Oncological and Functional Outcomes of Larynx-Preserving Surgery for Hypopharyngeal Cancer: A Comparison with Definitive Radiation-Based Treatment
Article information
Abstract
Purpose
Larynx-preserving surgery (LPS) have recently gained popularity and achieved comparable oncologic outcomes to conventional radical surgery for localized hypopharyngeal cancer (HPC). In the current study, the role of LPS has been assessed thoroughly in comparison with upfront radiation therapy (RT).
Materials and Methods
We retrospectively reviewed 185 candidates for LPS with cT1–2 disease; 59 patients underwent upfront LPS while 126 patients received upfront RT, respectively. Oncological and functional outcomes were investigated and compared.
Results
Following LPS, safe margin (≥ 5 mm) was achieved in 37.3% of patients. Overall, better clinical outcomes at 5 years were achieved following upfront LPS than those following upfront RT: overall survival (OS) (72.7% vs. 59.0%, p=0.045), disease-free survival (DFS) (59.8% vs. 45.0%, p=0.039), and functional laryngeal preservation (100% vs. 89.7%, p=0.010). Although similar outcomes were observed in patients with cT1 disease, better 5-year DFS was achieved following upfront LPS in patients with cT2 disease (57.0% vs. 36.4%, p=0.023) by virtue of better local control. Despite frequent cN2–3 disease in upfront LPS group, comparable outcomes were observed between upfront RT and LPS group. However, multivariable analyses revealed that performance status and double primary cancer diagnosed within 6 months of HPC diagnosis affected OS significantly, while treatment modality per se did not.
Conclusion
Although upfront LPS could provide better local control than upfront RT in patients with cT2 disease, overall outcomes were comparable following either modality. Treatment selection of larynx-preserving approach for HPC should be individualized based on tumor and patient factors.
Introduction
The incidence of hypopharyngeal cancer (HPC) is rare, less than 0.7 per 100,000 people [1,2], and is generally associated with poor prognosis [2,3]. This could be explained by the characteristics of early cancer cell spread into one or more subsites of the hypopharynx and deeper tissues [4,5] and frequent lymphatic metastasis even with the small primary tumor [6], which frequently lead to diagnosis at the loco-regionally advanced stages [1–3]. Moreover, approximately 40% of the patients with HPC are known to suffer from a secondary cancer during their clinical course [7].
Due to the anatomical vicinity, the local therapeutic extent frequently includes the uninvolved larynx [4,5]. Conventional radical surgery for advanced primary tumor usually requires total laryngectomy to achieve an acceptable surgical safety margin, resulting in the permanent loss of laryngeal voice. Thus, non-surgical approach, upfront radiation therapy (RT) with or without chemotherapy, has been accepted as a larynx-preserving approach [6,8–14]. In addition, larynx-preserving surgery (LPS) has become an alternative to upfront RT with the advances in surgical techniques [15–17]. However, most previous comparative reports, in which treatment modality selection was based on mainly tumor-node-metastasis (TNM) staging, should have had unavoidable selection biases [6,8,11,13,14]. Assuming that the patients had tumors of the same T classification, upfront surgery would have been more frequently recommended to the patients with localized tumors and with favorable performance status. Meanwhile, upfront RT would have been applied to those with rather diffuse or infiltrative tumor or with poor performance status. Therefore, larynx-preserving approaches including LPS and RT should be re-evaluated objectively, considering tumor and host factors.
In this context, we intended to evaluate the role of LPS, which has not previously been adequately addressed, in comparison with upfront RT. We specifically focused on the patients whose tumors could be a potential candidate for LPS, based on the initial tumor characteristics.
