Efficacy and Safety of Ceritinib 450 mg/day with Food and 750 mg/day in Fasted State in Treatment-Naïve Patients with ALK+ Non–Small Cell Lung Cancer: Results from the ASCEND-8 Asian Subgroup Analysis
Article information
Abstract
Purpose
Previous report from the ASCEND-8 trial showed consistent efficacy with less gastrointestinal (GI) toxicity in patients with anaplastic lymphoma kinase-rearranged (ALK+) advanced/metastatic non–small cell lung cancer (NSCLC) treated with ceritinib 450-mg with food compared with 750-mg fasted. In this subgroup analysis, we report outcomes in Asian patients of the ASCEND-8 trial.
Materials and Methods
Key efficacy endpoints were blinded independent review committee (BIRC)–assessed overall response rate (ORR) and duration of response (DOR) evaluated per Response Evaluation Criteria in Solid Tumors v1.1. Other efficacy endpoints were investigator-assessed ORR and DOR; BIRC- and investigator-assessed progression-free survival (PFS) and disease control rate; overall survival (OS). Safety was evaluated by frequency and severity of adverse events.
Results
At final data cutoff (6 March 2020), 198 treatment-naïve patients were included in efficacy analysis, of which 74 (37%) comprised the Asian subset; 450-mg fed (n=29), 600-mg fed (n=19), and 750-mg fasted (n=26). Baseline characteristics were mostly comparable across study arms. At baseline, more patients in 450-mg fed arm (44.8%) had brain metastases than in 750-mg fasted arm (26.9%). Per BIRC, patients in the 450-mg fed arm had a numerically higher ORR, 24-month DOR rate and 24-month PFS rate than the 750-mg fasted arm. The 36-month OS rate was 93.1% in 450-mg fed arm and 70.9% in 750-mg fasted arm. Any-grade GI toxicity occurred in 82.8% and 96.2% of patients in the 450-mg fed and 750-mg fasted arms, respectively.
Conclusion
Asian patients with ALK+ advanced/metastatic NSCLC treated with ceritinib 450-mg fed showed numerically higher efficacy and lower GI toxicity than 750-mg fasted patients.
Introduction
Aberrant activation of the anaplastic lymphoma kinase (ALK)–receptor tyrosine kinase via chromosomal rearrangement of the ALK gene is observed in 3% to 7% of patients with non–small cell lung cancer (NSCLC) [1]. Therapies targeting the ALK protein in this subset of patients with NSCLC provide greater clinical benefit than standard chemotherapy [2,3]. Crizotinib was the first ALK-inhibitor (ALKi) approved by the U.S. Food and Drug Administration (FDA) in 2011 for the treatment of patients with NSCLC whose tumors are ALK-activated (ALK+) [4]. In 2014, ceritinib, a second generation ALKi received FDA accelerated approval for the treatment of patients with ALK+ NSCLC who progressed on crizotinib or those who could not tolerate crizotinib [5]. Thereupon, multiple next-generation ALK inhibitors including alectinib, brigatinib, lorlatinib and ensartinib have been developed to circumvent challenges from acquired resistance, prolong survival of patients and minimize toxicities [6].
In 2017, ceritinib received regular approval by the FDA for the treatment of treatment-naïve and pretreated patients with metastatic ALK+ NSCLC as detected by an FDA-approved test [5,7]. The FDA approval for ceritinib was based on data from the phase 3 ASCEND-4 trial, which evaluated ceritinib in the first-line setting, and the ASCEND-5 trial, which evaluated ceritinib in pretreated patients using a 750-mg dose in a fasted state [3,8]. Because ceritinib administered at the initial approved dose of 750 mg/day under fasted state was associated with high incidence of gastrointestinal (GI) adverse events (AEs) [3,8–10], we evaluated the pharmacokinetics (PK), safety, and efficacy of lower doses of ceritinib at 450 and 600 mg/day taken with a low-fat meal versus 750 mg/day taken under a fasted state in the phase 1 ASCEND-8 study.
