Who Will Benefit from Charged-Particle Therapy?
Article information
Abstract
Charged-particle therapy (CPT) such as proton beam therapy (PBT) and carbon-ion radiotherapy (CIRT) exhibit substantial physical and biological advantages compared to conventional photon radiotherapy. As it can reduce the amount of radiation irradiated in the normal organ, CPT has been mainly applied to pediatric cancer and radioresistent tumors in the eloquent area. Although there is a possibility of greater benefits, high set-up cost and dearth of high level of clinical evidence hinder wide applications of CPT. This review aims to present recent clinical results of PBT and CIRT in selected diseases focusing on possible indications of CPT. We also discussed how clinical studies are conducted to increase the number of patients who can benefit from CPT despite its high cost.
Introduction
Radiation therapy is an essential element in cancer treatment, with some estimates that approximately 25% to 30% of cancer patients undergo radiotherapy during their treatment course in Korea [1]. Charged-particle therapy (CPT) using proton or carbon ion, has physical and biological advantages over conventional X-ray therapy. Bragg peak and sharp penumbra allows high tumor dose while saving normal tissue and reduced spread dose. Moreover, carbon ion has greater relative biological effectiveness (RBE) than photon or proton.
Unique physical properties of particle therapy enable dose escalation to the tumor while limiting toxicity in normal tissues, which increases the therapeutic window for cancer patients. Indication for CPT includes unresectable tumors located in eloquent areas or patients with poor organ function, which are of serious concern when treated with X-ray radiotherapy. Proton beam therapy (PBT) can also benefit pediatric cancer patients because it can reduce late side effects and secondary malignancy. In addition, because of the high linear energy transfer (LET) of carbon ion radiotherapy (CIRT), the general indications for CIRT are radioresistant tumor types [2–6].
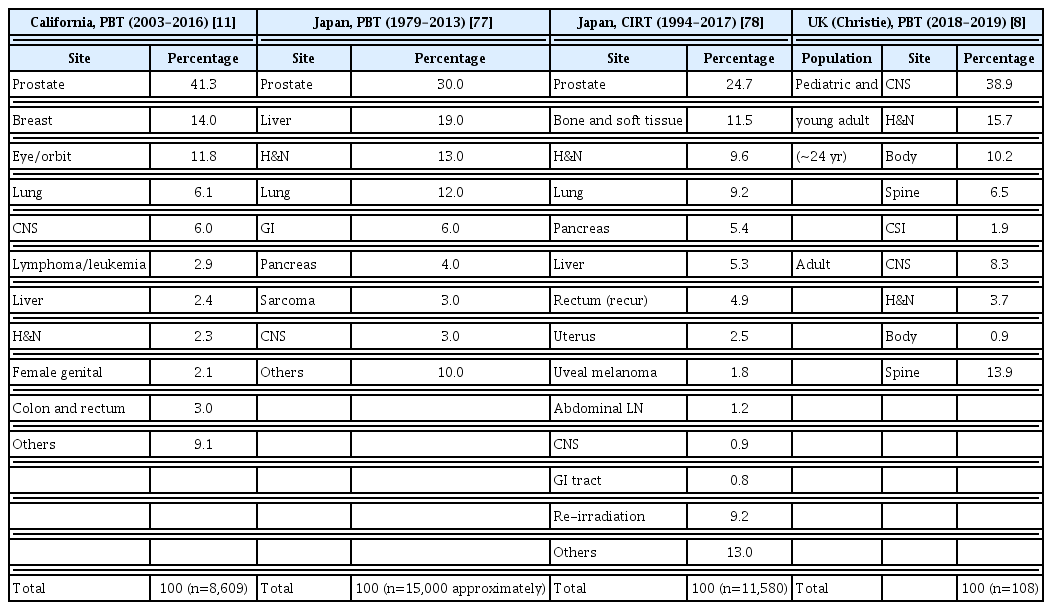
However, high set-up cost of particle therapy facilities makes it difficult to be widely applied to all cancer patients. Countries with CPT facilities have their own recommendation or public health insurance indication (Table 1) [7–10]. Pediatric cancer is the most common indication for PBT, as concerns regarding late effects. CPT is also indicated for certain rare cancers in adults such as tumors located in the eye, skull base and re-irradiation cases because of concern about radiation damage to adjacent normal tissues. Although there is a lack of consensus regarding the comparative effectiveness of CPT for common adult cancer types, the benefits of CPT may be demonstrated in a subset of patients with prostate, lung, liver, pancreas, and breast cancers [11].