Materials and Methods
1. Study patient
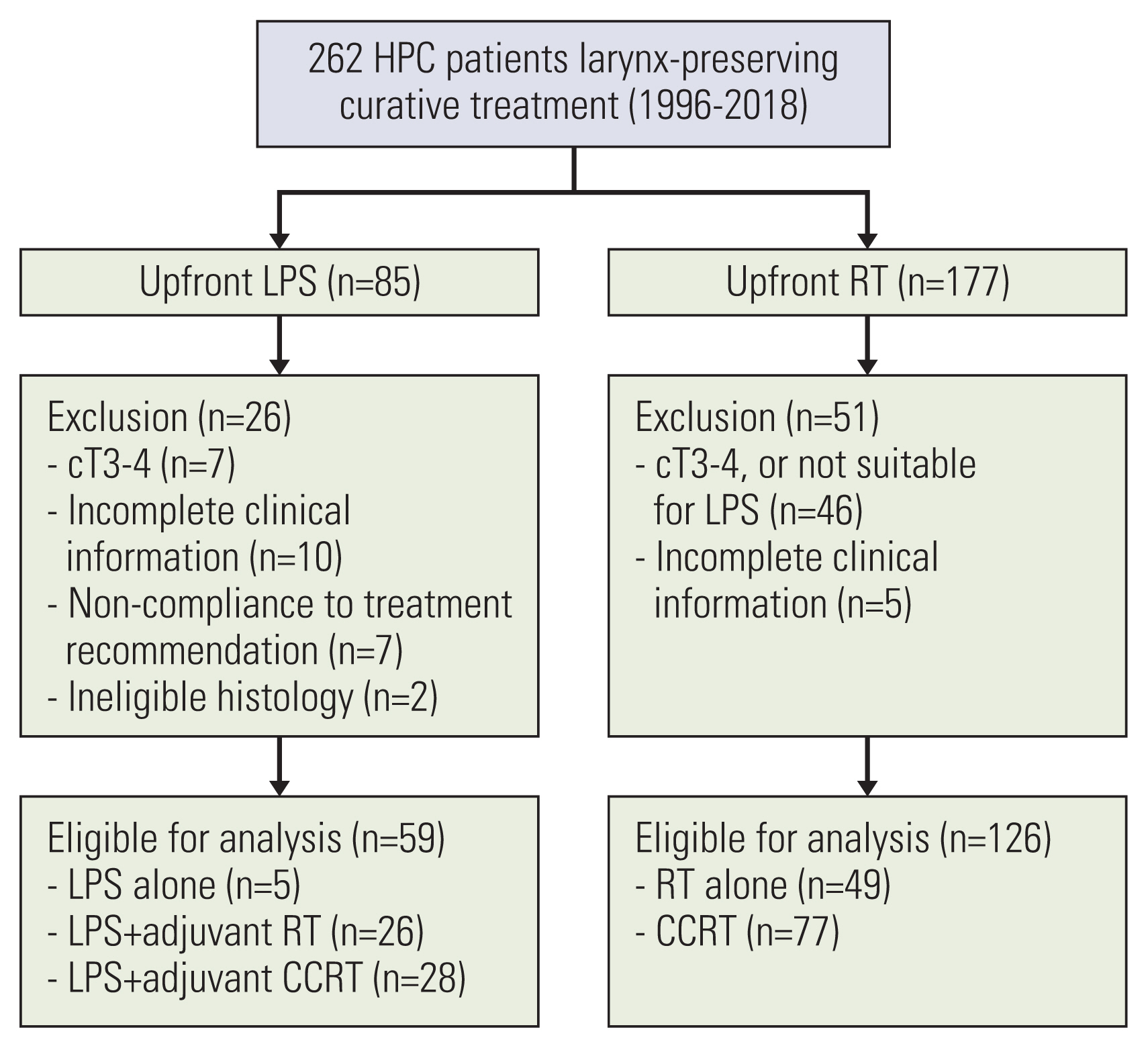
A full electronic search identified 262 patients who underwent curative larynx preserving treatment for biopsy-proven hypopharynx squamous cell carcinoma from 1996 till 2018 at the authors’ institute: 85 underwent upfront LPS; and 177 underwent upfront RT, respectively (Fig. 1). The actual treatment decision was made largely depending on the disease extent as well as the patients’ factors. In order to endorse the objective and unbiased comparisons, the suitability of LPS was thoroughly re-evaluated on each patient through a careful joint review by two head and neck surgeons (D.K., H.S.J.), based on clinical data, laryngoscopic findings, and radiological studies (computed tomography, positron emission tomography/computed tomography, or magnetic resonance imaging). Following criteria was considered for the suitability of LPS: the surface dimension of tumor, the depth of invasion, the involvement of larynx cartilage frame, function (movability) of vocal fold, the proximity to larynx cartilage (surgical safety margin), and the characteristics of tumor border. Taking the aforementioned criteria into consideration, 77 patients were excluded from the current analyses: 26 in the upfront LPS group; and 51 in the upfront RT group, respectively. Fifty-three patients (seven in upfront LPS group and 46 in upfront RT group) were mistakenly under-staged as cT2 tumors but actually turned out to have had cT3–4 tumor following the thorough reevaluation. Additional reasons for exclusion were incomplete clinical information in 15 patients (ten and five), non-compliance to the treatment recommendation by the patients in seven (seven and none), and ineligible histologies (synovial sarcoma and carcinoma in situ) in two (two and none), respectively. Finally, 185 patients (59 in the upfront LPS group and 126 in the upfront RT group) were analyzed.
2. Diagnostic work-ups and treatment decisions
The diagnostic work-ups included physical examination, endoscopic examination of the upper aerodigestive tract (laryngopharyngoscopy and esophagoscopy), computed tomography or magnetic resonance imaging, and positron emission tomography/computed tomography (or chest computed tomography, when necessary). Any suspicious metastatic lymph node detected by physical examination and/or radiological images was evaluated by aspiration cytology (ultrasonography guided or not).