Primary results from the global phase 1 ASCEND-8 trial (NCT02299505) showed that treatment with ceritinib 450 mg/day with a low-fat meal (450-mg fed) results in comparable steady-state exposure (primary end point) as the initially approved dose of 750-mg/day in a fasted state (750-mg fasted) in patients with ALK+ advanced/metastatic NSCLC [11]. In addition, results from the ASCEND-8 study also showed consistent efficacy of the ceritinib 450-mg fed arm compared with the 750-mg fasted arm, with a low incidence of GI-related toxicity (diarrhea, vomiting, and nausea) [12]. The overall response rate (ORR; 95% confidence interval [CI]) assessed by a blinded independent review committee (BIRC) was 78.1% (66.9 to 86.9) in the 450-mg fed arm, 72.5% (58.3 to 84.1) in the 600-mg fed arm, and 75.7% (64.3 to 84.9) in the 750-mg fasted arm. Patients in the 450-mg fed arm had the lowest incidence of any-grade GI-related toxicities (75.9%) compared with the 600-mg fed (82.6%) and 750-mg fasted (91.8%) arms. Grade 3/4 GI-related toxicities were reported in 2.8%, 8.1%, and 13.6% of patients in the 450-mg fed, 600-mg fed, and 750-mg fasted arms, respectively. Based on the results from the global ASCEND-8 trial, the recommended/ approved dose of ceritinib has been revised to 450 mg/day with a low-fat meal [5]. We have recently published data showing that the efficacy and safety of ceritinib administered at a dose of 750 mg/day in a fasted state in Asian patients was consistent with that of the global patient population in the phase 3 ASCEND-4 trial [13]. However, no study to date has evaluated the efficacy and safety of first-line ceritinib at 450 mg/day administered under fed conditions in Asian patients with advanced ALK+ NSCLC. This additional subgroup analysis assessed the efficacy and safety of ceritinib administered at a dose of 450 mg and 600 mg under fed conditions, and 750 mg under fasted conditions in treatment-naïve Asian patients with ALK+ NSCLC in the ASCEND-8 study.
Materials and Methods
1. Study design and patient population
ASCEND-8 was a randomized, multicenter (87 sites across 24 countries), open-label, parallel-design, phase 1 study (Fig. 1). Details of the ASCEND-8 study design were published previously [11,12]. Briefly, 306 patients (aged ≥ 18 years; both pretreated and treatment naïve) were randomized globally in a 1:1:1 ratio to receive one of the following treatments: ceritinib 450 mg/day with a low-fat meal (n=108), ceritinib 600 mg/day with a low-fat meal (n=87), and ceritinib 750 mg/day in a fasted state (n=111). Ceritinib was administered immediately (within 30 minutes) following a low-fat meal (food), defined as approximately 100 to 500 calories, and 1.5 to 15 g of fat (S1 Table) was used in this study because of the modest difference in PK between a light snack, low-fat meal, and high-fat meal after a single dose of ceritinib. Enrollment to the ceritinib 600-mg fed arm was closed early due to higher exposure and a less favorable safety profile. In the current subgroup analysis, data from treatment-naïve Asian patients with stage IIIB or IV NSCLC harboring the ALK rearrangement (confirmed by immunohistochemistry) who were evaluated for efficacy and safety in ASCEND-8 are reported. Eligible patients had a World Health Organization (WHO) performance score of 0 to 2 and at least one measurable lesion, as per Response Evaluation Criteria in Solid Tumors (RECIST) ver. 1.1, and could have asymptomatic or neurologically stable central nervous system metastases.

ASCEND-8 study design. ALK, anaplastic lymphoma kinase; BIRC, blinded independent review committee; CTCAE, Common Terminology Criteria for Adverse Events; DCR, disease control rate; DOR, duration of response; FDA, Food and Drug Administration; FISH, fluorescence in situ hybridization; IHC, immunohistochemistry; NSCLC, non–small cell lung cancer; OS, overall survival; PFS, progression-free survival; PK, pharmacokinetics; R, randomization; RECIST, Response Evaluation Criteria in Solid Tumors; WHO PS, World Health Organization performance status. a)Prior adjuvant or neoadjuvant therapy was allowed if relapse occurred > 12 months after chemotherapy, b)Patients could continue to receive treatment with ceritinib following disease progression, including cases of isolated brain progression, if continued treatment provided clinical benefit in the opinion of the investigator.
Ceritinib treatment was to be continued until disease progression, unacceptable toxicity, or consent withdrawal or at the discretion of the investigator. Patients could receive ceritinib following progressive disease, including cases of isolated brain progression, if continued treatment provided clinical benefit in the opinion of the study investigator.
2. Endpoints
The primary endpoint of the phase 1 ASCEND-8 showing similar exposure in patients treated with ceritinib 450 mg/day with low-fat meal compared with 750 mg/day under fasted conditions has been published [11]. We have also previously reported data from key secondary endpoints showing consistent efficacy and less GI toxicity with ceritinib 450 mg/day with a low-fat meal compared with 750 mg/day under fasted conditions in a global patient population of the ASCEND-8 study [12]. In this subgroup analysis, we assessed data on key secondary endpoints in the Asian subgroup of the ASCEND-8 study including BIRC-assessed ORR and duration of response (DOR) as per RECIST ver. 1.1 are reported in this analysis. Other secondary end points included investigator-assessed ORR and DOR per RECIST ver. 1.1, BIRC- and investigator-assessed progression-free survival (PFS), time to response (TTR) and DOR per RECIST ver. 1.1, overall survival (OS). Safety was assessed as the incidence and severity of AEs.