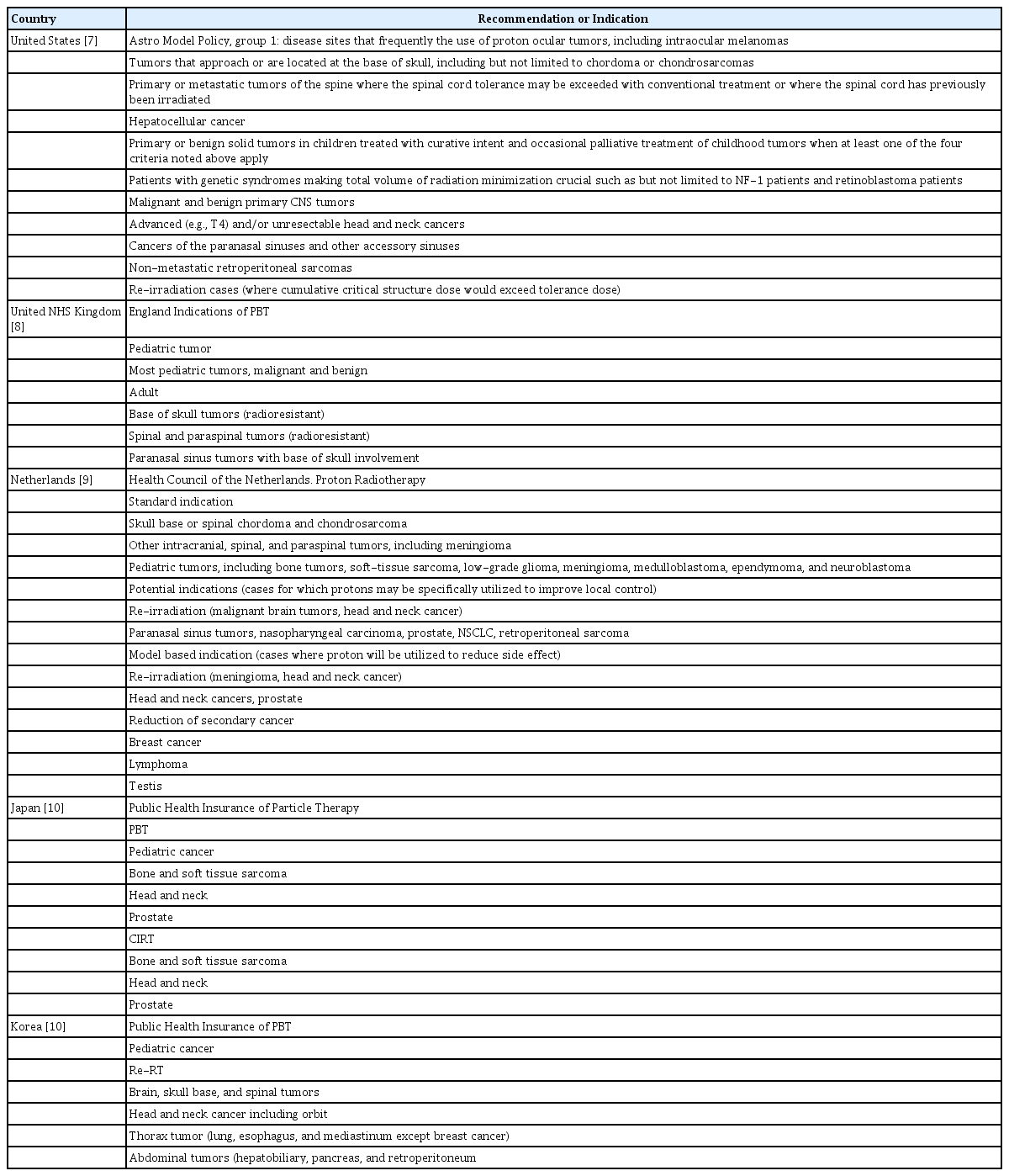
Recommended or public health insurance covered indications for charged-particle therapy from several countries
In the following sections, we present recent clinical outcomes of PBT and CIRT in selected diseases. In addition, we propose clinical research methods to increase the number of patients who can benefit from CPT, given the limitation of its high cost.
