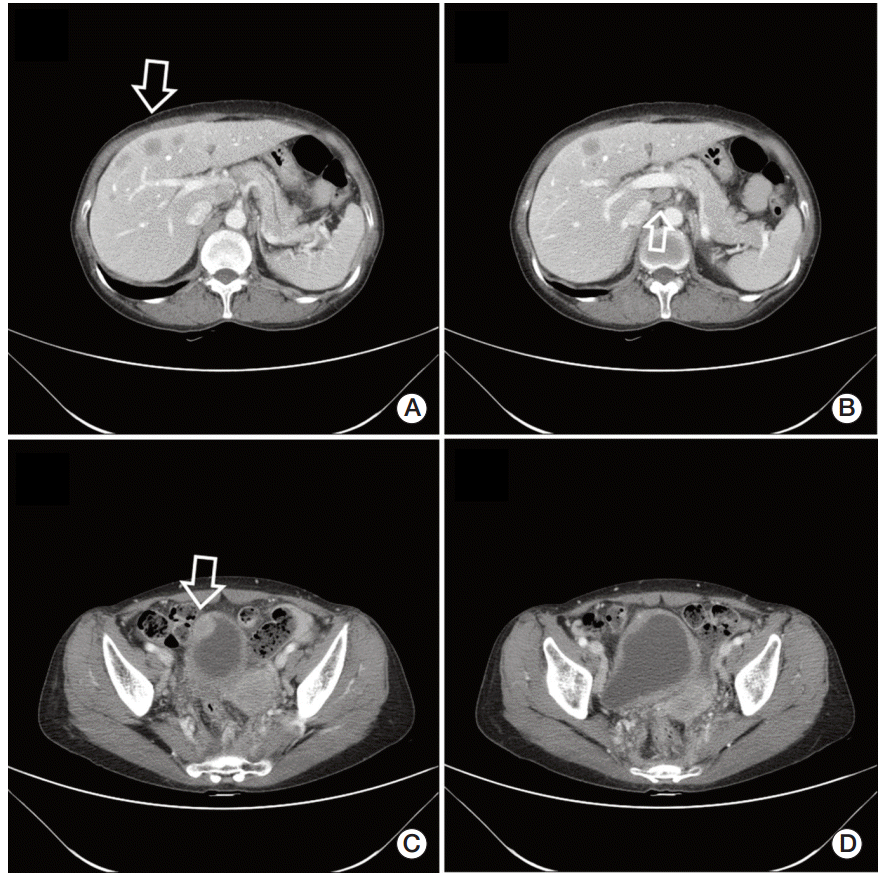
At Korea University Medical Center, abdominal CT was repeated and detected a nodular enhancing lesion in the right anterior wall of the bladder with suspicious perivesicular invasion. In addition, multiple ill-defined hypodense mass lesions reminiscent of metastasis were observed on the liver (Fig. 1A-D). Multiple enlarged lymph nodes were also noted in the external iliac, inguinal, aortocaval, paraaortic, portocaval, right pericardiac, and subphrenic areas.
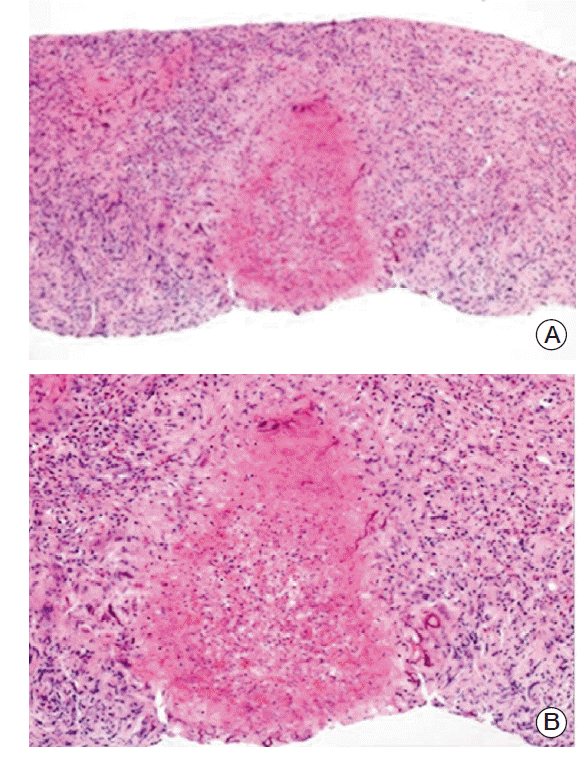
Because malignancy was suspected, gastrofiberscopy and colonoscopy were performed to search for the origins of the masses. No suspicious malignant lesion was observed. We referred the patient to an urologist for bladder examination. Cystoscopy was performed and showed no evidence of cancer on the bladder wall. Urine cytology was repeated three times and revealed no malignant cells. The urologist recommended a bladder biopsy to confirm eosinophilic cystitis, but the patient refused. For further evaluation of the liver findings, a core biopsy of a liver mass was taken. Histologic examination of the biopsy sample revealed the presence of eosinophilic necrotizing granuloma. No larvae were found in the tissue samples, and the other liver tissues showed non-specific reactive changes (Fig. 2A and B). These findings were suggestive of parasitic granulomas or reminiscent of larva migrans lesions. We tested for anti-Toxocara antibodies using an enzyme-linked immunosorbent assay (ELISA), but the result was negative. However, anti-Toxocara IgG was detected by western blotting showing two or more low molecular weight bands and intermediate molecular weight band in the ranges of 24-50 kDa (Toxocara western blot IgG, Ldbio Diagnostics, Lyon, France) (Fig. 3). Because the patient had urinary symptoms, we prescribed prednisolone at 15 mg/day for 21 days. The urinary symptoms resolved after prednisolone treatment. Abdominal CT scan was repeated after three months, and sizes of multiple liver masses and intraabdominal lymph nodes were decreased and bladder wall thickening had disappeared during the interval.