3. Assessments
Computed tomography of the chest and abdomen, and computed tomography or magnetic resonance imaging of the brain were performed at baseline in all patients. Tumor response assessments were performed starting at cycle 3 (1 cycle was of 21 days) and then every 2 cycles thereafter through cycle 9. Subsequently, the frequency of tumor assessments may be reduced as clinically indicated, at the discretion of the investigator, but no less than once every 4 cycles until the end of treatment. Tumor assessments in patients who discontinued therapy for reasons other than disease progression, loss to follow-up, pregnancy, or death were to be performed at least every 12 weeks following the end of treatment, until progressive disease as per RECIST ver. 1.1 (as confirmed by BIRC), withdrawal of consent for further tumor assessments, or death. AEs were coded using the Medical Dictionary for Regulatory Activities ver. 21.0 and graded as per the Common Terminology Criteria for Adverse Events ver. 4.03. Dose reductions (150 mg/day per dose reduction) were allowed in all the arms; a maximum of 2 dose reductions in the 450-mg fed arm and maximum of 3 dose reductions in the 600-mg fed and 750-mg fasted arms were allowed.
4. Statistical analyses
This subgroup analysis includes data from treatment-naïve patients in the Asian subset of the global phase 1 ASCE-ND-8 trial. Data are presented as descriptive summary statistics, e.g., for TTR. ORR, disease control rate (DCR), and the associated exact binomial 95% CIs were reported. The Kaplan-Meier methodology was used to determine the median values with the associated 95% CIs for DOR, PFS, and OS using the Brookmeyer and Crowley method. SAS ver. 9.4 (SAS Institute Inc., Cary, NC) was used to perform all statistical analyses.
Results
1. Patient characteristics and disposition
Overall, as of the data cutoff date (March 6, 2020), 198 treatment-naïve patients from the full analysis set (FAS) were included in the efficacy analysis; of these, 74 (37.4% of the treatment-naïve patients in the FAS) constituted the Asian subgroup. In the Asian subgroup, there were 29 patients in the 450-mg fed arm, 19 patients in the 600-mg fed arm and 26 patients in the 750-mg fasted arm. The median duration of study follow-up from randomization to the data cutoff date in the Asian subgroup was 36.7 months; 450-mg fed (36.5 months), 600-mg fed (40.1 months), and 750-mg fasted (31.8 months). Baseline demographics and disease characteristics were mostly comparable across the study arms in the Asian subgroup, except for sex, ethnicity, and incidence of brain metastases (Table 1). At baseline, more patients in the 450-mg fed arm had brain metastasis than the 750 mg fasted arm (44.8% vs. 26.9%) (Table 1). At the time of data cutoff, 38 patients (51.4%) in the Asian subgroup continued to receive treatment and were transferred to other patient programs or clinical trials to continue receiving ceritinib: 450-mg fed, 19 (65.5%); 600-mg fed, seven (36.8%); and 750-mg fasted, 12 (46.2%). Among patients who discontinued in the treatment phase, the most common reason (25/74 patients [33.8%]) for discontinuation was progressive disease (450-mg fed, 6 [20.7%]; 600-mg fed arm, 9 [47.4%]; and 750-mg fasted, 10 [38.5%]).
2. Efficacy
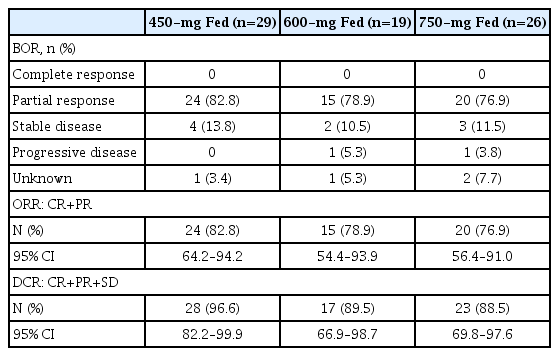
1) Response to ceritinib therapy
The best overall responses per BIRC in the treatment-naïve Asian subgroup are shown in Table 2. Across all arms, most patients had a partial response, which was the best response overall in the study (no complete response). The ORR and DCR per BIRC were numerically higher in the 450-mg fed arm compared with the 750-mg fasted arm, with overlapping CIs. The ORR and DCR per BIRC in Asian patients in this subgroup analysis were comparable with that reported in the global patient population of the ASCEND-8 study [12]. The investigator-assessed response rates were consistent with the BIRC-assessed response rates (S2 Table). In patients who showed response, the median DOR (95% CI) as per BIRC was not estimable (NE) (16.4 to NE) months in the 450-mg fed arm, 20.7 (4.2 to NE) months in the 600-mg fed arm, and 13.2 (4.5 to 17.9) months in the 750-mg fasted arm (Fig. 2). The DOR rate (95% CI) at 24 months per BIRC in Asian patients was 68.2% (44.5 to 83.4) in the 450-mg fed arm, 44.9% (16.4 to 70.1) in the 600-mg fed arm, and 20.3% (5.5 to 41.5) in the 750-mg fasted arm. The investigator-assessed median DOR and DOR rate at 24 months were consistent with the BIRC-assessed data (S3 Table). The median TTR as per BIRC was 6.0, 6.1, and 6.0 weeks in the 450-mg fed, 600-mg fed, and 750-mg fasted arms, respectively, corresponding with the first tumor assessment in these patients.
