Predictive Value of In Vitro Adenosine Triphosphate-Based Chemotherapy Response Assay in Advanced Gastric Cancer Patients Who Received Oral 5-Fluorouracil after Curative Resection
Article information
Abstract
Purpose
To assess the usefulness of adenosine triphosphate-based chemotherapy response assay (ATP-CRA) results in advanced gastric cancer patients receiving adjuvant chemotherapy.
Materials and Methods
Sixty-two patients underwent curative surgical resection between January, 2006 and December, 2008. Their highly purified surgical specimens were evaluated by ATP-CRAs. Of the 62, 49 had successful assay results and they received either oral 5-fluorouracil or other chemotherapies. We retrospectively analyzed data for 24 patients who were treated with oral 5-fluorouracil and whose assays were successful.
Results
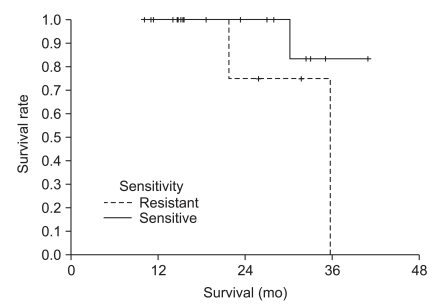
The median observation time was 24.6 months (range, 10.1 to 40.9 months). The median treatment time was 11.2 months (range, 1.2 to 17.7 months). The median age was 66 years (range, 30 to 81 years). Patients were grouped into sensitive- and resistant-groups according to adenosine triphosphate-based chemotherapy response results for fluorouracil. The sensitive-group showed a significantly longer time to relapse (not reached in the sensitive-group vs. 24.8 months in the resistant-group, p=0.043) and longer overall survival compared to the resistant-group (not reached in the sensitive-group vs. 35.7 months in the resistant-group, p=0.16, statistically insignificant).
Conclusion
Patients who receive curative surgical resection significantly benefit from sensitive adjuvant chemotherapy according to ATP-CRA results for time to relapse.
Introduction
Gastric cancer is the second most common cause of cancer-related death in the world, the fifth most prevalent cancer worldwide [1], and the leading cause of cancer-related death in eastern Asian countries. Curative resection has been considered the most important prognostic factor in advanced gastric cancer (AGC) [2], but most patients are at risk of recurrence. Adjuvant therapies adopting single or combined chemotherapy approaches with curative intent have been widely examined, and several meta-analyses have demonstrated modest survival benefits for adjuvant chemotherapy [3,4]. Considering the toxicity of chemotherapies, properly choosing drugs for patients who have undergone curative resection is important. By far, postoperative chemotherapy in AGC after curative resection has controversial survival benefits [5,6]. These facts have led to the concept of individualized tumor response tests (ITRT) [7] to increase the efficacy and feasibility of certain drugs and to predict the prognosis and survival benefits of sensitive drugs.
By definition, ITRT are any laboratory methods performed to evaluate malignant cell growth inhibition by conventional chemotherapeutic agents [8]. Several methodologies for ITRT have been developed including the adenosine triphosphate-based chemotherapy response assay (ATP-CRA) and the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) chemosensitivity test (MTT assay). These seem to be ideal tests that could help oncologists easily select effective chemotherapeutic agents for patients with advanced stage cancer. Surgeons in Japan have made use of the MTT assay and obtained positive results for AGC patients undergoing both adjuvant [9] and palliative chemotherapy [10]. In our study, we adopted an ATP-CRA to predict the response of chemotherapeutic agents. ATP-CRA is a sensitive assay that evaluates tumor cell viability by measuring the intracellular ATP levels of drug-exposed cells and untreated controls. This assay has been adopted for non-small cell lung cancer [11], ovarian cancer [12], and breast cancer [13] providing positive results in patients who underwent curative resection.
Several types of oral 5-fluorouracil (FU) have been developed for gastrointestinal (GI) tract cancer as a substitute for intravenous 5-FU with comparable efficacies but lower toxicities: S-1, UFT, capecitabine, and doxifluridine. S-1 and Tegracil are combinations of tegafur (a prodrug that is converted by cells to fluorouracil), inhibitors of dihydropyrimidine dehydrogenase (cholorodihydroxy pyridine in S-1 and uracil in Tegracil), and oxonic acid in S-1 [14]. Capecitabine (Xeloda) is a prodrug of doxifluridine, which is converted to the active FU by thymidine phosphorylase in tumor cells. The rate of response to treatment with each of the oral 5-FUs ranges around 30% [15].