The decision on the actual treatment modality, in principle, was to be made through the multidisciplinary tumor board, with the consideration not only of tumor extent, but also of the general condition and preference of each patient. However, the decision to conduct upfront LPS was mainly determined by the surgeon’s opinion on the expectancy of adequate surgical exposure and achieving clear resection margin while preserving the functioning larynx.
3. Surgical treatment: larynx-preserving partial hypopharyngectomy
Surgical approach was determined based on the primary tumor location and extent: trans-oral approach was chosen in 25 patients (42.4%); while trans-cervical pharyngotomy (open partial hypopharyngectomy) was in 34 (57.6%), respectively (S1 Table). Surgical microscope or endoscope was used, for magnification, in trans-oral approach, but did not use robotic assistance. The surgical defect was left denuded for secondary healing in 46 patients (78.0%), and was reconstructed with regional or free flap in 13 (22.0%), particularly in cases with connections between the hypopharyngeal and neck wounds, respectively. The extents of neck dissection were individually determined: radical neck dissection was performed in six patients (10.2%); modified radical neck dissection in 25 (42.4%); selective neck dissection in 16 (27.1%); and not done in 12 (20.3%), respectively. The addition of contralateral neck dissection and its extent was individually determined and applied in 39 patients (66.1%). The patients in upfront LPS group underwent curative surgical resection, with the intent to achieve adequate, defined as ≥ 5 mm tumor-free margins, through intra-operative frozen section analyses. Following LPS, the margins were adequate in 22 patients (37.3%), close (< 5 mm) in 19 (32.2%), and positive in 13 (22.0%), respectively.
4. Adjuvant or definitive radiation treatment
Among 59 patients in upfront LPS group, adjuvant RT with or without concurrent chemotherapy was performed in 54 (91.5%) with one or more pathologic risk factors: multiple lymph node metastases in 34 (57.6%); positive or close margin in 32 (54.2%); and extracapsular extension of involved lymph node in 28 (47.5%), respectively (S1 Table). Adjuvant concurrent chemoradiation therapy (CCRT) was performed in 28 patients (47.5%). Depending on the patient’s general condition and wound healing, RT was to start in 4–6 weeks, but not later than 8 weeks after surgery. The clinical target volume of RT/CCRT included the primary tumor bed with dissected lymph node levels. The typical RT/CCRT dose schedule was to deliver median 60.0 Gy (interquartile range [IQR], 59.4 to 66.0) with 2.0 Gy per fraction. Intensity-modulated RT (IMRT) without simultaneous integrated boost was applied to 38 patients (70.4%), while the rest underwent 3-D conformal RT (3DCRT).
Regarding definitive RT or CCRT, the gross tumor volume of the primary tumor and lymph node metastasis was delineated based on clinical information. Next, we adopted selective neck node irradiation in clinical target volume encompassing 2.0–2.5 cm margins from grossly involved lymph node station, followed by reduced field with 1.0–1.5 cm margins to lymph node metastasis after 36 Gy, as we previously reported [18]. IMRT was applied in 68 patients (54.0%), while 3DCRT was in 58 (46.0%) to deliver the median dose of 68.4 Gy (IQR, 68.4 to 70.0) with 2.0–2.2 Gy per fraction.
5. Chemotherapy regimens
The most common systemic therapy regimen consisted of cisplatin (40 mg/m2 every 3 weeks or 40 mg/m2 weekly) delivered to 99 patients (28 in LPS+CCRT, 71 in CCRT). Cetuximab (20 mg/m2) delivered to six patients (all in the CCRT group). Other regimens were combination therapy with cisplatin. Those regimens included Taxotere combination therapy (40 mg/m2) in 16 patients (two and 14 in the adjuvant and definitive CCRT), Cetuximab in one (LPS+CCRT), 5-fluorouracil (20 mg/m2) in seven (one and six in the adjuvant and definitive CCRT), or Pembrolizumab (20 mg/m2) in one (definitive CCRT), respectively.
6. Collection of clinical and functional data
We collected data regarding not only the oncologic outcomes (treatment failures and survival status), but also the post-treatment complications and functional status. Severe complications were defined as any clinical situation requiring hospitalization, intensive care, intervention or surgery, and included aspiration pneumonia, dyspnea, dysphagia, and wound problems (flap failure or necrosis). Restoration of oral diet of more than blended food without assistance was defined as normal swallowing without dysphagia. Functional laryngeal preservation was defined as voluntary speech pronunciation without any assisting device. Any patients who retained tracheostomy tube were regarded as having non-functional larynx. Both voice quality and swallowing function were assessed in 12–18 months of the completion of initial treatments. The voice quality was evaluated by an auditory-perceptual evaluation method for hoarseness. In this analysis, we used the G (Grade of hoarseness) score among the GRBAS scale (Grade of hoarseness, Roughness, Breathiness, Asthenia, and Strain): 0 is normal; 1 is a slight degree; 2 is a medium degree; and 3 is a high degree, respectively [19]. In addition, the swallowing function was tested by a modified barium swallowing test, giving a seven-point swallowing performance scale [20].
