INTRODUCTION
Despite its declining incidence in the western countries, gastric cancer remains among the most frequently diagnosed cancer worldwide and the most common causes of cancer death (1). In Korea, gastric cancer still ranks as the most common cancer (2). Systemic chemotherapy is widely accepted as palliative treatment for patients with advanced or metastatic disease, and this can lead to objective responses, improvement of the quality of life and prolonged survival when compared to the best supportive care (3~5). Many combination chemotherapy regimens have been studied in randomized trials, but the survival benefit was limited with the median survival consistently below 10 months (6~9). The results of using ECF (epirubicin, cisplatin and a continuous infusion of 5-FU) and DCF (docetaxel, cisplatin and a continuous infusion of 5-FU) were superior to those achieved with FAMTX (5-FU, doxorubicin and high-dose methotrexate) and CF (cisplatin and a continuous infusion of 5-FU) respectively, but substantial treatment-related toxicities were observed with these regimens. Oxaliplatin is a third-generation diaminocyclohexane platinum compound that has a wide range of antitumor activities and it appears to have a better toxicity profile compared to cisplatin. For treating advanced gastric cancer, various combinations of oxaliplatin and 5-FU have shown considerable antitumor activity in several recent phase II studies (10~14). As far as toxicity is concerned, FOLFOX-6 caused significant toxicities, including myelosuppression and peripheral neuropathy (10). When compared to FOLFOX-6, both weekly and biweekly reduced-dose combinations of oxaliplatin/5-FU/ FA without 5-FU bolus showed a more favorable toxicity profile with lower rates of peripheral neuropathy and myelosuppression (11~13). Further, the preliminary results of a study conducted by the Korean Cancer Study Group (KCSG) to assess the activity and toxicity of a biweekly combination of oxaliplatin/5-FU/FA without 5-FU bolus (oxaliplatin 100 mg/m2 2-hour infusion, FA 100 mg/m2 2-hour infusion, 5-FU 2,400 mg/m2 46-hour continuous infusion) were promising with a 52% tumor control rate and an acceptable toxicity profile (14).
Gastric cancer rarely occurs before the age of 40 years, and its incidence increases thereafter; this peaks in the 6th and 7th decades. Although the majority of gastric cancer patients are elderly, patients older than 65 years have been often excluded from or underrepresented in the study populations of combination chemotherapy trials (15,16). Consequently, little data is available on the efficacy and safety of chemotherapy regimens in elderly gastric cancer patients.
On this basis, we investigated a biweekly protocol of oxaliplatin/ 5-FU/FA (oxaliplatin 100 mg/m2 2-hour infusion , FA 100 mg/m2 2-hour infusion, 5-FU 2,400 mg/m2 46-hour continuous infusion) for treating elderly patients with metastatic or recurrent gastric cancer.
MATERIALS AND METHODS
1) Patient eligibility
The eligibility criteria of the patients included (1) pathologically confirmed, metastatic or recurrent adenocarcinoma of the stomach (except carcinoma of the esophagogastric junction) and a patient age >65 years old, (2) at least one measurable lesion (3) no prior chemotherapy except postoperative adjuvant chemotherapy that was received more than 6 months before entry into the study, (4) an Eastern Cooperative Oncology Group (ECOG) performance status 0~2, and (5) adequate baseline hematologic function [an absolute neutrophil count (ANC) ≥1.5×109/L, a platelet count ≥100×109/L], an adequate hepatic function [serum bilirubin ≤ 1.25×upper normal limit (UNL), serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) ≤ 2.5×UNL, serum alkaline phosphatase ≤5.0×UNL], and adequate renal function [serum creatinine ≤ 1.5 mg/dl]. The exclusion criteria included (1) pre-existing peripheral neuropathy, (2) concurrent or prior malignancy except curatively resected cervical carcinoma in situ or squamous cell carcinoma of the skin, (3) brain metastases (4) active infection, and (5) concurrent treatments that interfered with the study's evaluation.
2) Study design and dose modifications
(1) Chemotherapy protocol
Oxaliplatin 100 mg/m2 and folinic acid 100 mg/m2 were given as a 2-hour intravenous infusion followed by 5-FU 2,400 mg/m2 as a 46-hour continuous infusion. The cycles were repeated every 2 weeks and treatment was continued until disease progression, unacceptable toxicity, the patient's refusal or the physician's decision. Antiemetic prophylaxis was given according to a local protocol. Granulocyte colony-stimulating factor (G-CSF) was not routinely used in the present study.
(2) Treatment delays and dose modifications
Each chemotherapy cycle was started only if the ANC and platelet counts on the day of treatment were ≥1.5×109/L and ≥100× 109/L, respectively. In the case of any grade 2 or more toxicity, except alopecia, chemotherapy was delayed a week and then restarted after full recovery. A reduction of 25% in all drugs dosing was applied to grade 3/4 thrombocytopenia, grade 4 neutropenia, grade 3/4 febrile neutropenia and grade 3 non- hematologic toxicity in the preceding cycle of chemotherapy. Patients with unresolved grade 2 or more toxicity after two consecutive treatment delays or those patients experiencing grade 4 non-hematologic toxicity, except alopecia, went off the study.
