Development of a Tongue Immobilization Device Using a 3D Printer for Intensity Modulated Radiation Therapy of Nasopharyngeal Cancer Patients
Article information
Abstract
Purpose
This study aimed to reduce radiation doses to the tongue, a patient-specific semi-customized tongue immobilization device (SCTID) was developed using a 3D printer for helical tomotherapy (HT) of nasopharyngeal cancer (NPCa). Dosimetric characteristics and setup stability of the SCTID were compared with those of a standard mouthpiece (SMP).
Materials and Methods
For displacement and robust immobilization of the tongue, the SCTID consists of four parts: upper and lower tooth stoppers, tongue guider, tongue-tip position guide bar, and connectors. With the SCTID and SMP, two sets of planning computed tomography and HT plans were obtained for 10 NPCa patients. Dosimetric and geometric characteristics were compared. Position reproducibility of the tongue with SCTID was evaluated by comparing with planned dose and adaptive accumulated dose of the tongue and base of the tongue based on daily setup mega-voltage computed tomography.
Results
Using the SCTID, the tongue was effectively displaced from the planning target volume compared to the SMP. The median mucosa of the tongue (M-tongue) dose was significantly reduced (20.7 Gy vs. 27.8 Gy). The volumes of the M-tongue receiving a dose of 15 Gy, 30 Gy, and 45 Gy and the volumes of the mucosa of oral cavity and oropharynx (M-OC/OP) receiving a dose of 45 Gy and 60 Gy were significantly lower than using the SMP. No significant differences was observed between the planned dose and the accumulated adaptive dose in any dosimetric characteristics of the tongue and base of tongue.
Conclusion
SCTID can not only reduce the dose to the M-tongue and M-OC/OP dramatically, when compared to SMP, but also provide excellent reproducibility and easy visual verification.
Introduction
Radiation therapy (RT), usually combined with surgery and/or chemotherapy, plays an important and pivotal role in treating the patients with head and neck cancer (HNC) [1–3]. Delivery of curative high radiation dose is typically accompanied with the acute and delayed radiation toxicities, mainly because of the anatomical proximity of the radiation target to the surrounding normal organs at risk (OARs). For example, the radiation targets in treating the patients with nasopharynx cancer (NPCa) are usually located very close to several OARs, which include the temporal brain, brainstem, spinal cord, optic apparatus, major salivary glands, pharyngeal musculature, and mucosa of oral cavity (OC) and oropharynx (OP) (M-OC/OP).
The tongue plays very important roles of tasting, speech, mixing and swallowing food [4–7], and anatomically occupies the OC and the OP (base of tongue). Acute damage of the tongue by high dose RT is typically presented as confluent mucositis, usually associated moderate to severe pain and altered taste. Though acute inflammatory damages could usually recover in a few weeks of finishing RT, delayed damages, represented as fibrosis, however, can cause altered taste, mucosal dryness, and difficulty in food mixing, swallowing, and speech persistently. In order to maintain a descent quality of life level, it is very important to minimize the radiation damage of the tongue and consequently to decrease the severity of acute and delayed reactions.
Intensity modulated radiation therapy technique has contributed to significant reduction of the OARs’ damage risk [1,2,4,7,8]. Various additional approaches including the patient guidance [9] and the use of intra-oral devices (IOD) [10–16] have been proposed to avoid or lessen severe acute mucosal toxicity, all of which have some limitations in routine clinical application. We previously designed and developed a unique semi-customized tongue displacement device, using the 3D printing (3DP) technology, and reported the dosimetric profiles by actual application to seven patients (four with OC cancer and three with tonsil cancer) who underwent ipsilateral RT [17]. Using this device, the median tongue dose could be significantly reduced (< 30 Gy), with the capability of patient-specific customization. Based on our previous experience, we developed a unique semi-customized tongue immobilization device (SCTID) mainly to reduce radiation dose to the anterior part of the OC/OP in treating the NPCa patients needing bilateral RT. This is to report the manufacturing process, setup reproducibility, and dosimetric characteristics, when compared with using the standard mouthpiece (SMP).
Materials and Methods
1. Patient selection
With the approval from the institutional review board (IRB SMC 2020-02-134-001), 10 consecutive NPCa patients, who underwent definitive RT between October 2017 and January 2018 at the authors’ department with helical tomotherapy (HT; TomoHD, Accuray, Sunnyvale, CA), were included in this study (Table 1).