7. Statistical analysis
A Wilcoxon rank sum test was used to compare continuous variables between the two groups, because the data for each variable were not normally distributed. We used Fisher’s exact test or chi-squared test to examine differences in discrete variables.
All events were measured from the date of surgery (upfront LPS group) or the first date of RT (upfront RT group) to the time of the event. The rates of overall survival (OS) and disease-free survival (DFS) were estimated using the Kaplan-Meier method. Then, we conducted univariable and multivariable survival analyses using Cox proportional hazard models to identify potential prognostic factors. The outcomes are presented as p-values, hazard ratios (HRs) and 95% confidence intervals (CI). In the multivariable models, we included all variables with p < 0.05 in the univariable analysis. Potential multi-collinearity among variables was tested by calculating the variance inflation factor. In statistical comparisons, a p-value of less than 0.05 was considered significant. All statistical analyses were performed using IBM SPSS Statistics ver. 25 (IBM Corp., Armonk, NY) and R (ver. 3.6.3, R Foundation for Statistical Computing, Vienna, Austria).
Results
1. Baseline characteristics of patients in the comparison groups
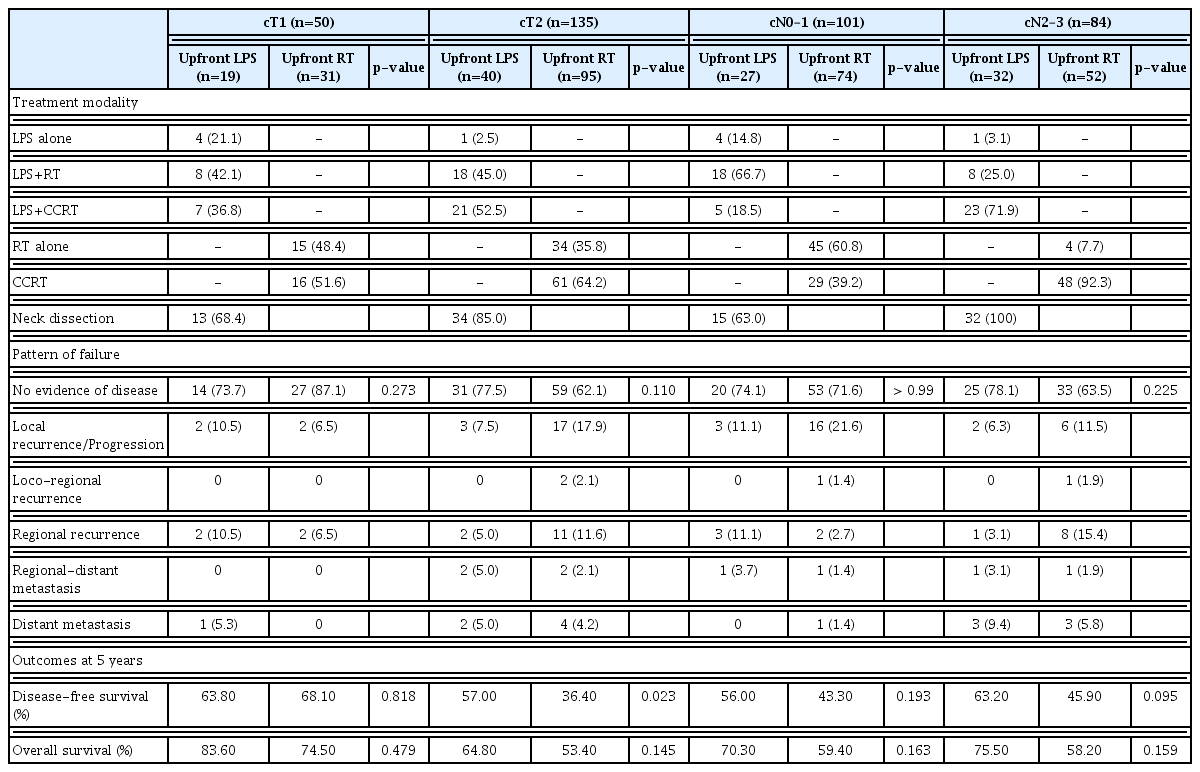
The baseline characteristics of the patients at diagnosis are summarized in Table 1. With a male predominance (95.7%), the median age of the entire cohort was 65 years (IQR, 58 to 70) and 165 patients (89.2%) had favorable Eastern Cooperative Oncology Group (ECOG) performance status of 0–1. There was no difference in ECOG performance status, underlying disease, history of alcohol drinking and underlying disease between treatment groups (Table 1, S2 Table). Although most baseline characteristics including cT stage were comparable, the proportion of cN2/N3 disease were more frequent in upfront LPS group (59.3% vs. 41.3%, p=0.001).