3) Response and toxicity assessment
The response to treatment was assessed according to the RECIST criteria (17). Computed tomography (CT) scans of the measurable lesions were carried out within 3 weeks before the start of the treatment. After every three cycle of chemotherapy, the patients underwent follow-up CT scans for assessment of response. Patients were considered evaluable for response if they had received at least three cycles of chemotherapy with at least one follow-up tumor assessment or they had early disease progression. Nonevaluable patients were included into the intention-to-treat analyses, but they reported as being not evaluable. For cases where treatment was discontinued before tumor progression, CT scans were repeated every 3 months. A complete history taking, physical examination, complete blood cell counts with the differential count and the serum biochemistry were performed at baseline. Complete blood cell counts with the differential count and serum biochemistry determinations were repeated just before the start of each chemotherapy cycle, and the toxicity was graded according to the National Cancer Institute Common Toxicity Criteria (NCI-CTC, version 3.0).
RESULTS
1) Patient characteristics
Between April 2004 and October 2006, we enrolled a total of 37 elderly patients with a median age of 72 years (range: 67~79) from Seoul Municipal Boramae Hospital and Seoul National University Bundang Hospital. The patient baseline characteristics are listed in Table 1. There were 23 men and 14 women, and 13 patients (35%) had an ECOG performance status of 2. Twenty-six patients (70%) had primary metastatic disease and 11 patients (30%) had recurrent disease. Lymph nodes, liver and peritoneum were the most common metastatic sites. The metastasis involved two organs in 15 patients (41%) and three or more organs in 10 patients (27%). Among the 11 patients who had previously received curative surgery, six patients received adjuvant chemotherapy after surgery with 5-FU-containing regimens.
2) Tumor response
Among the 37 patients who received the chemotherapy, the tumor response was evaluable according to the RECIST criteria in 34 patients. Three patients were excluded from the response analysis because they did not complete three cycles of chemotherapy and they did not show early progression; two patients refused continuation of treatment after the second cycle and one patient refused continuation of treatment after one cycle. Of the 34 evaluable patients, none achieved a complete response (CR) and 14 achieved a partial response (PR), resulting in an overall response rate of 41.2%. Twelve patients (35.3%) had stable disease and eight patients (23.5%) had progressive disease.
3) Toxicity and dose intensity
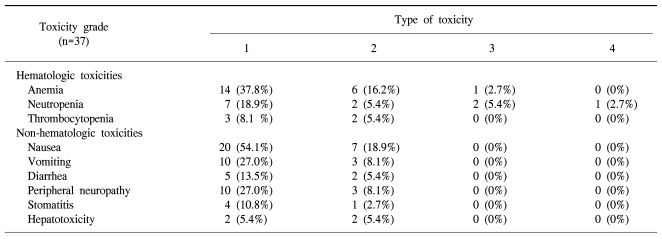
The incidences of main toxicities per patient are summarized in Table 2. Thirty-seven patients received a total of 214 treatment cycles. The median number of cycles administered was six (range: 1~12 cycles). The most common hematologic toxicity was anemia (56.7%) with one case of grade 3 anemia. Neutropenia occurred in 32.4% of the patients and grade 3/4 neutropenia was observed in 8.1% of the patients. Non patient experienced febrile neutropenia. There was no grade 3/4 thrombocytopenia. The non-hematologic toxicities were mild to moderate and no grade 3/4 toxicity was observed. These non-hematologic toxicities consisted mainly of nausea, vomiting and peripheral neuropathy. Peripheral neuropathy occurred in 35.1% of the patients and all cases were grade 1/2. Transiently impaired liver function was observed in four patients (10.8%). There was no treatment-related mortality. The relative dose intensities of oxaliplatin and 5-FU were 90.6% and 89.4%, respectively. The median cumulative dose of oxaliplatin was 567.5 mg/m2.
4) Survival
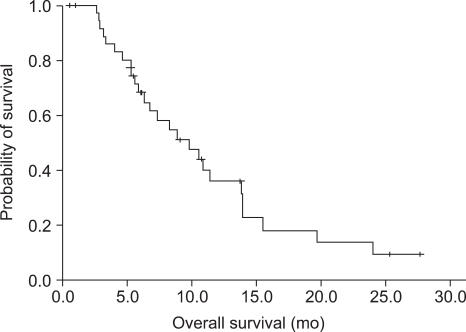
The median follow-up duration was 12.6 months, and the median time to progression (TTP) was 5.7 months [95% confidence interval (CI): 4.2~6.3 months] (Fig. 1). The median overall survival (OS) was 9.8 months [95% CI: 4.4-12.0 months] (Fig. 2). Second-line chemotherapy was administered to 16 patients (43%); five patients received capecitabine, five patients recieved docetaxel (±cisplatin), four patients received paclitaxel (±cisplatin), one patient received S-1 and one patient received irinotecan/5-FU/FA (FOLFIRI). At the time of analysis, 24 patients had died. The causes of deaths were progressive disease in 21 patients, severe infection in two patients and brain hemorrhage in one patient.