2. Design of SCTID
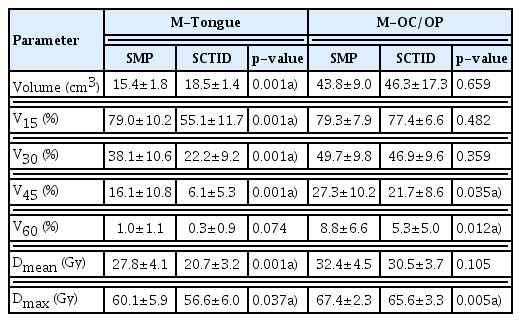
We designed the SCTID with the aim to reduce the radiation dose to the mucosal lining of the OC/OP in treating the NPCa patients. The manufacturing process is quite similar to that previously reported [17]. The SCTID consists of four parts: upper and lower tooth stoppers; tongue guider; tongue-tip position guide bar; and connectors, respectively (Fig. 1). Typically, a patient is asked to bite the tooth stoppers with the incisors, and to stick out his (or her) oral tongue beneath and along the tongue guider through the open window between the tooth stoppers until the tongue-tip touches the position guide bar, and to keep this position during each RT session (red dotted arrows in Fig. 1). The tongue guider, connected to the tongue-tip position guide bar, was designed to provide mild downward compression and immobilization of the tongue and to provide a simple visual verification of the tongue-tip position by the naked eyes during each RT session. The connectors were designed to attach the SCTID to the thermoplastic immobilization mask securely and easily. In order to accommodate each patient’s physical dimension of the tongue and the jaw opening and for patient-specific semi-customization, we fabricated several different sizes of the SCTID with varying combinations respects to the height (10–20 mm), width (30–45 mm), and length (50–65 mm), with 5 mm’s increment in each variable, respectively.
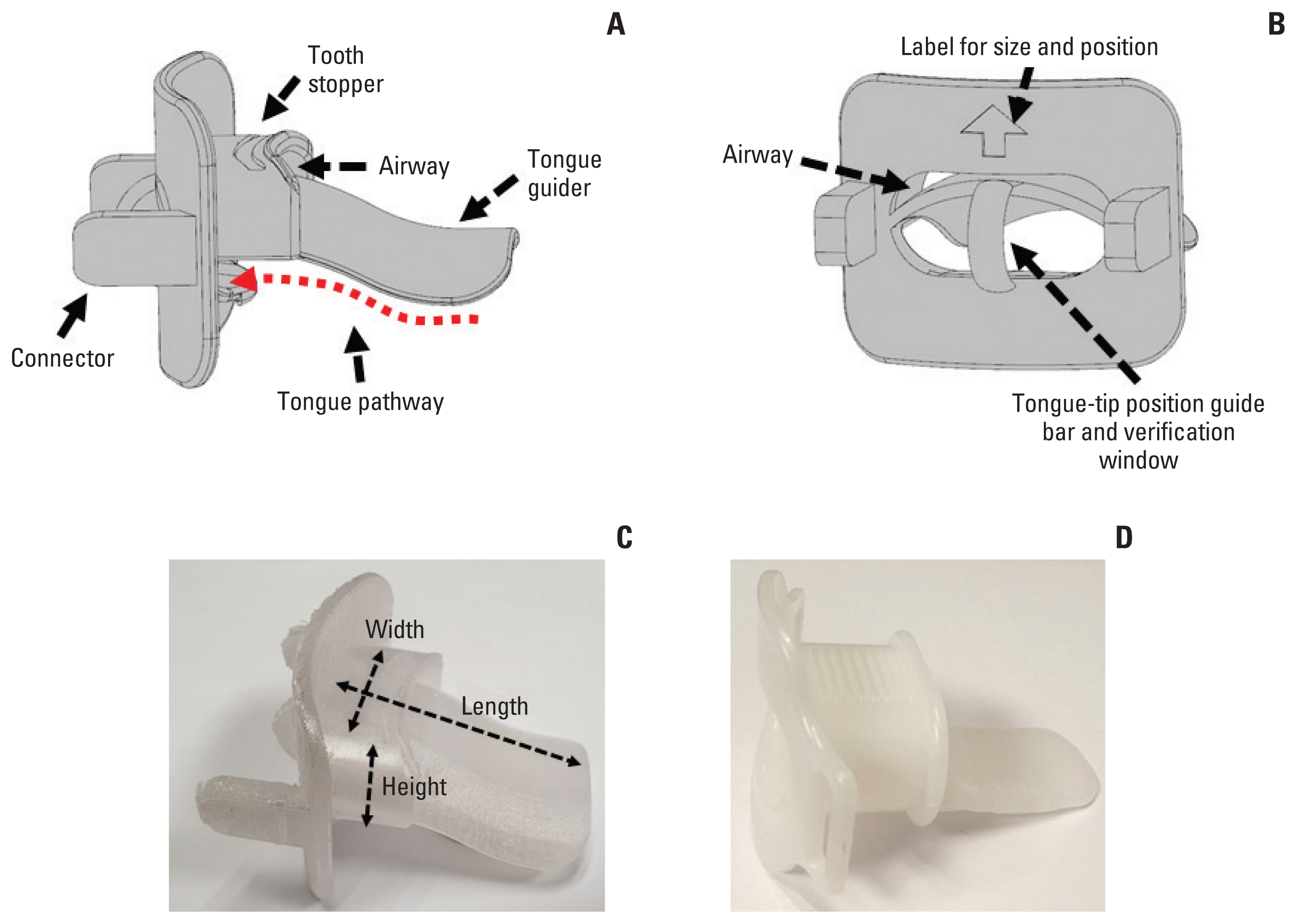
Side (A) and front (B) views of the 3D model for the semi-customized tongue immobilization device. It was printed using a three-dimensional printer with a biocompatible material (C). Commercially available standard mouthpiece (D), which has been the most commonly used device in head and neck cancer radiation therapy.
