Validation of Korean Version of the COmprehensive Score for Financial Toxicity (COST) among Breast Cancer Survivors
Article information
Abstract
Purpose
Little is known about the impact of financial toxicity in disease-free breast cancer survivors. We aim to validate the COmprehensive Score for financial Toxicity in Korean (COST-K) and evaluate financial toxicity among disease-free breast cancer survivors.
Materials and Methods
We conducted linguistic validation following a standardized methodology recommended by Functional Assessment of Chronic Illness Therapy multilingual translation (FACITtrans). For psychometric validation, we conducted a cross-sectional survey with 4,297 disease-free breast cancer survivors at a tertiary hospital in Seoul, Korea between November 2018 and April 2019. Survivors were asked to complete the COST-K and European Organization for Research and Treatment of Cancer Quality of Life Core 30 (EORTC QLQ-C30) questionnaires. The test-retest reliability, internal consistency, and validity of the COST-K were assessed using standard scale construction techniques.
Results
The COST-K demonstrated good internal consistency, with a Cronbach’s α of 0.81. The test-retest analysis revealed an intraclass correlation coefficient of 0.78. The COST-K had moderate correlation (r=−0.60) with the financial difficulty item of the EORTC QLQ-C30 and week correlation with the items on acute and chronic symptom burdens (nausea/vomiting, −0.18; constipation, −0.14; diarrhea, −0.14), showing good convergent and divergent validity. The median COST-K was 27 (range, 0 to 44; mean±standard deivation [SD], 27.1±7.5) and about 30% and 5% of cancer survivors experienced mild and severe financial toxicity, respectively. Younger age, lower education, lower household income was associated with higher financial toxicity.
Conclusion
The COST-K is a valid and reliable instrument for measuring financial toxicity in disease-free breast cancer survivors. Considering its impact on the health-related quality of life, more studies need to be conducted to evaluate financial toxicity in cancer survivors and design interventions.
Introduction
Cancer is one of the costliest medical conditions to treat, and individuals with cancer are at a higher risk of experiencing financial problems than those without cancer [1–3]. Financial toxicity is defined as the adverse economic consequence of medical treatment. The objective financial consequences of cancer as well as subjective financial distress are broadly termed as “financial toxicity.” However, studies on the individual financial effects of cancer treatment have largely focused on quantifying objective financial burdens, such as out-of-pocket expenses [4]. Recently, there has been increasing interest in subjective financial distress due to cancer treatment because it could lead to adverse health outcomes, both physical and mental [5–7].
To measure financial toxicity, the COmprehensive Score for financial Toxicity (COST) was developed and validated with patients with cancer in the United States [8,9]. This instrument contains 11 questions that are categorized into three themes: (1) two resource items, (2) one financial item, and (3) eight affect items specifically asking objective financial burden and subjective financial distress due to cancer care. The COST had involved patients with advanced and metastatic cancer undergoing active cancer treatment when it was developed [10–12]. Recently, there has been an increasing awareness about long-term financial burden and distress among cancer survivors [13]. The COST has been used in several studies to assess financial toxicity of cancer survivors as well as insured patients [9,14,15].
Breast cancer is the most common cancer diagnosed in women. Early detection and advanced treatment have dramatically improved survival rates, which might result in high level and long-term financial burdens [3,13,16]. Considering that female sex and younger age are risk factors for financial toxicity [17], it is important to evaluate financial toxicity among breast cancer survivors. In addition, little is known about the impact of financial toxicity in disease-free breast cancer survivors [18]. Thus, we aimed to validate the COST in Korean (COST-K) and evaluate financial toxicity among disease-free breast cancer survivors.
Materials and Methods
1. Linguistic validation
To validate the COST-K, we conducted linguistic validation before psychometric validation. First, we obtained copyright permission from the Functional Assessment of Chronic Illness Therapy multilingual translation (FACITtrans) and translated the COST measure using a standardized methodology recommended by FACITtrans. All documentation pertaining to the COST-K translation, including the item history and decisions about item rephrasing, were reviewed by FACITtrans before being finalized for cognitive testing. No translation issues related to the response options, linguistics, and conceptual equivalence to the original COST measure, as intended, were reported. The participants subsequently underwent cognitive debriefing for 30 minutes to evaluate comprehension; ease of response; and acceptability of the terminology, phrasing, and response options. Cognitive interviews were conducted by one oncology nurse and a behavioral scientist. We recruited cancer patients until saturation; 10 cancer patients with various types of cancers (three, hepatocellular carcinoma; two, lung cancer; two, colon cancer; two, gastric cancer; two, breast cancer) participated in the cognitive interviews; 50% of the patients were male, 60% were over 50 years old, and 50% had an education level of high school or lower.