2) Response to ceritinib therapy in patients with brain metastasis
Overall, 28 treatment-naïve patients had brain metastasis at baseline in the Asian subgroup of the ASCEND-8 study; 13 patients (44.8%) in the 450-mg fed arm, eight patients (42.1%) in the 600-mg fed arm, and seven patients (26.9%) in the 750-mg fasted arm. Among those patients, the BIRC-assessed ORR in the 450-mg fed arm (76.9%) was comparable with the 750-mg fasted arm (71.4%) and numerically higher with overlapping CIs compared with the 600-mg fed arm (62.5%). The ORR as per the investigator assessment in patients with brain metastasis was similar to the BIRC-assessed data (S4 and S5 Tables).
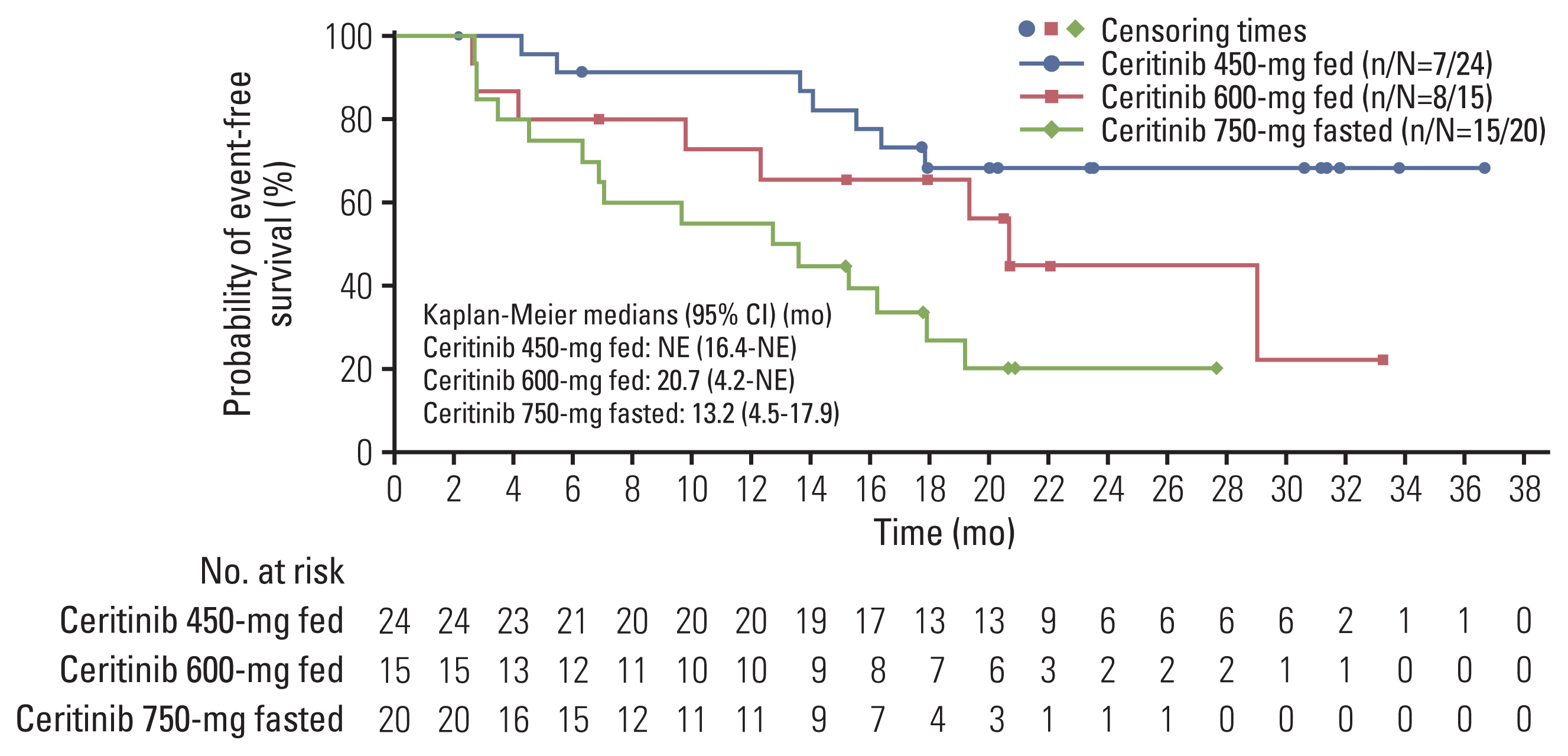
3) Progression-free survival
The median PFS (95% CI) per BIRC was NE (16.9 to NE) months in the 450-mg fed arm, 21.9 (4.1 to NE) months in the 600-mg fed arm, and 8.2 (5.4 to 16.6) months in the 750-mg fasted arm (Fig. 3A). The investigator-assessed median PFS (95% CI) was NE (19.2 to NE) months in the 450-mg fed arm, 16.6 (5.5 to NE) months in the 600-mg fed arm, and 11.9 (8.2 to 30.4) months in the 750-mg fasted arm (S6 Table). The 12-, 24-, and 