3. Simulation
Each patient underwent computed tomography (CT)–based simulation in the supine position two times: one with the SCTID, and the other with the SMP, respectively. Before the actual simulation, each patient’s distances between the upper and lower incisors (height) and between the mouth angles on both sides (width), and the length of the OC were measured, so that we could select a presumably proper size of the SCTID. For the adaptation and training, each patient was asked to keep sticking out the tongue using the pre-selected SCTID, under our guidance, for about 10 minutes before determining the patient-specific proper size. Once the patient was familiarized with the SCTID, the actual CT-based simulation process was undertaken and the SCTID was connected to the individually customized thermoplastic mask (Aquaplast RT, Q-fix, Avondale, PA). Among 10 patients, nine used the SCTID size with 25 mm height×45 mm width×55 mm length, while one did with 15 mm height×45 mm width×55 mm length, respectively. In each patient, another simulation process was undertaken under the same conditions, but with the SMP, instead of the SCTID. The SMP was designed mainly to immobilize the tongue by downward depression, and, however, visual verification of the tongue position was not possible (Fig. 1D).
4. Delineation of target volumes and OARs
On two sets of simulation CT images, the gross tumor volume (GTV), clinical target volume (CTV) were delineated based on all available clinical information including the diagnostic images. The OARs, including the spinal cord, brainstem, parotid gland, were delineated according to the previous guidelines [18,19]. The mucosa of tongue (M-tongue) was to include the mucosal surface of the entire tongue with 2 mm thickness from the tongue-tip anteriorly to the vallecula posteriorly. The M-OC/OP was to include the mucosal surface, with 2 mm thickness of the following structures: the buccal cheek, labial mucosa, outer and inner surfaces of gingiva, retromolar trigone, floor of mouth, hard and soft palate (including uvula), tonsillar fossa, and lateral walls of OP. The posterior wall of the OP was not delineated, considering that the damage of this region seemed less critically associated with painful symptomatic mucositis. The mucosa of the gingival mucosa and retromolar trigone region were contoured to the root levels of upper and lower teeth. The mucosa of the outer gingiva and buccal cheek were delineated together, since these were virtually in contact with each other. In order to guarantee consistency, all the mucosal contouring was done by one radiation oncologist. The planning target volumes (PTVs) were generated with the 3 mm’s isotropic expansion from the GTV (P-GTV) and CTV (P-CTV), respectively, which were edited in consideration of the actual anatomic boundaries, such as the spinal cord and skin surface.
5. Treatment planning
HT plans (Tomoplan, Accuray) were generated on two sets of CT images: one with the SCTID and the other with the SMP. The same dose schedule and constraints were applied to both plans along the internal guidelines. The typical dose schedule was to deliver 67.2 Gy to the P-GTV and 56.0 Gy to the P-CTV in 28 fractions using the simultaneously integrated boost and adaptive re-plan for the later 12 fractions, respectively. In accordance with the internal planning guideline, two constraints were set at the highest priority level for the PTVs and planning spinal cord (P-cord) (Table 2). The constraint set at the second priority level was to limit the mean dose (Dmean) to the parotid under 26 Gy. The constraints set at the third priority level were related to the M-tongue and M-OC/OP: the Dmean < 30 Gy; and the volume receiving 30 Gy or higher (V30 Gy) < 30%, respectively. The lowest level constraint was to limit the Dmax to the brainstem not to exceed 54 Gy.
For equal dosimetric comparison between the SCTID and SMP, the same dose optimization strategy was applied based on the same plan conditions, including the number of iterations, dose constraints, dose calculation algorithm (collapsed-cone convolution) [20,21], and beam parameters (field width of 2.5 cm, modulation factor of 2.0, and pitch of 0.287) [22], respectively. Once the PTV dose met the goal, the optimization was continued to reduce the doses to the OARs as low as possible until the iteration limit, while maintaining the PTV dose.