Overall, the cognitive debriefing revealed that the participants generally comprehended the COST-K well. About 20% of the participants experienced comprehension difficulty for item 4 and item 11, possibly due to cultural differences. Item 4, “I feel I have no choice about the amount of money I spend on care,” queried about the feeling of helplessness due to the financial burden. However, most of our study participants perceived that the item queried about the level of acceptance or willingness to pay for medical costs. It can be understood that patients have to accept treatment decisions regardless of their financial burden. Korean breast cancer survivors’ attitudes toward cancer treatment or treatment cost might be different from those of Western survivors who live in different social and healthcare environments. In addition, item 4 contained a negative expression, which made the phrasing awkward when translated into Korean. Our participants might have been confused and did not know the correct way to answer. About 20% of the participants experienced comprehension difficulty in the phrasing of “I feel in control of my financial situation” (item 11). A survivor in our studies believed that she could control the financial situation as she had enough money to pay for the cost of treatment; however, she did not feel she could manage the future financial situation well because she was not the main source of household income. In conclusion, we did not change any item, and the final version of the COST-K was reviewed and approved by the study team and FACITtrans.
2. Psychometric validation
1) Study participants
We conducted a cross-sectional study of consecutive breast cancer survivors who visited the outpatient breast cancer/oncology clinic at the Samsung Medical Center in Seoul, Republic of Korea, between November 2018 and April 2019. We included breast cancer patients aged above 19 years at the time of diagnosis who had completed active treatment with curative intent within the last 5 years. The exclusion criteria were the presence of secondary cancer, metastasis, and recurrence.
2) Measures
We asked study participants’ socio-demographic characteristics, including marital status, education level, monthly family income, and current working status. Clinical information was obtained from electronic medical records. Linguistically validated COST-K scores of the study participants were evaluated. Low COST-K scores indicated higher financial toxicity.
3) Reliability and validity
To assess the reliability of the COST-K, we calculated the internal consistency of each domain using Cronbach’s α and the item-total correlation of each domain. An α value of 0.8 or higher indicates very good reliability [19]. We invited 250 study participants to participate in the retest, of whom 223 survivors participated (response rate, 89.2%). The survivors who participated in the retest took the COST-K survey again 7 days after the initial survey. For the final test-retest analysis, we excluded 21 participants who had experienced financial events between the test and retest periods, resulting in the inclusion of 202 patients. The test-retest analysis was performed using the intraclass correlation coefficient (ICC) with a 1-way random-effects model [9].
Principal component analysis (PCA) with varimax rotation was performed to determine the underlying structure of the COST-K [20]. Convergent and divergent validities were assessed by determining Pearson’s correlations between the COST-K and relevant and irrelevant domains of the European Organization for Research and Treatment of Cancer Quality of Life Core 30 (EORTC QLQ-C30) questionnaire. We used the Korean version of the EORTC QLQ-C30 questionnaire, which has been validated and used in many studies [21,22]. We hypothesized that the COST-K will have moderate or strong correlation with the financial difficulty item of the EORTC QLQ-C30 questionnaire (during the past week: Has your physical condition or medical treatment caused you financial difficulties?) and a weak correlation with items on acute symptoms, such as nausea and vomiting, constipation, and diarrhea. We also evaluated the association between the COST-K and socio-demographic and clinical factors that were found to be significantly associated with financial toxicity, such as age, marital status, education, employment status, and time since surgery, to confirm the construct validity (“known groups”).
3. Statistical analysis
Data analyses included descriptive statistics (frequencies, means, and standard deviations) and statistics for assessing the questionnaire’s psychometric properties. Correlations between 0.30 and 0.50 were defined as mild; 0.50 and 0.70, moderate; and 0.70 and 0.90, strong. We used a multivariable logistic regression model to evaluate the association between financial toxicity and socio-demographic and clinical factors, adjusting for age, sex, marital status, education level, employment status, monthly family income, disease stage at diagnosis, time since surgery, and type of surgery. For logistic regression, a cutoff of 14 indicated moderate and severe financial toxicity, as suggested by the original developer [23]. All significance tests were two-sided, and a p-value < 0.05 was considered significant. All data analyses were performed using STATA ver. 15 (StataCorp LP, College Station, TX).