Clinical Applications of CPT
1. Pediatric cancer
Childhood cancer survivors who undergo radiotherapy are at high risk of deleterious effects on growth and development and of tissue late effects, including secondary malignancies.
In addition to the long life expectancy of children, germline predisposition to second malignancy of the childhood cancer is also the reason we should reduce normal tissue irradiation as much as possible in pediatric cancer patients. Hence, PBT has recently attracted worldwide attention as a radiotherapy modality for pediatric cancer.
Long-term outcome data showing favorable toxic profile of PBT for the treatment of pediatric central nervous system cancer such as medulloblastoma has been published [12–14]. Furthermore, the relationship between PBT and secondary cancer has been clarified. Sethi et al. [15] reported that the incidence of secondary cancer in 86 patients with retinoblastoma who were treated with PBT (n=55) or photon radiotherapy (n=31). Ten-year cumulative incidence of in-field secondary malignancies with PBT was lower than with conventional photon radiotherapy (0% vs. 14%, p= 0.015).
There is only limited research on the role of CIRT for pediatric cancer. Even though it could be efficient for radioresistant tumors of the children, long-term safety data is needed because of dosimetric uncertainty different from PBT.
2. Uveal melanoma
Uveal melanoma is a rare disease that has traditionally treated with surgery, brachytherapy, and particle therapy. Treatment with PBT was started in 1975 and has been beneficial in preserving vision and controlling disease [16,17]. A meta-analysis reported a superior local control (LC) rate and fewer adverse events in PBT compared to brachytherapy [18]. Treatment using carbon ions also showed promising results, with an LC rate of 97.4% at 3 years [19].
3. Head and neck cancer
CPT has been applied to radioresistant tumors other than squamous cell carcinoma of the head and neck. Adenoid cystic carcinoma arising from the salivary gland is a typical example with promising results using particle therapy. Although surgery and postoperative radiotherapy are the standard of care, it is still difficult to treat cancerous lesions in locations unfit for surgery, and locally advanced cancer with perineural invasion makes treatment difficult. PBT [20,21] or CIRT [22,23] used as a boost or primary treatment showed a high LC rate with acceptable toxicity.
Another suitable indication for particle therapy for head and neck cancer is malignant melanoma [24,25]. A multicenter retrospective study in Japan reported promising results in 260 patients with mucosal melanoma of the head and neck when treated with CIRT [25]. Seventy-eight patients had lesions in the nasal cavity, and concurrent chemotherapy was administered to 129 patients. The LC rate and overall survival (OS) rates at 2 years were 83.9% and 69.4%, respectively. Grade-3 or higher late toxicities were reported in 19% of the patients.
In addition, the treatment outcomes of particle therapy have been reported for tumors arising from the paranasal sinus and nasal cavity, including squamous cell carcinoma and rare histologic tumors in the head and neck [26–29]. A systematic review and meta-analysis concluded that particle therapy could be associated with better outcomes in tumors in the paranasal sinus and nasal cavity [26]. In the case of re-irradiation, particle therapy could have advantages owing to its dose gradient and decreased integral dose [30].
4. Bone and soft-tissue sarcoma
Chordoma is a rare bone tumor with slow growth. However, these tumors tend to be locally aggressive and have a high local recurrence rate. Moreover, they are located along the neural axis, clivus, and sacrum, where total resection is almost impossible to achieve. For skull-base chordoma, the proximity to vital structures also makes it difficult for definitive radiotherapy to be applied without complications. Therefore, particle therapy is preferred over conventional radiotherapy therapy to deliver a sufficient dose to the designated site. The LC rate has been reported to be 69.7%–87.9% [31–34] with an OS rate range of 80%–88.3% at 5 years [31–33]. For sacral chordoma, a similar outcome has been reported for PBT [34–36]. Although direct comparison of photon and particle therapy has not been reported, a recent analysis of 863 patients with chordoma and chondrosarcoma from the National Cancer Center Database showed that PBT was associated with an improved OS compared to photon radiotherapy [37]. CIRT results from the treatment of skull-base chordoma and chondrosarcoma were summarized in a recent meta-analysis [38]. An estimated LC rate of 80% and 89% at 5 years was reported in chordoma-only and chondrosarcoma-only studies, respectively. Estimates of OS probability at 5 years in chordoma-only studies and chondrosarcoma-only studies were 94% and 95%, respectively. An active investigation of randomized trials comparing PBT and CIRT of skull-base chordoma and chondrosarcoma is ongoing [39,40]. CIRT of the sacral chordoma also showed promising results, with OS rates ranging from 78% to 84% at 5 years [41–44].