6. Evaluation of geometric and dosimetric effects of SCTID versus SMP
The geometrical effect was compared by measuring the volumes of the M-tongue and M-OC/OP in both the SCTID and SMP plans. Several PTV-related dosimetric parameters were chosen for comparative evaluation on the quality of two plans: the minimum dose received by 98% (D98); 50% (D50); and 2% (D2) of the PTVs, respectively. The homogeneity index (HI) [17] and conformity index (CI) [23] were also compared. In addition, several OAR-related dosimetric parameters were measured for comparison: Dmean of the parotid; Dmax of the P-cord and brainstem; and the percentages of M-tongue and M-OC/OP volume that received a dose of 15 Gy (V15), 30 Gy (V30), 45 Gy (V45), 60 Gy (V60), or more; and Dmax and Dmean of the M-tongue and M-OC/OP, respectively. Statistical analysis of the dosimetric comparison between the SMP and SCTID plan was done using a paired t test. A probability level with a p-value < 0.05 was considered significant.
7. Setup reproducibility of the tongue with SCTID
In order to evaluate the setup reproducibility of the tongue during the definitive RT course, we used the daily setup mega-voltage computed tomography (MVCT) images of seven NPCa patients, who underwent HT using the SCTID from August to September 2019. On each patient, 16 sets of MVCT images were retrieved and registered to the planning CT images by deformable image registration algorithm using an adaptive plan function (PrecisionART, Accuray) [24]. We calculated accumulated adaptive doses and obtained accumulated-adaptive-DVHs (AA-DVH) for the tongue and base of the tongue and compared these with the planned DVHs (P-DVH). To evaluate setup reproducibility of the tongue with SCTID, we compared the panned doses with the accumulated adaptive doses based on mean DVHs (P-DVH vs. AA-DVH), V15, V30, V45, V60, and Dmean of the tongue and tongue base. The same statistical analysis described in the previous session was done between the two groups.
Results
1. Geometrical effect of SCTID