Results
1. Study participants
Among the 4,366 eligible participants, 69 did not answer the COST-K survey (missing rate, 1.5%); thus, the final analysis included 4,297 participants. The mean age of the participants was 50.4 (range, 20 to 84 years; mean±SD, 8.6). About 82.7% of the participants were married, 61.2% had a job, and 51.0% had a monthly family income of < US$4,000. The median COST-K was 27 (range, 0 to 44; mean±SD, 27.1±7.5). Breast cancer survivors who were not married, had a low education level, were unemployed, and had a low household income were more likely to report financial toxicity than those who were married, had a high education level, were employed, and had a high household income, respectively (Table 1).
2. Validity and reliability
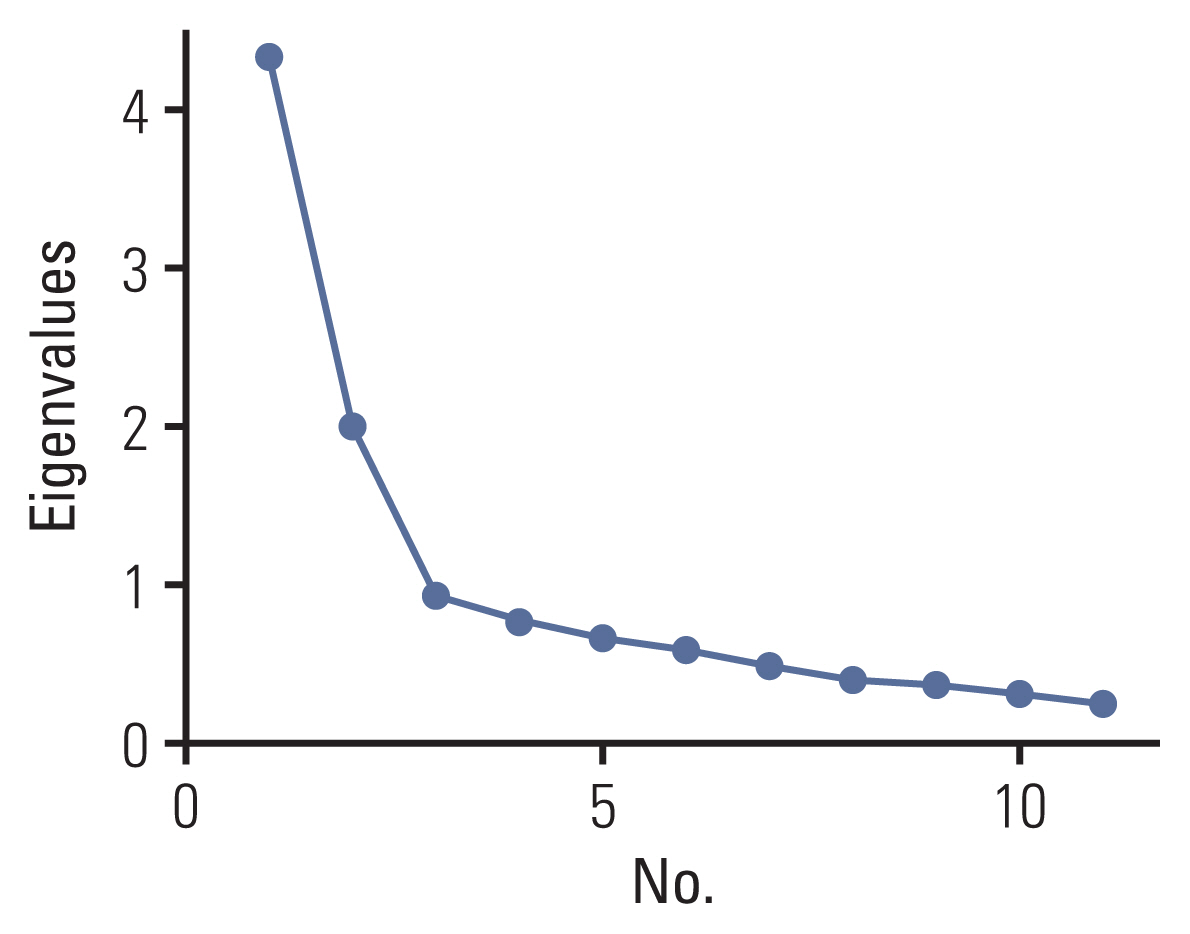
The COST-K demonstrated good internal consistency, with a Cronbach’s α of 0.81. The test-retest analysis revealed an ICC of 0.78 (95% confidence interval, 0.72 to 0.82). The PCA with 1-factor solution, while other items revealed high loadings (0.65 to 0.79), 3 items had loadings lower than 0.50 (item 2, 0.39; item 4, 0.04; item 11, 0.17) (Table 2). Without limiting the number of factors to be retained, the PCA with varimax rotation resulted in a 2-factor solution with an eigenvalue of > 1.0 (Table 3, Fig. 1). The first factor included item 3, item 5, item 8, item 9, and item 10, and the second factor included item 1, item 4, item 6, and item 7. However, item 2 was not related to factors 1 or 2.