In this study, we retrospectively analyzed 24 patients who underwent curative resection and postoperatively were treated with oral 5-FU. To assess the feasibility of using the ATP-CRA assay in adjuvant chemotherapy, we compared the relapse rate and survival between sensitive and insensitive groups.
Materials and Methods
1. Patients
Sixty-two patients, who were diagnosed as having AGC underwent curative surgery in Dong-A University Hospital between January, 2006 and November, 2008. ATP-CRA was done on tumor tissue specimens obtained from patients with pathologically confirmed AGC. The eligible patients for this study were subjects from whom we had successfully obtained assay results, who received extended surgery for AGC, and who received more than one month of 5-FU per os. The other eligibility criteria were: 1) aged at least 18 years; 2) histologically or cytologically proven AGC; 3) stage IB, II or III [16]; 4) an Eastern Cooperative Oncology Group performance status (ECOG PS) of 2; 5) adequate bone marrow function (neutrophils≥1.5×103/µL, platelets≥100×103/µL, and Hb≥10.0 g/dL), adequate renal function (serum creatinine≤1.5×upper normal limit), and adequate liver function (serum bilirubin≤1.5×upper normal limit, aspartate aminotransferase, and alanine aminotransferase≤1.5×upper normal limit); and 6) no history of other malignancies during the observation time. This clinical trial was approved by the appropriate Institutional Review Board, and all patients in the study gave written informed consent.
2. ATP-based chemotherapy response assay methodology
1) Isolation of cancer cells, elimination of normal cells, and tissue culture
Tumor tissues stored in Hank balanced salt solution (Gibco, Rockville, MD), containing 100 IU/mL penicillin (Sigma, St. Louis, MO), 100 µg/mL streptomycin (Sigma), 100 µg/mL gentamicin (Gibco), 2.5 µg/mL amphotericin B (Gibco), and 5% fetal bovine serum (FBS, Gibco) were delivered to the laboratory. These tissues were washed, quantified, and minced, and then incubated with extracellular matrix degrading enzymes, such as dispase (Sigma), pronase (Sigma), and DNase (Sigma), at 37.8℃ for 12 to 16 hours. Cells were harvested using a cell strainer (BD Falcon, Bedford, MA). To eliminate red blood cells and dead cells, the cell suspensions were subjected to Ficoll (1.077 g/mL) gradient centrifugation at 400 g for 15 minutes. The viability of isolated cells was tested using Trypan blue exclusion. The histological types of the tumor tissues as well as qualitative and quantitative analyses of the cancer cells were evaluated by pathologists.
2) ATP measurement
Separated tumor cells were diluted to 2,000-20,000 viable cells/100 µL using Iscove modified Dulbecco medium (IMDM, Gibco), including 10% FBS, and seeded in triplicate into wells of a 96-well ultra-low attachment plate (Costar, Cambridge, MA), which restricted the growth of normal cells (e.g., fibroblasts). For the treated groups, 100 µL of chemotherapeutic agents were added to the seeded cell cultures and incubated for 48 hours in a CO2 incubator. For the untreated control groups, 100 µL of IMDM, without chemotherapeutic agents, were added. For quality control, a negative control group of three to six wells (containing only seeding medium but without cells), and two positive control groups were included in the culture plate. Each positive control group was composed of three wells that contained the minimal (105 pg ATP) and median (280 pg ATP) amounts of ATP, as measured in 1,000 tumor cells harvested from tissue. Treated drug concentrations (TDC, concentrations of treatment drugs) were determined by preliminary experiments. They exhibited a scattered distribution of cell death from each specimen. Drugs were used in triplicate at ×0.2, ×1, and ×5 TDC. The TDCs used were: 5-FU (10 µg/mL), oxaliplatin (2.9 µg/mL), irinotecan (4.7 µg/mL), cisplatin (2.5 µg/mL), carboplatin (12 µg/mL), paclitaxel (8.5 µg/mL), docetaxel (3.7 µg/mL), gemcitabine (16.9 µg/mL), and vinorelbine (0.18 µg/mL), which are all active drugs widely used in combination with 5-FU in AGC. Eighteen cells from the untreated control and from the treated groups were lysed, and the amount of ATP in the cell lysates was measured using flash luminescence measurements on a Victor 3 multilabel counter (PerkinElmer, Boston, MA). The cell death rate for each drug was calculated using the following formula:
Cell death rate (%)=

A chemosensitivity index (CI) was calculated using the sum of the percentages of cell deaths for all concentrations tested: CI=(300-sum of % cell death rate at ×0.2, ×1 and ×5 TDC).