In most patients, use of the SMP resulted in posterior displacement of the tongue apparently, so that the posterior tongue and the nasopharynx target became closer to each other (red arrows in Fig. 2). In contrast, the SCTID allowed the tongue to depress and move anteriorly along the tongue guider, and the distance between the posterior tongue and the nasopharynx target became larger, when compared with the SMP use (green arrows in Fig. 2). Additionally, the mean M-tongue volume was larger when using the SCTID, mainly because of the tongue protrusion and elongation, compared with the SMP use (18.5±1.4 cm3 vs. 15.4±1.8 cm3, p=0.001) (Table 3). Meanwhile there was no significant difference in the M-OC/OP volume (46.3±17.3 cm3 vs. 43.8±9.0 cm3, p=0.659).
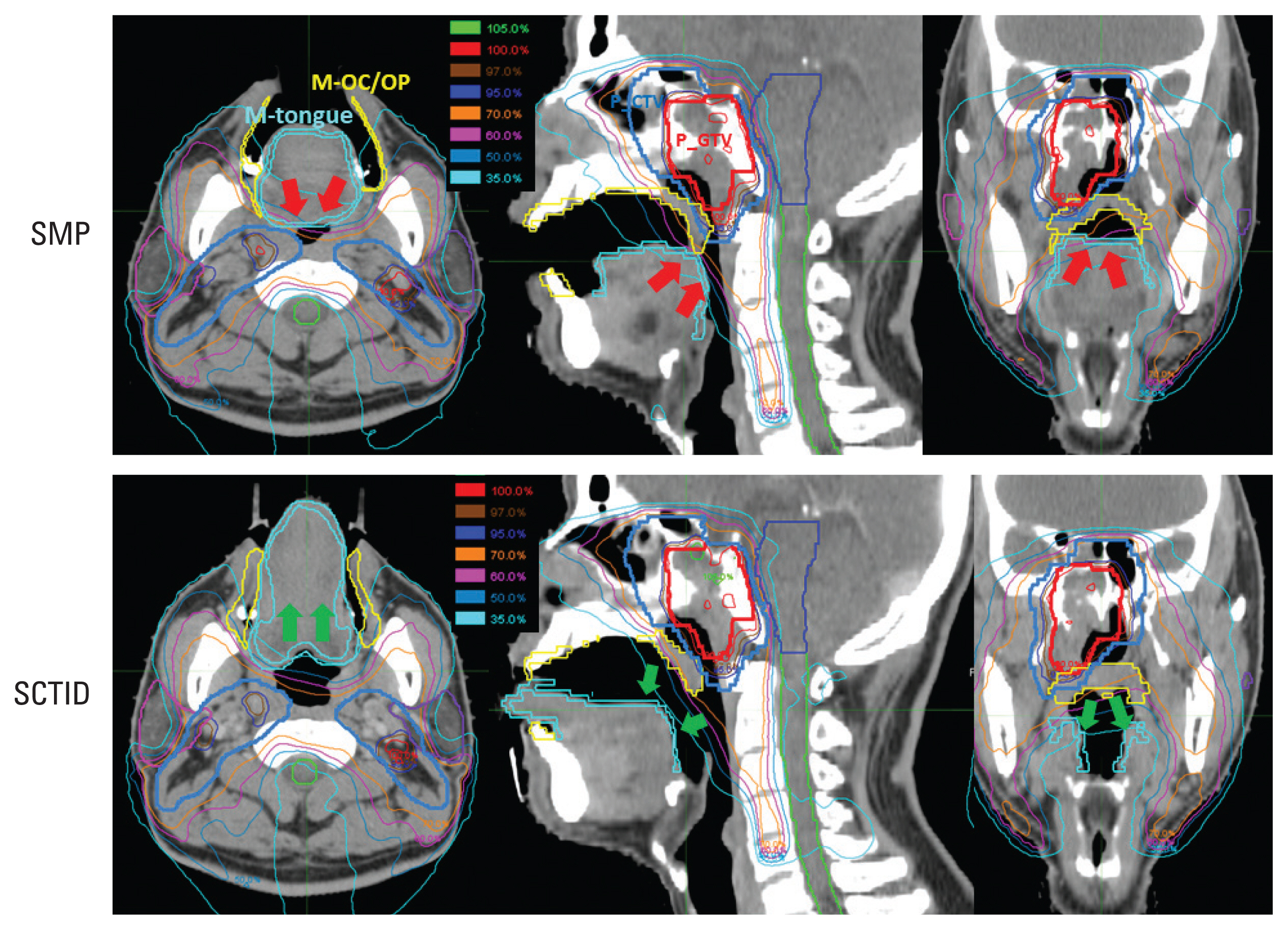
Isodose distributions for the standard mouthpiece (SMP, upper) and semi-customized tongue immobilization device (SCTID plans, lower). The tongue was effectively stuck out along the tongue guider by using the SCTID, it led to displacement of the mucosa of the tongue (M-tongue) from target (green arrows) while a partial volume of the M-tongue received a high dose equivalent to the prescribed dose (red arrows) in the SMP plan because depression of tongue caused the tongue to push back toward posterior neck region.
2. Dosimetric comparison
In connection with geometrical change of the tongue, the M-tongue was effectively displaced away from the high dose regions and smaller volume of the M-tongue received high dose by using an SCTID, when compared with the SMP use (Fig. 2). While the mean DVH profiles of the targets were almost the same between two groups, those of the M-tongue and M-OC/OP dramatically decreased when using the SCTID, compared with SMP (Fig. 3).
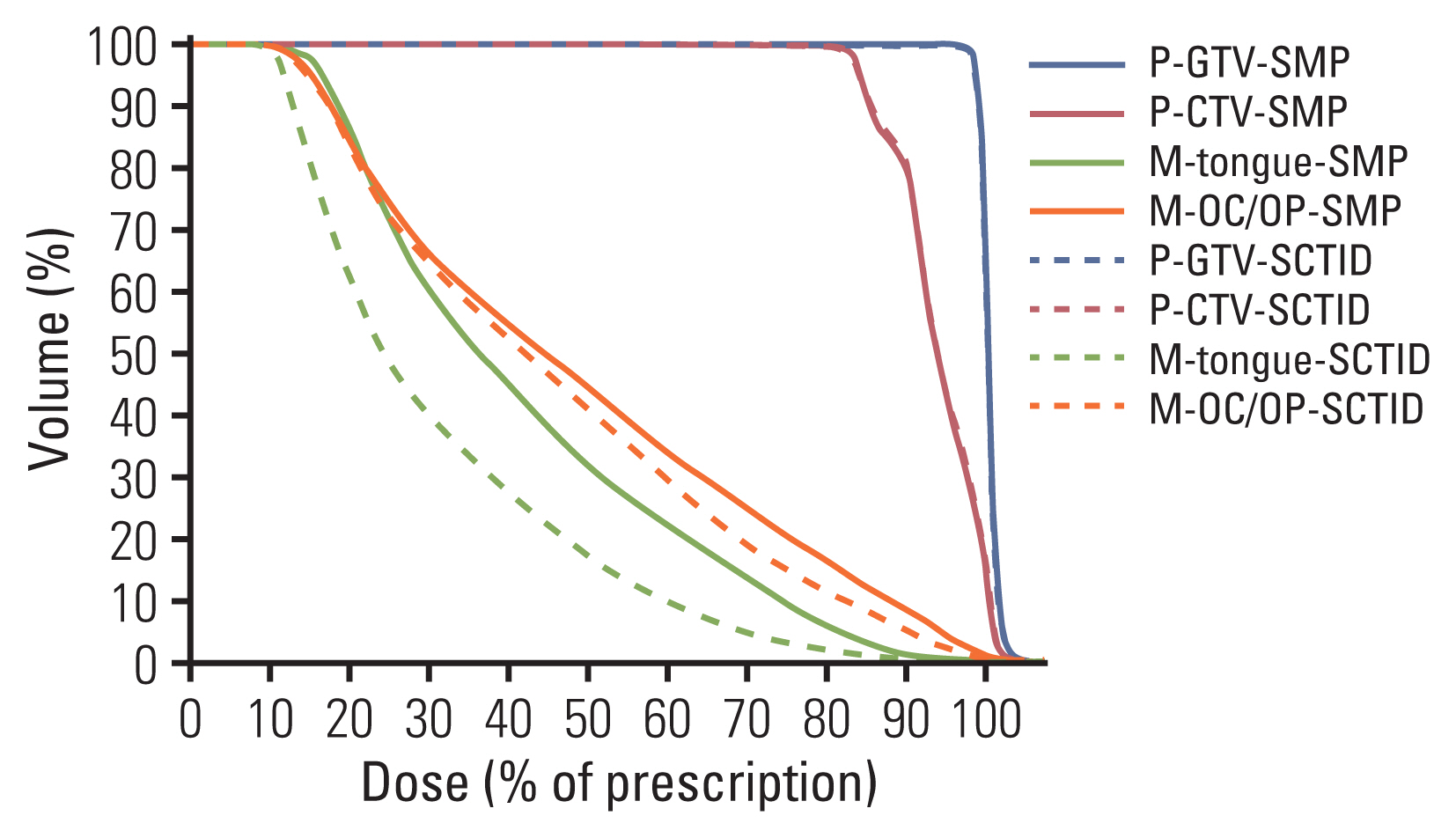