36-month PFS rates (95% CI) as per both BIRC and investigator assessments in the 450-mg fed arm of the Asian subgroup were numerically higher with overlapping CIs compared with 750-mg fasted arm (Fig. 3A, S6 Table).
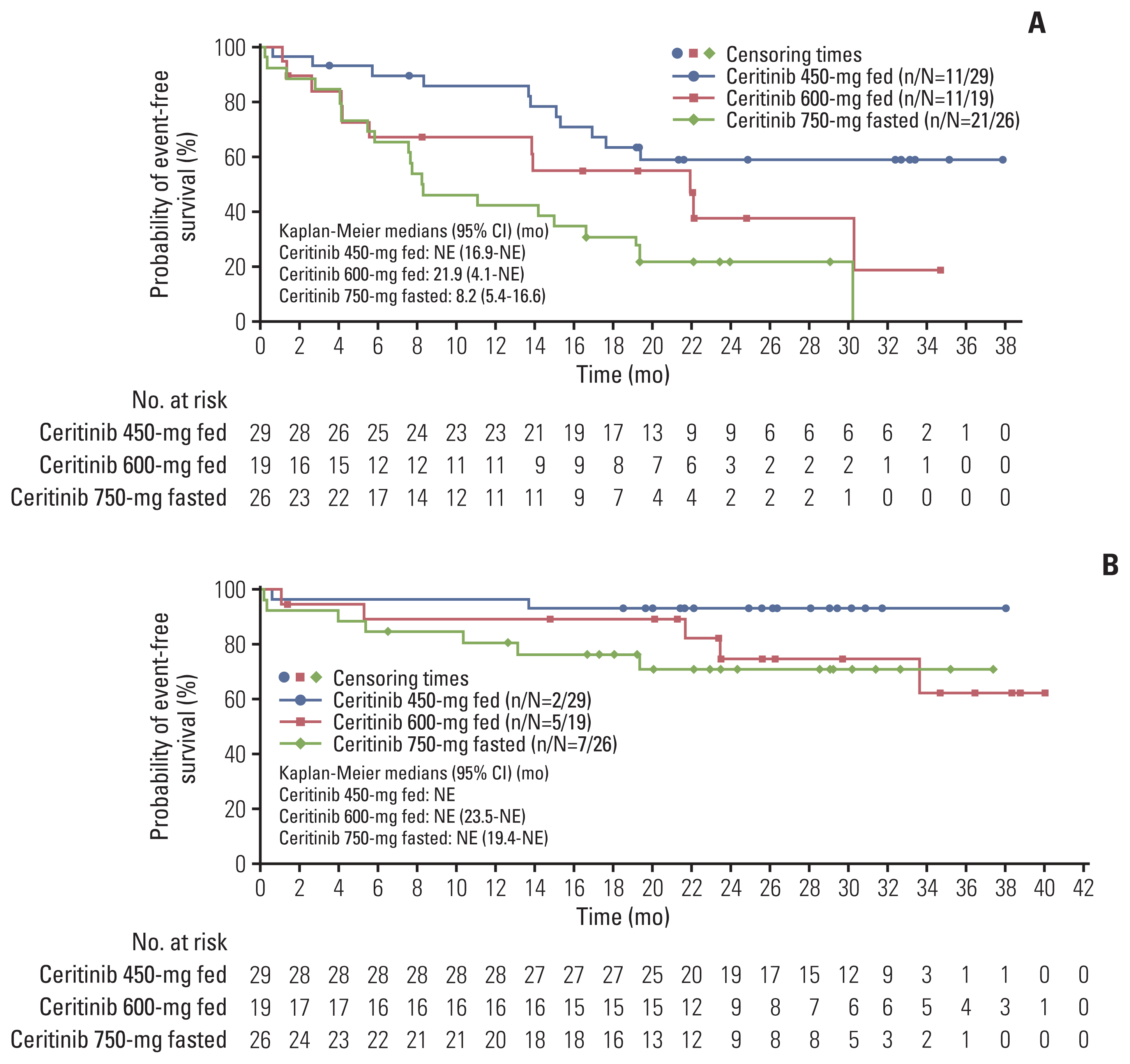
4) Overall survival
The median OS was not estimable for any of the three arms (Fig. 3B). The 24-month OS rate (95% CI) in the 450-mg fed arm was 93.1% (75.1 to 98.2) compared with 74.8% (45.3 to 89.9) in the 600-mg fed and 70.9% (47.9 to 85.1) in the 750-mg fasted arm. The 24-month OS rate (95% CI) was numerically higher with overlapping CIs in the 450-mg fed arm of the Asian subgroup compared with treatment-naïve patients in the 450-mg fed arm of the FAS. The efficacy outcomes assessed by the investigator were consistent with BIRC-assessed outcomes.
(1) Drug exposure
Patients in the 450-mg fed arm received a higher median relative dose intensity of ceritinib (96.7% [range, 34.1 to 100.0]) compared with patients in the 600-mg fed arm (77.8 [range, 25.6 to 100.0]) and 750-mg fasted arm (85.82% [range, 49.5 to 100.0]). Patients in the 450-mg fed arm had a longer median duration of exposure compared with those in the 750-mg fasted arm (96.14 [range, 2.3 to 165.1] weeks vs. 82.21 [range, 0.3 to 162.6] weeks).