Possible indication for CIRT include soft-tissue sarcomas arising from the axial skeleton or retroperitoneal space because they are difficult to approach using surgical methods [45–47].
5. Lung cancer
1) Early-stage non-small-cell lung cancer
For inoperable stage I–II non–small cell lung cancer (NSCLC), stereotactic body radiotherapy (SBRT) of photons is widely used in the clinic. By delivering an ablative radiation dose to the gross tumor within a few fractions, we can achieve an LC rate of over 90%. However, a high tumor-control rate through SBRT using photons cannot always be achieved in clinical practice. Such conditions include patients who have large or centrally located diseases and are prone to severe side effects due to poor general conditions, decreased pulmonary function, or recurrent diseases [48]. Particle therapy could be advantageous over SBRT under such conditions. For large T2 tumors, Iwata et al. [49] reported a 4-year LC rate and OS of 75% and 58% with CPT, respectively. Moreover, particle therapy can safely obtain high LCs in cases of centrally located tumors [50]; in patients with poor pulmonary function, such as those with idiopathic pulmonary fibrosis [51–53]; or the elderly [54]. There are no randomized controlled trials (RCTs) that directly compare SBRT with particle therapy. However, Chi et al. [55] conducted a systematic review and meta-analysis, and reported that even with more advanced tumors in terms of size and T category, patients treated with particle therapy were found to have significantly better OS and progression-free survival (PFS) than those treated with photon SBRT in the weighted multivariate analysis [55]. A retrospective comparison of CIRT with SBRT demonstrated a positive efficacy profile of CIRT for early-stage NSCLC over SBRT [56].
When CIRT was used, 18 fractions over 6 weeks were reduced to nine fractions over 3 weeks and finally to four fractions over one week with acceptable efficacy and safety [57–59]. A recent study on CIRT reported that even one fraction could achieve favorable outcomes with acceptable toxicity [60,61]. In addition to the advantages of providing convenience to the patient by providing single-day treatment, CIRT can provide an advantage in terms of cost-effectiveness because the number of particle therapy facilities is limited.
2) Locally advanced NSCLC
It is well known that improved LC in locally advanced NSCLC is associated with improved OS. The Radiation Therapy Oncology Group 0617 trial failed to show that increased doses guarantee improved outcomes. However, excessive doses to the lungs and heart are considered to be associated with poor outcomes [62,63]. Particle therapy can deliver an adequate radiation dose to the target while avoiding normal tissue in patients with locally advanced NSCLC. A retrospective and phase-I/II study reported the feasibility of PBT [64,65] and CIRT [66–70] for locally advanced NSCLC.
Even with the known advantages of particle therapy, one randomized phase-II trial performed to date reported that PBT did not have benefits over photons in advanced NSCLC [71]. In this study, PBT did not show benefits in LC or incidence rates of grade-3 pneumonitis in patients with stage IIB - oligometastatic diseases. However, the results of this study should be interpreted carefully. The sample of the study was very heterogeneous, with variability in disease states and target coverage, and randomization was performed after the initial screening with treatment plans of the two modalities. Finally, the learning curve of PBT delivery was developed through the study period instead of being established before the study began [72]. The patient population that will benefit from particle therapy should be well defined, and studies that integrate chemotherapy and immunotherapy should be conducted in the future to reflect current clinical practices.
There are many limitations in delivering particle therapy to organs that move due to respiratory motion, such as the lungs. The uncertainty arising from the interplay between the scanning beam and moving target needs to be controlled. Recently, technical advances have been made to control respiratory motion, such as four-dimensional computed tomography, respiratory-gated systems, and fast scanning systems, as previously discussed [73,74].