Mean DVHs for mucosa of the tongue (M-tongue, green) and oral cavity and oropharynx (M-OC/OP, orange) dramatically decreased using a semi-customized tongue immobilization device (SCTID, dotted line) compared with standard mouthpiece (SMP, solid line), but mean DVHs for the targets showed almost same shape between two groups. M-OC/OP, mucosa of the oral cavity and oropharynx; M-tongue, mucosa of the tongue; P-cord, planning spinal cord; P-CTV, planning clinical target volume; P-GTV, planning gross target volume.
For target dose coverage, no significant differences between the SCTID and SMP uses were observed in D98%, D50%, D2%, HI, and CI for all PTVs, except D98% and HI for P-GTV (Table 4). By using the SCTID, however, D98% of the P-GTV decreased (66.4 Gy vs. 66.2 Gy, p=0.005) and HI of the P-GTV increased (0.035 vs. 0.039, p=0.011), respectively (Table 4). In addition, no significant dose difference was observed in Dmean of the parotid (30.8 Gy vs. 30.7 Gy, p=0.779) or Dmax of the P-cord (30.6 Gy vs. 30.5 Gy, p=0.973), and the brainstem (40.8 Gy vs. 40.2 Gy, p=0.624), respectively.

Comparison of dosimetric characteristics between the SMP and the SCTID for planning targets and OARs
The mean Dmean and Dmax of the M-tongue were significantly reduced by 25.5% and 5.8% in the SCTID (20.7 Gy and 56.6 Gy) compared with the SMP (27.8 Gy and 60.1 Gy), respectively (p < 0.05) (Table 3). Moreover, mean V15, V30, and V45, of the M-tongue were significantly lower in the SCTID (55.1%, 22.2%, and 6.1%, respectively) than the SMP (79.0%, 38.1%, and 16.1%, respectively) (all p < 0.05). However, no significant difference in V60 of the M-tongue was observed between the SCTID (0.3%) and the SMP (1.0%) (p=0.074).
In the M-OC/OP, volumes that received high dose (mean V45 and V60) and mean Dmax were significantly lower in the SCTID (21.7%, 5.3%, and 65.6 Gy, respectively) than the SMP (27.3%, 8.8%, and 67.4 Gy, respectively) (p < 0.05). However, although the mean values of the V15 and V30 that received low dose and Dmean were lower in SCTID (77.4%, 46.9%, and 30.5 Gy, respectively) than SMP (79.3%, 49.7%, and 32.4 Gy, respectively), but no significant difference were observed in statistical analysis (p > 0.05).