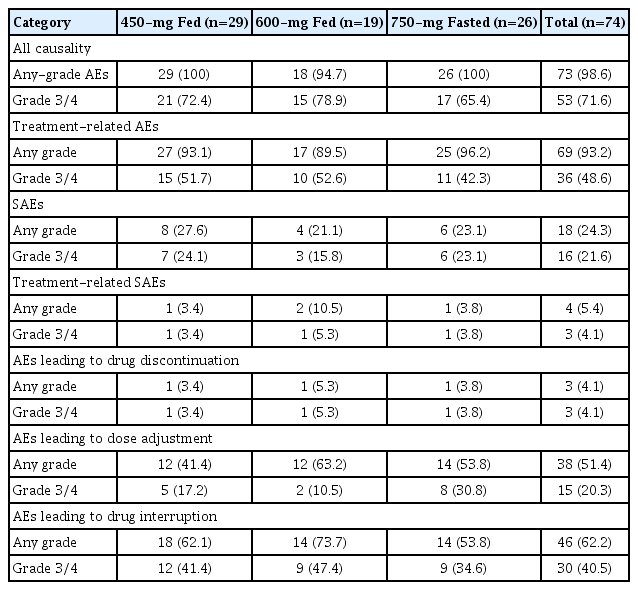
3. Safety
Any-grade AEs were experienced by 73 patients (98.6%). Key safety results are summarized in Table 3. The most frequent AEs regardless of study drug relationship occurring in ≥ 25% of all patients (S7 Table) were diarrhea (any grade, 68.9%; grade 3/4, 2.7%), increased alanine aminotransferase (any grade, 56.8%; grade 3/4, 31.1%), vomiting (any grade, 55.4%; grade 3/4, 1.4%), increased aspartate aminotransferase (any grade: 48.6%; grade 3/4: 18.9%), nausea (any grade, 47.3%; grade 3/4, 1.4%), increased gamma-glutamyl transferase (any grade, 31.1%; grade 3/4, 18.9%), and abdominal pain (any grade, 28.4%; grade 3/4, none). Any-grade GI toxicity (group event for nausea, diarrhea, and vomiting) occurred in 82.8%, 78.9%, and 96.2% of patients in the 450-mg fed, 600-mg fed, and 750-mg fasted arms, respectively (Table 4). Grade 3/4 GI toxicity occurred in one patient (3.4%) in the 450-mg fed arm, one patient (5.3%) in the 600-mg fed arm, and two patients (7.7%) in the 750-mg fasted arm.