There were 84 patients (45.4%) who developed malignancy other than HPC: 28 (47.4%) in the upfront LPS group; and 56 (44.4%) in the upfront RT group, respectively. Twenty-six patients (14.1%) were diagnosed with another malignancy within 6 months before or after diagnosis of HPC. The most common cancer types included esophageal cancer in 49 patients (26.5%), followed by other head and neck cancer in 26 (14.1%), lung cancer in 10 (5.4%), and stomach cancer in nine (4.9%), respectively. All patients with history of other malignancy was judged to tolerate the treatment plans for HPC, and treatment interruption was necessary in no patient.
2. Oncological outcomes
With a median follow-up of 37.0 months (IQR, 11.0 to 70.0) for the entire cohort, there was no difference in follow-up period between the upfront LPS group (median, 37.0 months [IQR, 13.0 to 65.0]) and the upfront RT group (median, 35.0 months [IQR, 10.0 to 71.0]) (Table 2). There were no differences in the rates of local recurrence, regional recurrence, and distant metastasis between the groups. Forty-three patients developed local and/or regional recurrence component without distant metastasis: nine in the upfront LPS group and 34 in the upfront RT group, respectively. Among these, 38 patients (88.4%) underwent local salvage therapy, where 28 (five and 23) underwent mainly surgery, and 10 (three and seven) did mainly RT, respectively. Among 26 patients who developed local recurrence component, salvage total laryngopharyngectomy was performed in nine patients, all of who belonged to the upfront RT group. The 5-year rates of OS and DFS following upfront LPS were better than those following upfront RT (72.7% vs. 59.0%, p=0.045, and 59.8% vs. 45.0%, p=0.039, respectively) (Fig. 2).
3. Functional outcomes
Functioning larynx was more frequently preserved in the upfront LPS group (100% vs. 89.7%, p=0.010) (Table 2), however, swallowing function was similar in two groups. Among the patients with functioning larynx, the scores for voice quality and swallowing function were comparable between the groups. The incidences of severe complications, which required hospitalization, intensive care, or surgical intervention, were comparable between groups (10.2% vs. 8.7%, p=0.788).
4. Subgroup analysis
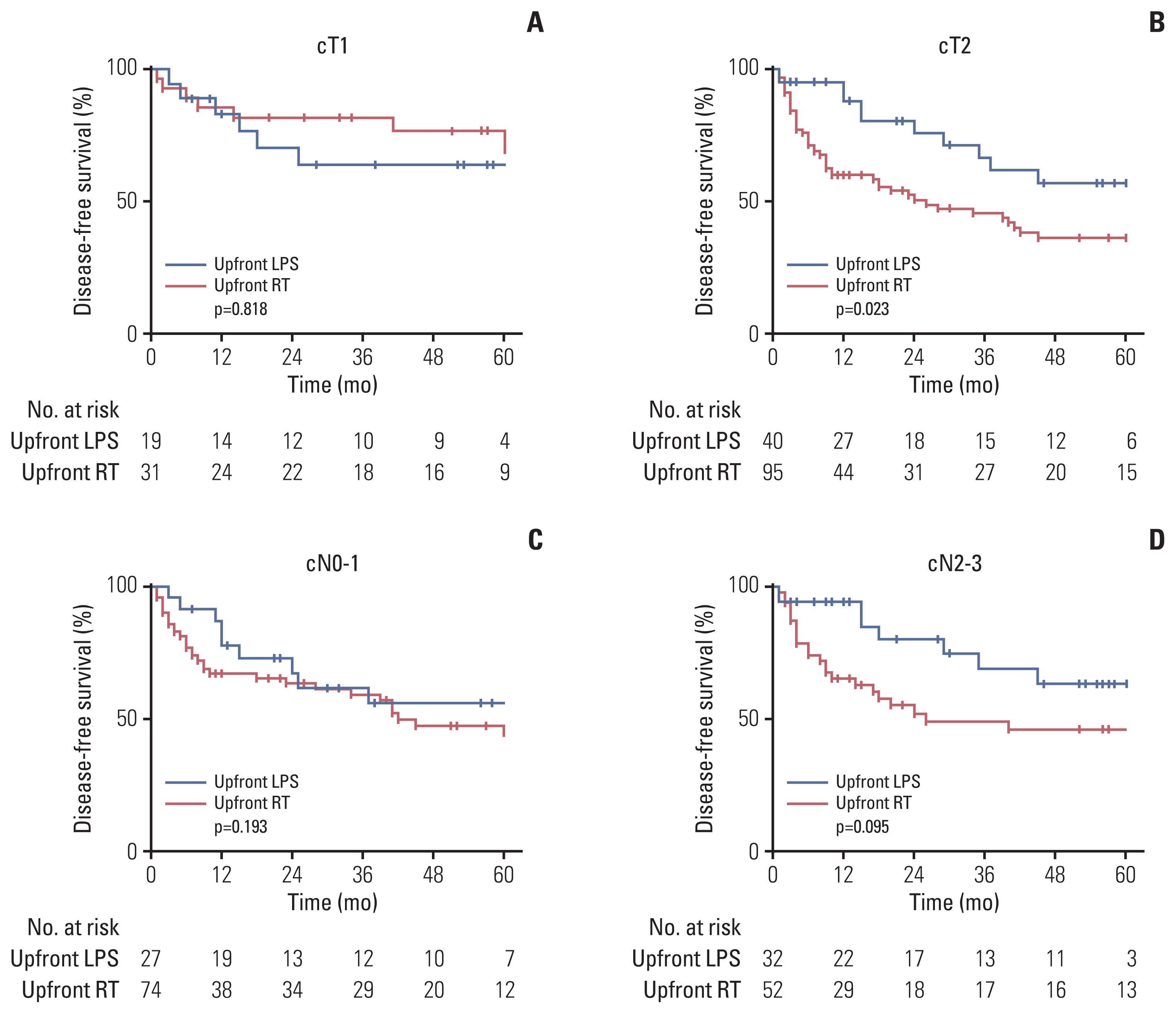