3. Setup reproducibility of the tongue with SCTID
Total 112 setup MVCT image sets of seven NPCa patients with SCTID were compared with their plan CT image to confirm setup reproducibility of the tongue with the SCTID. In most patients, the shape and position of the tongue (red in Fig. 4A) matched well with the original plan CT image (gray in Fig. 4A). These are well represented in mean AA-DVH of the tongue and tongue base (Fig. 4B). The mean AA-DVH of the tongue was almost in line with the P-DVH of the tongue, while the mean AA-DVH of the tongue base was slightly higher than the mean P-DVH of the tongue base in 30%–70% of the prescription dose regions. No significant differences, however, was observed between the planned dose and the accumulated adaptive dose in any dosimetric characteristics of the tongue and tongue base (p > 0.05) (Table 5).
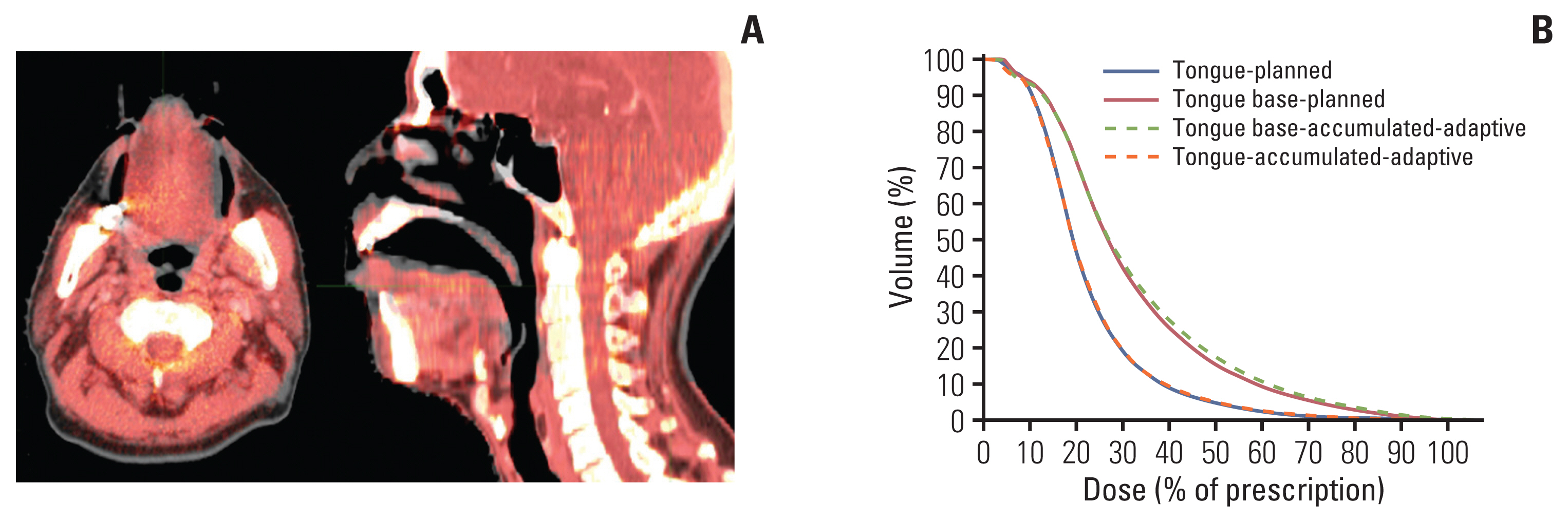
In most patient, shape and position of the tongue in daily setup mega-voltage computed tomography (MVCT) image (red) matched well with original one in plan computed tomography image (grey) (A). (B) The mean accumulated-adaptive-DVH (AA-DVH) for tongue (orange dotted line) was almost in line with the mean planned DVH (P-DVH) for tongue (blue solid line), while the mean AA-DVH for tongue base (green dotted line) was slightly higher than the mean P-DVH for tongue base (red solid line), but no significant differences was observed between two groups (p > 0.05).
Discussion
It is well known that the acute and delayed radiation damage of the oral tongue can cause significant deterioration of the patients’ quality of life. There were several reports regarding the dosimetric co-relations with the morbidity risk, which generally increased along the increasing dose and volume of the OC structures including the tongue. For example, high mean dose of the tongue was related to the tongue movement dysfunction, altered taste, speech quality, and even weight loss [5,6]. And a positive dose-response relationship was reported regarding the relative taste loss with the threshold at around 20 Gy [25,26]. By using the SCTID, which we developed, we could lower the mean M-tongue dose close to this threshold, when compared to the SMP use (20.7 Gy vs. 27.8 Gy, p=0.001), and the clear dose reduction effects were achieved in all dosimetric indicators except V60. In addition, it was reported that V30 < 65% and V35 < 35% for the anterior OC were the predictive factors for swallowing dysfunctions [27], while the mean dose to the OC (representing the minor salivary gland dose) was a significant factor of dry mouth [7]. In the current study, by using the SCTID, we could follow these dose limits without sacrificing the plan quality or patient setup stability, when compared to the SMP use.