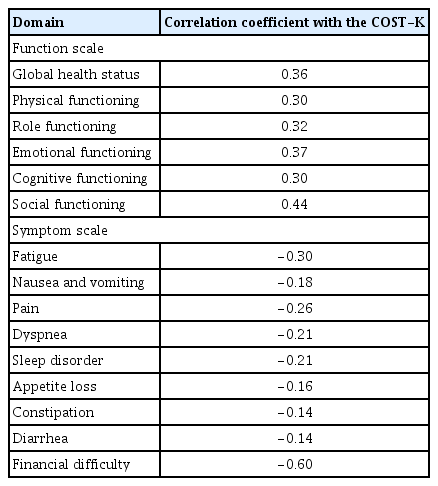
The COST-K showed good convergence with the financial difficulty item of the EORTC QLQ-C30 questionnaire (r= −0.60, p < 0.01) (Table 4), and it was weakly correlated with the items on acute symptom burdens, such as nausea and vomiting, constipation, and diarrhea, showing good divergent validity (nausea/vomiting, −0.18; constipation, −0.14; diarrhea, −0.14) (Table 3).
3. Factors associated with financial toxicity
After adjusting for potential confounders, cancer survivors who were aged < 40 years (odds ratio [OR], 3.90; 95% confidence interval [CI], 2.17 to 7.00), were single (OR, 1.72; 95% CI, 1.23 to 2.39), had an education level of high school or lower (OR, 2.37; 95% CI, 1.40 to 4.02), were unemployed (OR, 1.56; 95% CI, 1.13 to 2.16), and had a monthly family income of ≤ $2,000 (OR, 8.39; 95% CI, 4.42 to 15.94) were more likely to experience financial toxicity than those who were aged ≥ 60 years, were married, had university education, were employed, and had a monthly family income of ≥ $6,000, respectively (Table 5).
Discussion
Here we described the COST-K was easily administered to and completed without discomfort by the breast cancer survivors. The COST-K was found to be a reliable and valid measure of financial toxicity among breast cancer survivors. It had good internal consistency and test-retest reliability, and the convergent and divergent validities were also good.
In a development study, all items had moderate or high loadings (> 0.5) with a 1-factor solution on PCA, supporting the factorability of the items [8]. However, in our study, three items revealed low loadings (items 2, item 4, and item 11). Therefore, we performed PCA without limiting the number of factors to be retained and found two factors, but item 2 did not belong to either factor. The items loaded on the first factor seemed to be related to psychological distress, and the items loaded on the second factor seemed to be related to perceived resource availability. Item 2 (“My out-of-pocket medical expenses are more than I thought they would be”) had low loading on PCA with varimax rotation, regardless of the number of factors retained. This may be due to the national healthcare system in Korea. South Korea has a national health insurance system that covers all people in the nation. In addition, all patients with cancer have special coverage programs and only pay 5%–10% of the total health expenditure. Thus, our study participants might have had relatively little concern about out-of-pocket medical expenses, and item 2 was not sensitive enough to assess financial toxicity. In addition, the low loading of item 2 might be due to the characteristics of the study population. We included breast cancer survivors who had completed their treatment and had no signs of recurrence. They might have little concern about out-of-pocket medical expenses. In fact, less than 5% of our study participants agreed or strongly agreed that their out-of-pocket medical expenses were more than they thought they would be (S1 Table). While the comprehension issues related to item 4 and item 11 were due to cultural differences during the linguistic validation process, we did not exclude these items to ensure consistency of the COST measure as a standardized instrument across the country for the psychometric analysis. Then, on PCA, item 4 and item 11 had much lower loadings than other items, confirming our findings with linguistic validation. However, the overall reliability and test-retest reliability of the COST-K were good. Further studies are necessary to modify these items and their responses.