Luminescence measurements were directly related to ATP levels; they allowed for evaluation of the percentage of cell deaths with respect to the untreated control. We did not use background subtraction because the quantity of ATP in the "media only" well (dispensed medium without cells) and the quantity of extracellular ATP were always negligible. The intra-assay mean coefficient of variation (CV) was calculated using the luminescence value measured three times in each specimen. If microorganism contamination was present, if the intra-assay mean CV exceeded 30, or if the measurement luminescence in the untreated control group was lower than that in the positive control group (105 pg of ATP), the test was considered a failure.
3. Chemotherapy
Patients were administered oral 5-FU four weeks after an operation for AGC with curative intent. Oral 5-FUs that were administered were Tegracil (Shinpoong Pharmaceutical, Ansan, Korea) 300 mg/m2/day in 15 patients, TS-1 (Jeil Pharmaceutical, Seoul, Korea) 30 mg/m2 bid. in two patients, Capecitabine (Roche Pharmaceutical, Nutley, NJ) 1,250 mg/m2 bid. in three patients, and Doxifluridine bid in four patients. The mean duration of chemotherapy administration was 10.76 months: 11.5 months in the S-group and 9.3 months in the R-group (p=0.978).
4. Endpoints and statistical analysis
The endpoints of our study were the correlations between (1) the ATP-CRA results and (2) time to relapse (TTR) or overall survival (OS). TTR was defined as the time from commencement of chemotherapy until disease progression or patient death. OS was defined as the time from chemotherapy to death from all causes.
All statistical calculations were carried out using the SPSS ver. 11.5 (SPSS Inc., Chicago, IL). All p-values were two-sided, and the p-value for statistical significance was set at 0.05. Means and medians were calculated when appropriate, continuous variables were compared by Student's t-test, and categorical variables were analyzed by χ2 test. Survival was calculated using the Kaplan-Meier method. A log-rank test was used to compare survival between subgroups. Prognostic variables found by univariate analysis were used in multivariate analysis by the Cox proportional hazard regression model.
Results
1. Patient characteristics
From January, 2006 to December, 2008, 62 patients underwent curative surgical resection for AGC. The success rate of the ATP-CRA assay was 49 out of 62 (79%). Among the 49 successful ATP-CRA patients, 21 received other chemotherapies and four patients did not receive chemotherapy. A total of 24 patients was retrospectively analyzed (Fig. 1). The median age was 66 years (range, 30 to 81 years) and the male to female ratio was 2 : 1. Patients were grouped into sensitive- (S-) and resistant- (R-) groups according to ATP-CRA results for 5-FU. There were no significant differences in pretreatment factors between the subgroups. The clinicopathologic data of patients are shown in Tables 1 and 2.

Patient selection. Non-oral 5-fluorouracil (5-FU) contains patients who did not receive chemotherapy and who were treated with other chemotherapies. CST, chemosensitivity test.
2. TTR and OS according to ATP-CRA results
The median observation time was 22.6 months (range, 10.1 to 40.9 months). Four of 24 patients underwent disease progression. Of these four, three patients expired due to cancer progression. The median TTR did not reach the end point, with a two year TTR of 75%. The median OS was 23.3 months (95% confidence interval, 10.29 to 36.31) with a two year OS rate of 91%.
Following the ATP-CRA results, the S-group showed significantly longer TTR (not reached in the S-group vs. 24.8 months in the R-group, p=0.043) and an insignificantly longer OS compared to the R-group, (not reached in the S-group vs. 35.7 months in the R-group, p=0.16) (Figs. 2 and 3).
3. Predictors for clinical response and prognostication for progression or survival
Chemosensitivity result according to the ATP-CRA assay was the only significant predictor for TTR, whereas there were no significant pretreatment predictors in univariate analysis. In multivariate analysis; however, there were no significant factors for prediction of TTR and OS (data not shown).