Further analyses based on subgroup stratified by cT and cN status were performed (Table 3). The rates of loco-regional recurrence and DFS among cT1 subgroup were similar between the groups. In contrast, among cT2 subgroup, loco-regional recurrence was more frequently observed in the upfront RT group (12.5% in the upfront LPS group vs. 31.6% in the upfront RT group, p=0.030), and the 5-year DFS rate was better following upfront LPS (57.0% vs. 36.4%, p=0.023) (Fig. 3A and B). Although neck dissection was more frequently performed in the cN2–3 patients than cN0–1 patients (100.0% vs. 63.0%), the crude incidence rates of regional recurrence were similar between groups: 14.8% vs. 5.4% in cN0–1 subgroup (p=0.205); and 6.3% vs. 19.2% in cN2–3 subgroup (p=0.120), respectively. In addition, comparable rates of OS and DFS were achieved in cN subgroups following both approaches (Fig. 3C and D).
5. Prognostic factors affecting the OS and DFS rates
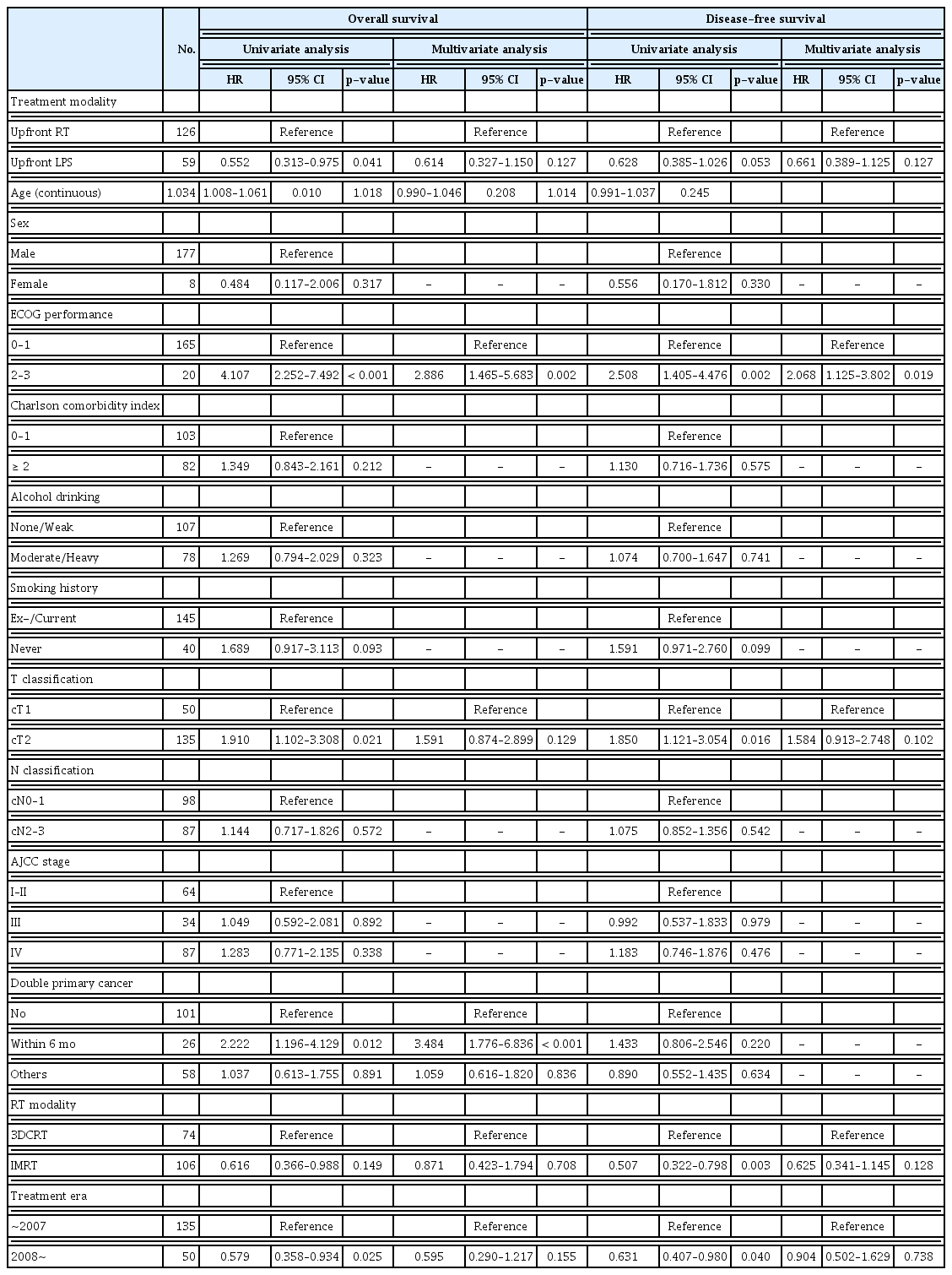
Univariate analyses revealed that upfront LPS showed benefit over upfront RT in OS (p=0.041) and DFS (p=0.053), which, however, were not proven in multivariate analysis (HR, 0.61; 95% CI, 0.33 to 1.15; p=0.127 and HR, 0.66; 95% CI, 0.39 to 1.13; p=0.127), respectively (Table 4). In addition, treatment modality had little impact on HPC-specific survival in univariate analysis (p=0.122) (S3 Table). In multivariate analyses, poor performance status of ECOG 2–3 (HR, 2.89; 95% CI, 1.47 to 5.68; p=0.002) and diagnosis of subsequent malignancy within 6 months (HR, 3.48; 95% CI, 1.78 to 6.84; p < 0.001) were significantly factors to worse OS. Furthermore, poor performance status of ECOG 2–3 remained as significantly adverse factors in DFS (HR, 2.07; 95% CI, 1.13 to 3.80; p=0.019) (Table 4) and HPC-specific survival (HR, 2.53; 95% CI, 1.12 to 5.73; p=0.026) (S3 Table).
Discussion
In this study, we investigated the clinical outcomes following larynx-preserving approach, either upfront LPS or RT, in patients with cT1–2 HPC. Within the same cT status, the actual tumor burden could be different based on the nature of tumor growth, and well-defined tumors are easily assumed to have smaller tumor burden than diffuse and/or infiltrative ones. Even though we thoroughly re-evaluated all patients’ information with regard to cT status, there might have been a preference toward upfront LPS over upfront RT for patients with favorable performance status and localized