Regarding convergent and divergent validities, there was a moderate correlation between the COST-K and the financial difficulty item of the EORTC QLQ-C30 questionnaire, whereas weak correlations were noted between the acute symptom items and COST-K, confirming its divergent validity. In addition, the COST-K showed a mild correlation with the health-related quality of life (HRQoL) assessed by the EORTC QLQ-C30 questionnaire, indicating that financial toxicity is an important patient-centered outcome that affects the survivors’ HRQoL. In particular, there was a mild correlation between physical symptoms such as fatigue, pain, and sleep disturbance. The results are similar to those of the previous studies. Financial toxicity was found to be related to symptom burden in both cancer patients and survivors [7,9,24,25]. Similar to other studies, we also found socio-demographic and clinical factors that were associated with financial toxicity on multivariate logistic regression, confirming the validity of the COST-K [5,9,13,26].
One study found that 14.8%–78.8% of cancer survivors experienced financial toxicity, especially in low-income populations [27]. In our study, over 30% and 5% of cancer survivors experienced mild and severe financial toxicity, respectively. Korean breast cancer survivors experience less financial toxicity than survivors in other countries due to relatively low medical costs in South Korea owing to coverage by the national health insurance system. However, survivors with low income were more likely to experience financial toxicity than those with high income. Survivors with low income would still experience substantial financial toxicity because of the various direct and indirect costs of cancer treatment [13]. For example, they would have more financial burden due to reconstruction surgery, additional expensive treatment on their own or having private insurance. They might experience high levels of financial toxicity because of a lack of savings and assets and unexpected medical expenditure [27]. Further study would be necessary to find factors associated with financial toxicity and develop interventions for these vulnerable populations as early as possible to minimize financial toxicity.
Our study had several limitations. First, given the cross-sectional design, participants would answer differently depending on time and environment. Therefore, we conducted a test-retest reliability test and confirmed the consistency of the COST-K in our population. Second, the convergent validity of the COST-K was not examined using a separate instrument for financial toxicity among cancer survivors. However, there was no standardized instrument for measuring subjective financial distress or financial toxicity in cancer survivors [5]. Instead, we tested the correlation between the COST-K and the financial difficulty item of the EORTC QLQ-C30 questionnaire, which is a well-known, frequently used, and a validated measure [5,28,29] of financial toxicity in cancer patients. The findings of this study might not be generalizable to other populations in other settings because this study was conducted with breast cancer survivors at a single institution in Seoul, South Korea. Female cancer survivors might experience more psychological distress due to financial difficulties than male cancer survivors. Furthermore, survivors in other countries with health insurance systems different from the one in South Korea might experience different types or levels of financial toxicity.
Our study demonstrated that the COST-K is a valid and reliable instrument for measuring financial toxicity in disease-free breast cancer survivors. To the best of our knowledge, this is the first validation study of the COST involving disease-free cancer survivors. Considering its impact on the HRQoL, more studies are necessary to evaluate and intervene financial toxicity among breast cancer survivors who would experience more financial challenges due to younger age.
Electronic Supplementary Material
Supplementary materials are available at Cancer Research and Treatment website (https://www.e-crt.org).
Notes
Ethical Statement
The study was approved by the Institutional Review Board of the Samsung Medical Center, Seoul, Republic of Korea, and informed consent was obtained from all study participants (IRB number: SMC-2018-08-070).
Author Contributions
Conceived and designed the analysis: Shim S, Kang D, Kim N, Park J, Lee SK, Cho J.
Collected the data: Shim S, Park J, Lee JE, Kim SW, Yu J, Chae BJ, Ryu JM, Nam SJ, Lee SK.
Contributed data or analysis tools: Shim S, Kang D, Kim N, Han G, Lim J, Kim H, Lee M, Lee JE, Kim SW, Yu J, Chae BJ, Ryu JM, Nam SJ, Lee SK, Cho J.
Performed the analysis: Shim S, Kang D, Han G, Lim J, Kim H, Lee M.
Wrote the paper: Shim S, Kang D, Lee SK, Cho J.
Conflicts of Interest
Conflict of interest relevant to this article was not reported.
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT, and Future Planning (2017R1E1A1A0107764214).