6. Liver cancer
The role of radiotherapy in the management of hepatocellular carcinoma (HCC) is increasing in clinical practice. Recent progress in radiation oncology has enabled the concentration of higher-radiation-dose photon beams to the tumor surrounded by relatively radiosensitive normal liver tissue, and SBRT has become one of the ablative treatment options for HCC [75]. The results of particle therapy for HCC using protons have been reported at Tsukuba University in Japan [76]. The second most common cancer treated with PBT is HCC in Japan [77]. The CIRT facility in Japan has also started treatment of HCC, and currently 5%–6% of the treated patients are HCC patients [78]. Pooled analyses of the results of HCC patients treated with particle therapy have been reported [79]. By analyzing 16 studies containing reports from 17 cohorts with a total of 1,516 patients treated with protons or carbon, the authors concluded that the LC rate was 87% and OS was 59% at 3 years. This study showed favorable outcomes similar to those of SBRT [80], but it should be noted that the population of this study included patients with large tumors, portal vein tumor thrombosis (PVTT), and liver cirrhosis. The indications for particle therapy of HCC have not yet been determined, but by exploiting superior dose profiles, we can apply particle therapy in difficult cases of clinical situations that are ineligible for photon therapy [81,82]. These conditions include large tumor sizes [83–85], central lesions near the porta hepatis [86–88], tumors with PVTT [89–91] or inferior vena cava tumor thrombosis [92], tumors with close proximity to gastrointestinal (GI) organs [93], patients with poor liver function or cirrhosis [94], and elderly patients [95].
Selected studies in particle therapy reported excellent tumor control and OS [96,97]. Sanford et al. [82] reported a superior OS with PBT over photons-based therapy in their retrospective studies. Moreover, studies comparing the efficacy of particle therapy with other local treatment modalities, such as radio-frequency ablation [98] and transarterial chemoembolization [99,100] have been reported recently. Regarding the fractionation schedule of CIRT, an attempt to reduce the fraction schedule from four to two would increase the convenience to patients receiving the therapy [97,101]. Based on these results, studies should be conducted to clarify the benefits of particle therapy in the management of HCC.
The effectiveness of particle therapy in treating unresectable cholangiocarcinoma has also been investigated [102–104]. In these studies, particle therapy showed a high LC rate, but distant metastasis still limited the OS gain. Further studies are needed to establish optimal treatment strategies for unresectable cholangiocarcinoma by combining chemotherapy and particle.
7. Pancreatic cancer
1) Unresectable locally advanced disease
Multi-agent chemotherapy is the standard treatment for unresectable, locally advanced pancreatic cancer. The role of local therapy remains controversial. The LAP07 trial showed significantly longer period without treatment (6.1 months vs. 3.7 months) and reduced local tumor progression (32% vs. 46%, p < 0.05) in the chemo-radiotherapy group after induction chemotherapy, even though it did not show an OS benefit compared to the chemotherapy-alone group. Several studies that performed autopsies reported that approximately 30% of patients with pancreatic cancer did not show evidence of distant metastasis at death. In addition, the possibility of OS improvement by improving the LC through SBRT or dose escalation has been shown in recent studies [105,106]. Therapeutic gains can be obtained in pancreatic cancer from particle therapy because of the limitations in sufficient dose escalation of X-rays due to close proximity to the duodenum, stomach, etc. With these theoretical advantages, favorable outcomes of unresectable pancreatic cancer treated with PBT have been reported [107–113]. These studies used diverse fraction sizes of 1.8–4.5 Gy, and reported favorable toxicity profiles [114]. Moreover, pancreatic cancers are known to be hypoxic; thus, the radiobiological advantages of carbon ions with high-LET property can be maximized [115]. A few clinical trials and retrospective studies have evaluated CIRT for unresectable pancreatic cancer in Japan [116–121]. Shinoto et al. [117] reported the treatment results of 64 unresectable pancreatic cancer patients treated with CIRT. Chemotherapy was administered before CIRT to 78% of patients, and 88% of the patients were treated with concurrent gemcitabine and/or S-1. In 75% of patients, the distance between the tumor and the GI tract was within 5 mm. The prescribed dose was 55.2 Gy (relative biological effectiveness, RBE) with 12 fractions. They reported an excellent 2-year OS rate of 53%, and grade-3 ulcer/bleeding was reported in two patients. Two multi-institutional prospective phase-III trials comparing photon intensity modulated radiotherapy (IMRT) and CIRT are ongoing (NCT03403049 and NCT03535182).