DISCUSSION
Although elderly patients comprise a significant proportion of those patients with gastric cancer, and the elderly population is increasing (18), any large trials of palliative chemotherapy for elderly patients are almost lacking. Several phase II trials have been reported so far (19~24). In these trials, the eligibility criteria were quite stringent and a proportion of elderly patients could not be enrolled, and so the number of patients enrolled in most of these trials did not exceed 30 in each of them. Consequently, the results in terms of efficacy and safety were heterogeneous, but in general, the tumor control rate was promising and no treatment-related mortality was observed. The largest phase II trial reported by Graziano et al. investigated the weekly combination of cisplatin, 5-FU and folinic acid (PLF) in 58 elderly patients (>65 years old) with advanced gastric cancer (24). The regimen showed a promising tumor control rate (43%) and it was safely delivered to nonfrail elderly patients on an outpatient basis. About half of the patients who experienced grade 2/3 (35%/17%) neutropenia received filgrastim, which was maintained until the end of the treatment program according to the treatment protocol. No grade 3/4 non-hematologic toxicity was observed, except for one case (2%) of grade 3 fatigue.
In the present study, we assessed the clinical efficacy and safety of a biweekly oxaliplatin/5-FU/FA regimen (oxaliplatin 100 mg/m2 2-hour infusion, FA 100 mg/m2 2-hour infusion, 5-FU 2,400 mg/m2 46-hour continuous infusion) for elderly patients with metastatic or recurrent gastric cancer. This regimen showed a response rate of 41.2%, a median time to progression of 5.7 months and a median overall survival of 9.8 months. These results are comparable to those studies of other chemotherapeutic agents like FAMTX, ECF, EFL and oxaliplatin-based combinations (6~14), in which the majority of patients were young and had a good performance status. We confirm the activity of this biweekly oxaliplatin/5-FU/FA regimen in our elderly patients with a median age of 72 years (range: 67~79), including 13 patients (35%) with an ECOG performance status of 2. In terms of toxicity, our regimen showed an acceptable toxicity profile and it was well-tolerated. Neutropenia occurred in 32.4% of the patients, and grade 3/4 neutropenia was observed in 8.1%. FOLFOX-6, which included a 5-FU bolus on day 1, caused grade 3/4 neutropenia in 38% of the patients with an 11% rate of febrile neutropenia (10). As already observed for several oxaliplatin-based combinations (11~13), avoiding the 5-FU bolus administration has led to a dramatic reduction of hematologic toxicity. In our study, the 5-FU bolus was omitted, and consequently, the rate of grade 3/4 neutropenia was below 10% and any febrile neutropenia was not observed. In general, higher rates of neutropenia and leucopenia were reported with using other chemotherapeutic regimens (6~9), including a weekly PLF regimen for elderly gastric cancer patients (24). The oxaliplatin-induced neurotoxicity was also mild and this occurred in 35.1% of the patients and there was no grade 3/4 neuropathy. This was probably related to the low median cumulative dose of oxaliplatin (567.5 mg/m2) we used in our study, with a median of six administered cycles of chemotherapy. More recently, Santini et al. investigated a weekly combination of oxaliplatin/5-FU/FA (OXALF) for the treatment of 42 elderly patients (>70 years old) with advanced gastric cancer (25). The regimen consisted of weekly oxaliplatin 40 mg/m2, 5-FU 500 mg/m2 and folinic acid 250 mg/m2, and 5-FU/FA was administered as a bolus instead of a 24-h infusion because of the ease of administration. The overall response rate was 45.2%, the median time to progression was 5.0 months and the median overall survival was 9.0 months. These results are comparable to those of our regimen. The toxicity was generally mild with few grade 3 toxicities and no grade 4 toxicities. Neutropenia was reported in 9.6% of the patients, including two (4.8%) cases of grade 3 toxicity. Peripheral neuropathy occurred in 33.3% of the patients, including one (2.4%) case of grade 3 toxicity with the median cumulative dose of oxaliplatin of 480 mg/m2. The results of the weekly OXALF and our regimen confirm the activity and safety of various combinations of oxaliplatin/5-FU/FA in elderly patients with advanced gastric cancer.
CONCLUSIONS
In conclusion, the biweekly oxaliplatin/5-FU/FA regimen administered in this study was found to be well-tolerated, and it provided similar survival benefit, for the elderly patients with metastatic or recurrent gastric cancer. Therefore, it may be a valuable treatment option for these patients.