In case of ipsilateral RT, such as in treating the OC cancer patients, displacement of the tongue to the opposite side of the target was quite effective in decreasing the tongue dose [17]. However, lateral displacement of the tongue seemed less effective when applying bilateral neck HNC RT. We intended to develop a unique IOD, to be used in bilateral HNC RT delivery, with the capability of normal structure sparing from moderate to high dose radiation, robust setup reproducibility, easy visual verification, and patient-specific customization. First, we focused on the ‘stick-out’ tongue position [9] and adapted to use the tongue guider and the tongue position guide bar with verification window. As a result, we could achieve improved dose profile around the normal structures with easy verification of reproducible immobilization.
When using the IOD, reproducible and reliable immobilization of the tongue is very important [13,28]. It is ideal to compare the setup reproducibility between SMP use and SCTID use on the same patients, which, however, was practically impossible. It would have been better to select the same patients included in this dosimetric analysis for the setup reproducibility of the tongue; however, we had to use the data on seven different NPCa patients who used the SCTID, because of a technical problem that could not bring up the existing setup MVCT due to the system upgrade. It should be considered that the dosimetric comparison between different IODs is independent issue from the setup reproducibility. Though with the above mentioned limitations, SCTID as a unique IOD proved to have provided excellent reproducibility. In our experience, to achieve these goals, it is necessary to cooperate with the patient who is asked to stick out the tongue properly and with the therapists who need to confirm the tongue position. The tongue position guide bar and verification window were very useful to achieve these two goals. However, SMP could not provide these, and the tongue was pushed posteriorly to reach the soft palate, close to the target region, which not infrequently caused high dose radiation damage, mainly because of absence of verification window and poor cooperation by the patient.
Patient-specific customization of the IOD is another important and desirable issue in order to cover various physical dimensions of each patient’s OC and jaw opening, which, however, is not easily achieved when using the conventional IODs. In-house manufacturing of the individualized IODs either by consulting dentistry department or by skilled RT staff seems ideal [29], which, however, is usually difficult and time-consuming. In this point, our semi-customized manufacturing process, with the fixed basic model and minor modification per each patient, seems the realistic alternative. Furthermore, 3DP technology has made it possible to manufacture the proper SCTID among 27 different sizes within a short time when needed at low cost. In addition, 3DP is suitable for manufacturing complex structure (narrow entrance and wide inside such as the tongue guider and verification window), which is very difficult to produce by the conventional molding process. From this experience, we have developed a unique manufacturing process that could allow printing the patient-specific RT device for routine clinical use.
The IOD is placed with a patient’s mouth open. As the course of RT progresses, oral discomfort or pain may increase because of oral mucositis and trismus caused by RT. In order to minimize discomfort, Hong et al. [17] recommended several conditions for the IOD to be equipped. Although SCTID fulfilled most of the conditions, we were concerned gag reflex by tongue guider. However, all patient included in this study completed RT with SCTID without any complaint. Based on our experience, only less than 1% of total NPCa patients used SMP instead of SCTID because of oral discomfort.
We have developed a unique SCTID to reduce M-tongue and M-OC/OP dose with robust setup stability based on semi-customization by using 3DP technology. SCTID can not only reduce the dose to the M-tongue and M-OC/OP dramatically, when compared to SMP, but also provide excellent reproducibility and easy visual verification.
Notes
Ethical Statement
The institutional review board at Samsung Medical Center (file no. SMC 2020-02-134-001, April 2, 2020). Informed consent was waived.
Author Contributions
Conceived and designed the analysis: Ju SG, Ahn YC.
Collected the data: Kim YB, Park SG, Na CH, Choi YM, Hong CS, Oh D, Kwon DY, Kim CC, Kim DH.
Performed the analysis: Kim YB, Park SG, Na CH, Choi YM, Hong CS, Oh D, Kwon DY, Kim CC, Kim DH.
Wrote the paper: Ju SG, Ahn YC.
Conflicts of Interest
Yong Chan Ahn, a contributing editor of the Cancer Research and Treatment, was not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Acknowledgements
This work was supported by the research program, NRF-2018R1D1A1B07043445 (Republic of Korea). This idea is protected by a Korean patent (No. 10-1983786).