Discussion
Since the development of the oral prodrug form of FU in 1967, numerous studies have been conducted to demonstrate the efficacy of the oral form of FU compared with protracted intravenous infusion of FU. The discovery of dihydropyrimidine dehydrogenase (DPD) in the catabolic pathway of FU led to the use of combinations of inhibitors of DPD with the oral prodrug of FU, yielding a prolonged activity of FU [15]. The invention of another form of oral prodrug of FU, capecitabine, boosted the investigation of the oral form of FU in GI tract tumors and other solid tumors of epithelial cell origin. A recent study comparing continuous infusion of 5-FU and capecitabine, the REAL-2 and ML17032 trials, showed that oral fluoropyrimidine is non-inferior to 5-FU for OS and progression free survival, respectively, in advanced oesophago-gastric cancer [17]. However, combinations of these various types of oral FU prodrugs as a single agent have not undergone large scale randomized studies. But, some pilot studies have shown that the response rate to each of the prodrugs of GI tract tumors was approximately 30% [18,19]. In this study, patients who underwent curative resection for AGC were treated with various prodrug forms of oral fluoropyrimidine without combination with other infusional chemotherapeutic agents as adjuvant therapy.
Previous intravenous combination chemotherapy for adjuvant purposes in AGC revealed a negative result for disease free survival or OS, but showed a high response rate [5]. However, studies in Japan, ones making use of another in vitro assay (the MTT-assay), produced a positive result in both response rate and in OS [9,10]. In our study, although the number of patients who had undergone ATP-CRA assays and had successful results was small, the clinical outcomes of the chemosensitive group were more favorable than those for TTR. The TTR and OS were not significantly different as a function of cancer stage. The patients in the S- and R-groups had similar pretreatment parameters in age, stage, depth of invasion, nodal metastasis, and histological types, and there were no significant differences in these factors. Previous studies have shown that an anchorage-independent culture method, such as an agar underlayer, inhibits the growth of fibroblasts, allowing tumor cells to survive and proliferate. In our study, we adopted the use of another ultra-low attachment plate, one that inhibits fibroblasts better than the agar plate, which may have allowed a positive result. The beneficial aspect of oral prodrug forms of FU in TTR may have additional significance for the role of oral forms of FU in adjuvant chemotherapy of AGC, which is the primary concern of most oncologists.
Although ITRT-guided chemotherapy seems to be ideal, in general clinical practice these methods are not widely accepted because they require high technical skill, a large number of tumor cells, and a long turnaround time for testing. ATP-CRA has a relatively short turnaround time (a few days) and high success rates even with bronchoscopic biopsy specimens in lung cancers [11]. We have shortened the turnaround time to 48 hours and raised the drug concentrations, which has been tried in other studies [20]. Hence, clinicians can simply adopt the ATP-CRA in clinical trials.
The validity of a diagnostic test to be applied in the clinical setting is assessed by positive predictive value (PPV) and negative predictive value (NPV). For the ITRT used in our study, the ATP-CRA assay showed a PPV of 94% and an NPV of 38%. This result is opposite that of previous studies which have reported higher NPV than PPV values [9,10]. Higher PPV than NPV values means that the ATP-CRA assay can detect drug sensitivity better than drug resistance. Although the heterogeneity of drug sensitivity in a tumor specimen in patients may vary, complete removal of the tumor may provide a comprehensive chemosensitivity of the malignant tissue. The low NPV of the ATP-CRA assay may make clinicians avoid the resistant drugs by the sensitivity test. The small number of patients analyzed may have resulted in NPV that is less reliable, so studies on chemosensitivity tests with a large number of patients are needed to demonstrate the validity of the ATP-CRA.
Conclusion
A limitation of our study was that the type of oral 5-FUs and the duration of administration were not the same, creating different pharmacokinetics and different drug levels in different individuals. In this study, we compared very small numbers of patients that were chemo-sensitive or resistant, and this assay cannot be easily generalized to predict the outcome of chemotherapy. However, the positive correlation of ATP-CRA results and clinical outcomes should encourage randomized, prospective studies with more patients that compare ATP-CRA results and conventional adjuvant chemotherapies in AGC patients that have undergone curative surgical treatments.
Acknowledgments
This paper was supported by the Dong-A University Research Fund. This work was supported by a Korea Science and Engineering Foundation (KOSEF) grant funded by the Korean government (MEST; R13-2002-044-05001-0).
Notes
Conflict of interest relevant to this article was not reported.