Discussion
The patient described in this report was referred to Korea University Medical Center with urinary symptoms, peripheral eosinophilia, diffuse bladder wall thickening, and multiple hepatic mass lesions on CT images. First, we considered bladder cancer with liver metastasis as a differential diagnosis. However, urologic examination and liver biopsy showed no malignant lesion. Based on the liver histology, we next considered inflammatory or non-neoplastic bladder lesions such as eosinophilic cystitis. The urologist recommended bladder biopsy to confirm the diagnosis; however, because the patient refused biopsy, we were unable to perform histologic examination. The diagnosis of VLM syndrome was confirmed via histopathologic examination of the liver and detection of anti-Toxocara antibodies by western blotting.
Several cases in which the imaging findings of VLM syndrome mimicked malignant lesions have been reported [4,8]. In addition, owing to its non-specific appearance on CT images, hepatic toxocariasis can often be mistaken for other conditions. Differential diagnoses for multiple low-density liver nodules include microabscesses, other granulomatous diseases (i.e., sarcoidosis) as well as hepatocellular carcinoma or liver metastasis [3]. Hepatic lesions associated with VLM usually appear as multiple, ill-defined, oval-shaped, low-attenuating nodules that are best seen in the portal phase on CT images. These findings seem to be somewhat characteristic of eosinophilic infiltration or granuloma and can thus distinguish such processes from malignant lesions [8]. Most of all, histologic confirmation is the most important process for the differential diagnosis.
Unlike hepatic toxocariasis, VLM syndrome involving the bladder has rarely been reported. In this case, the patient showed mass lesions on both liver and bladder and the biopsied hepatic lesion was described in the pathology report only as a non-specific lesion associated with an eosinophilic infiltrate, with no cancer cells. In addition, the patient showed peripheral eosinophilia and history of allergy. Therefore, we considered inflammatory or non-neoplastic conditions for bladder lesions as a differential diagnosis. There are several conditions in this category, including pseudosarcomatous fibromyxoid tumor, eosinophilic cystitis, bladder endometriosis, and various bladder infection with tuberculosis or schistosomiasis [9]. Among these, we strongly suspected eosinophilic cystitis because the patient described in this report resembled previously reported cases in having urinary symptoms, peripheral eosinophilia, a history of allergy, and imaging findings of diffuse bladder wall thickening and focal mass-like lesions [10]. Because the patient did not show cyclic hematuria or pyuria, we could exclude other disease like bladder endometriosis or various bladder infections.
If a finding of eosinophilic granulomas by tissue biopsy is strongly suggestive of VLM, as in this case, then the causative organism should be considered. However, samples rarely contain intact Toxocara larvae and additional tests are required to obtain a diagnosis. There are two types of tests available for the immunodiagnosis of toxocariasis: ELISA and western blotting. Both methods use T. canis excretory-secretory antigens for detection of anti-Toxocara antibodies. An ELISA that detects antibodies against a second-stage larval excretory-secretory antigen has been one of the best indirect methods for diagnosis, with a sensitivity of 78-91%and specificity of 86-93% [11]. However, ELISA might not be specific due to the cross-reactivity of T. canis excretory-secretory antigens with antibodies against parasites of the genera Anisakis or Ascaris [12]. Therefore, western blot testing after a negative or even a positive initial ELISA result has recently been recommended for more exact evaluation [13].
As in this case, lower molecular weight bands demonstrated a high level of specificity while higher molecular weight bands did not because these are suggestive of cross-reactions with other helminthes in western blot testing [14].
Most cases of toxocariasis are benign and self-limiting, and medical treatment is unnecessary for even prolonged mild cases [3]. Albendazole is the treatment of choice in more severe cases, and a 14-day or 5-day treatment course of albendazole (10 mg/kg daily for 14 days or 15 mg/kg daily for 5 days) is recommended [15]. In addition, symptomatic treatment, including administration of corticosteroids, can be helpful for suppressing the intense allergic manifestations of the infection. In this case, because the patient complained of urinary symptoms and showed peripheral eosinophilia and inflammatory eosinophilic bladder lesions were suggestive, we prescribed corticosteroid, which was helpful for suppression of symptoms and regression of mass lesions.
In conclusion, we have reported a case of VLM syndrome causing bladder wall thickening and multiple hepatic masses mimicking malignancy. In cases where the imaging pattern of the mass is atypical of malignancy and peripheral eosinophilia is present, VLM syndrome should be included asa differential diagnosis.