2) Potentially resectable disease
In resectable or borderline-resectable pancreatic cancer, the role of neoadjuvant treatment is actively being investigated [122–124]. The potential increase in R0 resection rate, decrease in locoregional recurrence rate, and better patient compliance could be the advantages of incorporating radiotherapy in the neoadjuvant setting. Retrospective and phase-I/II trials of PBT [107,122,124–127] and CIRT [119,128] have been reported, but more prospective trials are needed for radiotherapy, including particle therapy, to be established as a standard treatment.
8. Prostate cancer
Radiotherapy is used as a definitive treatment for patients with prostate cancer. Due to the dose-dependent tumor-control rate and frequent late GI and genitourinary toxicity, IMRT is commonly used in the clinic. PBT and CIRT have advantageous physical properties over photon-based radiotherapy because they can provide adequate dose distribution with fewer beam portals. Theoretically, PBT and CIRT may seem ideal for subsequent dose escalation with minimal toxicity. However, due to the lack of clinical trials that directly compare the effects of particle therapy with photon beam radiotherapy, it is difficult to evaluate the superiority of particle therapy. The limited number of institutions that can conduct clinical studies involving particle therapy is also a major limitation.
Despite these limitations, many studies have reported that PBT [129–133] and CIRT [134–138] show lower toxicity compared to IMRT [139]. Prostate cancer is characterized by a very low α/β ratio, thus leading to the theoretical superiority of particle therapy with a hypofractionated schedule over conventional photon radiotherapy. With 12 fractions at 3 weeks, CIRT showed superior 5-year biochemical-free survival (88%–92%) in high-risk prostate cancer patients compared to the previously reported result of the IMRT group [134,135].
As particle therapy is a limited medical resource, there are constraints on its widespread use for prostate cancer. With a gradual decrease in the cost of the facility, efficiency of the hypofractionation schedule, low toxicity profile, high biochemical control rate of intermediate or high-risk disease, and reduced secondary cancer risk [140], it is expected that particle therapy of prostate cancer will become a cost-effective treatment in the near future. Since April 2018, particle therapy for prostate cancer has been covered by the Japanese National Health Insurance System [139]. The majority of patients treated with CPT in Japan were prostate cancer patients [77,78].
9. GI malignancy
1) Esophageal cancer
There is still little evidence on the role of particle therapy in the treatment of esophageal cancer. However, many studies have shown that toxicity can be reduced by reducing the dose to the heart and lungs during particle therapy, and recent studies have reported that acute hematologic toxicity of grade 4 or higher can be reduced using PBT compared to IMRT in the treatment of esophageal cancer [141]. Moreover, PBT reduced the risk and severity of adverse events compared to IMRT while maintaining a similar PFS in a randomized phase-IIB trial [142]. Regarding oncological outcomes, there are no RCTs that directly compare PBT and photon therapy, and a retrospective study has reported the improved OS of PBT over IMRT [143]. Xi et al. [143] reported that patients who received PBT had significantly higher 5-year OS rates than IMRT (41.6% vs. 31.6%), and PBT was a significant factor for OS in multivariate analysis. Akutsu et al. [144] reported a clinical trial of a preoperative short-course CIRT for esophageal cancer. In their report, a complete response was achieved in 39% of the patients, and 42% of patients achieved a partial response after treatment with a total dose of 28.8 Gy (RBE) and up to 36.8 Gy (RBE) in 8 fractions. Further studies are needed to select the optimal patient to benefit from particle therapy in the treatment of esophageal cancer.
2) Recurrent rectal cancer
Local recurrence occurs in 5%–10% of patients after surgical resection of rectal cancer [145]. Therapeutic options for salvage treatment of locally recurrent rectal cancer are limited, especially in patients who have undergone preoperative radiotherapy [146]. CIRT has shown promising results in these diseases. Shinoto et al. [147] reported multi-institutional retrospective data of 224 patients treated with CIRT in Japan. They reported an LC rate of 93% and an OS rate of 73% at 3 years with limited toxicity. In the case of re-irradiation, CIRT shows promising results [148–150].