primary tumor. Consequently, favorable outcomes following LPS could have reflected aforementioned selection bias that happened in real-world clinical practice. Subsequent subgroup analyses based on cT status also revealed that DFS advantages following upfront LPS were apparent only in cT2, but not in cT1 subsets. However, there was no significant difference between treatment groups for survival outcomes after adjusting baseline clinical characteristics.
The basic assumption of larynx preserving partial hypo-pharyngectomy seems contrary to the infiltrative and extensive mucosal/submucosal spreading nature of HPC [21]. It also differs from the surgical concept of endolaryngeal surgery for laryngeal cancer, which has minimal nearby tissue invasion and justifies a narrow surgical safety margin [22]. Recently, LPS via trans-oral techniques has been introduced for the management of selected HPC cases (usually T1–2, but also selected T3) [23–26]. However, there has not been enough evidence to determine whether LPS itself (or LPS alone) is adequate for locally aggressive HPC or whether it is really beneficial to change the RT dose from the definitive (usually 70 Gy) to the adjuvant setting (around 60 Gy). In oropharyngeal cancer, an RT dose reduction of 10–15 Gy had only a small effect on patients in terms of RT toxicity [27].
Therefore, the current study addresses an important clinical issue in the management of HPC. Our first question was whether LPS is oncologically safe. Based on the currently accepted guideline, LPS seems to be suboptimal (clear resection in 37.3% of cases in our series), and frequently requires adjuvant treatments (91.5%). In cT2 tumors, LPS with adjuvant treatment seemed to offer better disease control than the upfront RT, perhaps because the addition of surgery to RT/CCRT led to a rapid decrease in the tumor burden. However, the surgical wound itself can have a negative impact on the effectiveness of RT for tumor control [28], because most wounds after LPS in our series healed secondarily without reconstruction (78.0%). Furthermore, LPS can distort the anatomy and deteriorate laryngeal function further even with the preservation of functional speech and swallowing, as indirectly revealed by the occurrence of treatment-related complications in the upfront LPS group. Thus, the risks and benefits of LPS should be weighed carefully.