Discussion
Overall, the results from this subgroup analysis of ASCEND-8 in treatment-naïve Asian patients with ALK+ advanced/metastatic NSCLC showed that ceritinib administered at a dose of 450 mg/day with a low-fat meal yielded high response and clinical efficacy, comparable with the efficacy observed in the general treatment-naïve study population of the ASCEND-8 study [12]. In this subgroup analysis of Asian patients, those in the 450-mg fed arm showed a numerically higher ORR per BIRC (82.8%) compared with those in the 750-mg fasted arm (76.9%). Furthermore, the objective response to ceritinib in Asian patients in the 450-mg fed arm of the ASCEND-8 study was numerically higher than in Asian patients in the ASCEND-4 study who were administered ceritinib at a dose of 750 mg/day under fasted conditions (65.8%) [13]. These data should be interpreted with caution as this is a cross-trial comparison of two studies, and this subgroup analysis was performed on a small sample size. It is noteworthy to mention that the ORR by BIRC observed with ceritinib 450-mg fed in Asian patients (82.8%) in the present study is consistent with the ORR reported in Asian patients treated with crizotinib in clinical trials (ALESIA, 77% [14]; ALEX, 76.8% [15]; PROFILE 1014, 70% [16]; PROFILE 1029, 87.5%) [17]. Furthermore, the ORR in Asian patients in the 450-mg fed of this study is also consistent with Asian patients treated with alectinib in the ALEX (81.2%) and ALESIA (91%) trials [14,15].
Although the 450-mg fed arm appears favorable in a Kaplan-Meier analysis of PFS and OS, no conclusions can be drawn from this subgroup analysis regarding the survival benefits of ceritinib 450 mg/day administered with a low-fat meal compared with 750 mg/day administered under a fasted state due to the small sample size and lack of statistical power for the comparison by the study design. While acknowledging the limitation of cross-trial comparison, the 3-year OS rate (93.1%; 95% CI, 75.1% to 98.2%) observed in the ceritinib 450-mg fed arm of this study is numerically higher than that of crizotinib (57%; 95% CI, 42.1% to 65.9%) and alectinib (67%; 95% CI, 59.1% to 74.8%) reported in the phase 3 ALEX trial [18]. Furthermore, the results from a recent network meta-analysis study showed ceritinib at a dose of 450 mg/day with a low-fat meal was more cost-effective compared with crizotinib and alectinib as a front-line therapy in patients with advanced ALK+ NSCLC [19].
The brain is a frequent site of metastasis, disease manifestation, and disease progression in patients with ALK+ NSCLC [20–24] and is associated with poor prognosis [21,22,24]. The results from the ASCEND-7 trial evaluating ceritinib in ALK+ patients with active brain metastases and/or leptomeningeal carcinomatosis further substantiate the intracranial/extracranial antitumor activity of ceritinib [25]. In this subgroup analysis, Asian patients with brain metastasis at baseline showed a high ORR to ceritinib across study arms. This is consistent with previous studies showing the promising clinical activity of ceritinib in treatment-naïve and pretreated patients with brain metastasis [3,8]. It is noteworthy that patients with brain metastasis at baseline in the 450-mg fed arm showed numerically higher ORR per BIRC (76.9%; 95% CI, 46.2% to 95.0%) compared with the 750-mg fasted arm (71.4%; 95% CI, 29.0% to 96.3%); however, the small sample size limits any conclusive interpretations from these results.
The safety profile of ceritinib in Asian patients was consistent with that of the global patient population in the ASCEND-8 study [12]. Importantly, the incidence of all-cause any-grade GI toxicity (group event for nausea, diarrhea, and vomiting) was lower in the 450-mg fed arm (82.8%) compared with the 750-mg fasted arm (96.2%). Furthermore, grade 3/4 GI toxicity was infrequent in both the arms, with one patient (vomiting) in the 450-mg fed arm and two patients (diarrhea) in the 750-mg fasted arm.
Factors such as diet, lifestyle and genotype may affect drug metabolism, efficacy, safety and clinical outcomes of a drug, and therefore, a subgroup analysis in Asian patients is of significant importance for the clinical management of Asian patients with ALK+ NSCLC treated with ceritinib. Although the sample size was low, which is an inherent limitation of this ASCEND-8 Asian subgroup analysis and the efficacy outcomes were not powered to evaluate efficacy outcomes, the benefit to risk profile of ceritinib 450 mg/day taken with food as observed in this study is of clinical relevance for treatment-naïve Asian patients with ALK+ NSCLC.
In this subgroup analysis of the ASCEND-8 study in treatment-naïve Asian patients with advanced ALK+ NSCLC, the ceritinib 450-mg fed dose demonstrated a favorable benefit-risk profile, supporting the use of ceritinib 450-mg fed as the recommended dose of ceritinib in this patient population.