10. Breast cancer
The largest proportion of patients receiving radiotherapy are breast cancer patients. It is well known that radiation dose to the heart is associated with late cardiovascular disease after breast radiotherapy [151]. The use of protons would spare the heart from radiation and could result in fewer morbidities later in life. Thus, PBT can be cost-effective for patients with one or more cardiac risk factors. The risk factors include left breast cancer, post-mastectomy chest wall irradiation, and node-positive disease receiving for regional nodal irradiation [152].
Clinical Research to Broaden the Indication of CPT
We have to admit that there are lack of high level of evidence to support superiority of particle therapy over conventional photon therapy. RCTs are crucial in defining the role of CPT but cannot and should not be used exclusively to investigate this. RCTs between CPT and conventional photon treatment in a population that are not particularly abundant as individuals who are likely to experience a difference are not a suitable tool to the identification and quantification of the clinical benefit of CPT. Several different methodological approaches are needed. Establishing the relative role of CPT will include estimating the magnitude of benefit, which is likely to be different in different patient groups. In addition, understanding the scientific principles, and uncertainties or ensuring safety, underlying CPT is crucial to achieving greatest clinical benefit for patients [8]. These clinical studies involve clinical cohort study with long-term outcome tracking of every patient treated. Since the number of CPT center is limited, introduction of an appropriate referral system and multicenter registry are essential to successfully carry out these studies. Beside clinical studies, translational studies to maximize the possible benefit of particle therapy should be conducted in parallel. These studies include the application of ultra-high dose-rate (FLASH) therapy of proton beam, clinical application potent immunogenic effect of CPT and studies to understand basic biologic interaction of heavy ions such as carbon or oxygen ions.
There are several factors that influence the optimal number of particle therapy facilities needed in one society. The expansion of the particle therapy facility should be accompanied by studies necessary to select patients who can benefit from the CPT as described above, and at the same time, cost-effective analysis considering nationwide medical cost and changes in healthcare insurance policy should be carefully considered [153]. The proportion of the patients treated with PBT or CIRT in California, Japan, and United Kingdom were summarized in Table 2 [8,11,77,78]. Recently in United States, the number of proton centers has expanded rapidly from two centers in 2004 to 27 centers in 2020. However, decline of insurance coverage and shortage of patient brought financial problem to one-third of the institutions [154,155]. On the other hand, in United Kingdom, two proton centers opened in 2018 and 2020 and started to treat patients focusing on pediatric patients after decade-long careful planning [8]. In Nethelands, the three new proton beam therapy centers will use a normal tissue complication probability model to select patients who are expected to benefit significantly from PBT on the basis of comparative planning for individual patients [156]. In Japan, there are 18 proton centers and six carbon centers, and coverage of national health insurance for particle therapy are expanding including prostate cancer.
In Korea, two proton centers have been established from 2007 (National Cancer Center, Goyang) and 2015 (Samsung Medical Center, Seoul). National Cancer Center is planning the installation of the second cyclotron for PBT. Moreover, Seoul National University Hospital and Yonsei University Hospital decided to set up CIRT centers in Busan and Seoul, respectively, and these centers will open within 3 to 4 years. At the beginning of the expanding CPT facilities in Korea, efforts should be made to build high-level evidence that can confirm clinical benefits of particle therapy. Along with these efforts, it is necessary to carefully increase the number of patients who can benefit from CPT in consideration with the nationwide medical cost. In addition, the initial set-up cost of these facilities is gradually decreasing. Eventually, with these efforts, CPT will improve the survival rate and quality of life of cancer patients and contribute to the decline of secondary malignancy among cancer survivors.
Notes
Author Contributions
Conceived and designed the analysis: Wu HG.
Collected the data: Kim KS, Wu HG.
Contributed data or analysis tools: Wu HG.
Performed the analysis: Kim KS.
Wrote the paper: Kim KS.
Conflicts of Interest
Conflict of interest relevant to this article was not reported.
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) with a grant funded by the Korean government (MSIT) (No. 2019R1F1A1040583) and the Radiation Technology R&D program through the National Research Foundation of Korea, funded by the Ministry of Science and ICT (2019M2A2B4095126).