The next clinical question was the effectiveness of each treatment for the high nodal disease which is commonly seen in HPCs. Since the neck node metastases could also affect the therapeutic decision, upfront neck dissection was frequently performed in patients with bulky neck tumor (cN2–3). However, there was no apparent difference in oncologic outcomes based on subgroup analyses regarding cN status (Tables 3 and 4). Thus, even with bulky or multiple neck diseases, non-surgical treatments achieved regional control comparable to an initial neck dissection in HPC.
In real clinical practice, both performance status and clinical tumor extent are recognized as determinants of the treatment modality. In the current study, the survival outcomes did not differ significantly according to the treatment modality, with multivariable analyses. Therefore, we could postulate that the treatment modality selection based on both patient and tumor factors is appropriate in real-world practice. The treatment and prognosis for multiple primary cancers in HPC patients could also affect therapeutic decision. The oncological outcomes of patients with other primary malignancies following upfront LPS were superior to those following upfront RT (data not shown) suggesting that multiple primary cancer patients with favorable prognostic factors could have been more frequently included in the upfront LPS group.
Another factor for treatment decision is functional preservation of voice and swallowing. It is easily agreeable that the voice quality tends to be better following non-surgical therapy than following LPS. Although we did not investigate the quality of voice in detail, the ultimate rate of preserving functional larynx in the upfront RT group was inferior to that of upfront LPS group (Table 2). This could have been related with salvage treatment modality for local recurrence, which can be largely dependent on the initial modality. The patients developing local recurrence following upfront LPS could have another chance of larynx preservation by applying salvage RT. However, those following upfront RT are generally recommended surgical salvage, which frequently includes total laryngopharyngectomy. In addition, an unnoticed selection bias could have existed in real-world practice setting: the patients with advanced primary tumor could have been allocated to upfront RT and the necessity of total laryngopharyngectomy for local recurrence following upfront RT could have increased.
However, our findings are limited by the small number of patients, unnoticed selection bias, the retrospective nature of our study, and the experiences, resources, and preferences of our individual clinic. Although moderate to high alcohol consumption and presence of comorbidity were well-known unfavorable factors in the HPC patients, there was no significant correlation between outcomes and these factors in the current study [29,30]. As our study is in retrospective nature, there might have been ‘recall bias’ by the patients regarding quantitative alcohol drinking history, which could have caused difficult subgroup analyses on the degree of alcohol assumption (i.e. none, weak, moderate, and heavy drinker). Likewise, information on comorbidity could have been inaccurate and insufficient in our study.
Similar to technical advances in RT, LPS procedures are still evolving from the use of conventional magnification (microscope or endoscope) to robotics and image-guided surgery (we did not use robotics for the trans-oral approach in our series) [25,26]. The surgical precision offered by these modern surgical tools could achieve better outcomes with less morbidity than current findings, which could lead to a different conclusion from our study.
We could suggest that upfront LPS with adjuvant treatment as an effective treatment option for disease control and functional preservation in selected patients. However, upfront LPS may need to be limitedly applied in consideration of tumor factor, patient performance status, and the presence of multiple primary cancers, as this approach frequently required additional adjuvant treatment. Thus, the selection of treatments for resectable HPC should be individualized based on both patient and tumor factors. This preliminary finding should be validated in a further prospective trial with large series.
Electronic Supplementary Material
Supplementary materials are available at Cancer Research and Treatment website (https://www.e-crt.org).
Notes
Ethical Statement
The current study was approved by Institutional Review Board, and the written informed consent was waived owing to the retrospective nature. The clinical data used in the study were de-identified.
Author Contributions
Conceived and designed the analysis: Jeong HS, Ahn YC.
Collected the data: Kim D, Kim N, Koh S, Chung MK, Son YI.
Contributed data or analysis tools: Chung MK, Son YI, Oh D, Jeong HS, Ahn YC.
Performed the analysis: Kim D, Kim N, Oh D, Jeong HS, Ahn YC.
Wrote the paper: Kim D, Kim N, Jeong HS, Ahn YC.
Conflicts of Interest
Yong Chan Ahn, the editor-in-chief of the Cancer Research and Treatment, was not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Acknowledgements
This work was supported by a grant from the National Research Foundation of Korea (NRF) funded by the Korean government (MEST) (no. 2018R1A2B6002920).
The funders had no further role in the study design; collection, analysis and interpretation of data; writing of the manuscript; or decision to submit this manuscript for publication.