Supplementary Information
Supplementary materials are available at Cancer Research and Treatment website (https://www.e-crt.org).
Notes
Electronic Supplementary Material
Supplementary materials are available at Cancer Research and Treatment website (https://www.e-crt.org).
Ethical Statement
The study protocol was approved by the independent ethics committee and/or institutional review board for each center as per the local regulations. This clinical study was designed and implemented in accordance with the principles of the Declaration of Helsinki and the Good Clinical Practice guidelines of the International Conference on Harmonization, with applicable local regulations. Written informed consent was obtained from each patient before screening.
Author Contributions
Conceived and designed the analysis: Cho BC, Geater S.
Collected the data: Cho BC, Kim DW, Batra U, Park K, Kim SW, Yang CT, Voon PJ, Sriuranpong V, Babu KG, Geater S.
Contributed data or analysis tools: Cho BC, Kim DW, Batra U, Park K, Kim SW, Yang CT, Voon PJ, Sriuranpong V, Babu KG, Amin K, Wang Y, Sen P, Slimane K, Geater S.
Performed the analysis: Cho BC, Kim DW, Batra U, Park K, Kim SW, Yang CT, Voon PJ, Sriuranpong V, Babu KG, Amin K, Wang Y, Sen P, Slimane K, Geater S.
Wrote the paper: Cho BC, Kim DW, Batra U, Park K, Kim SW, Yang CT, Voon PJ, Sriuranpong V, Babu KG, Amin K, Wang Y, Sen P, Slimane K, Geater S.
Conflicts of Interest
Byoung Chul Cho has received research funding from Novartis, Bayer, AstraZeneca, MOGAM Institute, Dong-A ST, Champions Oncology, Janssen, Yuhan, Ono, Dizal Pharma, Merck, Sharp & Dohme, Abbvie, Medpacto, GIInnovation, Eli Lilly, Blueprint medicines and Interpark Bio Convergence Corp. He also receives royalty from Champions Oncology. He is a consultant for Novartis, AstraZeneca, Boehringer-Ingelheim, Roche, Bristol Myers Squibb, Ono, Yuhan, Pfizer, Eli Lilly, Janssen, Takeda, Merck, Sharp & Dohme, Janssen, Medpacto, Blueprint medicines and serves on the advisory board of KANAPH Therapeutic Inc, Brigebio therapeutics, Cyrus therapeutics, Guardant Health, Joseah BIO. He is on the board of directors for Gencurix Inc and Interpark Bio Convergence Corp. He is a founder of the DAAN Biotherapeutics and owns stocks of TheraCanVac Inc, Gencurix Inc, Bridgebio therapeutics, KANAPH Therapeutic Inc, Cyrus therapeutics and Interpark Bio Convergence Corp.
Dong-Wan Kim declares research funding to his institution from Novartis, Alpha Biopharma, Amgen, Astrazeneca/Medimmune, Boehringer-Ingelheim, Daiichi-Sankyo, Hanmi, Janssen, Merus, Mirati Therapeutics, Merck, Sharp & Dohme, Ono Pharmaceutical, Pfizer, Roche/Genentech, Takeda, TP Therapeutics, Xcovery, Yuhan, Chong Keun Dang, Bridge BioTherapeutics and GlaxoSmithKline. He serves on the advisory board of Amgen, AsatraZeneca, Bristol Myers Squibb/Ono, Daiichi-Sankyo, GlaxoSmithKline, Merck, Sharp & Dohme, Janssen, Pfizer, SK Biopharm, Takeda, Yuhan and also reports travel support for advisory board meetings from Amgen and Daiichi-Sankyo.
Virote Sriuranpong has received research grants from AsraZeneca, Merck, Sharp & Dohme, Novartis, Regeneron, Roche and honoraria for lectures from AsraZeneca, Astellas, Bristol Myers Squibb, Novartis, Pfizer, and Roche.
Ullas Batra, Keunchil Park, Sang-We Kim, Cheng-Ta Yang, Pei-Jye Voon, K. Govind Babu, Sarayut Geater have nothing to disclose.
Khalid Amin, Yingbo Wang, Paramita Sen and Khemaies Slimane are employees of Novartis and own stocks/shares.
Acknowledgments
This study was funded by Novartis Pharmaceuticals Corporation. The authors express their sincere gratitude to the participating patients, their families, all of the study co-investigators, and research contributors. Medical writing support was funded by Novartis Pharmaceuticals Corporation and was provided by Varunkumar Pandey, Novartis Healthcare Pvt Ltd. (Hyderabad, India).