INTRODUCTION
Gastric carcinoma remains a major cause of cancer death worldwide. While its overall incidence appears to be decreasing there has been a dramatic rise in the incidence of upper third gastric cancer in the west (1). These changes in gastric cancer subsite have focused attention on the management of upper third gastric cancer and, in particular, the optimal extent of resection (total gastrectomy or proximal gastrectomy), and in the extent of lymph node dissection.
The traditional approach to surgery with curative intent has been total gastrectomy with extended lymph node dissection (2). Recently, reflecting the trend toward function-preserving or limited surgery, proximal gastrectomy is accepted by many Japanese surgeons as the most preferred method for the upper third gastric cancer, and this is particularly true for cases of early gastric cancer (3,4). However, there is still no general agreement on the choice of surgical method for the upper third gastric cancer. Whether total gastrectomy or proximal gastrectomy is the most effective surgical procedure for cure is controversial because of the lack of controlled, randomized studies comparing these two procedures.
The purpose of this retrospective study is to present a 11-year experience with upper third gastric cancer from one institution treated by either total gastrectomy or proximal gastrectomy. Comparisons of operative mortality and morbidity, recurrence, and long-term survival rates are presented.
MATERIALS AND METHODS
The records of 1,520 patients with primary gastric adenocarcinoma operated on the authors' department from January 1990 to December 2000 were reviewed. Among these were 259 patients (17%) who underwent resection for the upper third gastric cancer. Upper third gastric cancer was defined as adenocarcinoma of the upper one third of the stomach with or without involvement of the esophagogastric junction, according to the classification of Japanese Gastric Cancer Association (JGCA), (5). The location of the primary cancer was identified by esophagogastroscopy or radiographic contrast examination. Patients with a remnant gastric cancer, and those who required a transthoracic esophagectomy were not considered for this study.
The extent of gastric resection depended on the judgement of the attending surgeon. In addition, tumor location and intraoperative verification of tumor-free resection margins were used to select patients for total or proximal gastrectomy. Proximal gastrectomies were performed in 74 patients. The operative procedures included resection of the upper two-thirds of the stomach and the distal esophagus, followed by esophagogastrostomy with a 25-mm circular stapler. Total gastrectomies were performed in 185 patients followed by Roux-en-Y esophagojejunostomy with a 25-mm circular stapler, as well.
A standard D2 lymph node dissection of nodal stations 1~11 (excluding the suprapyloric and infrapyloric stations 5 and 6 in proximal gastrectomy), in accordance with the guidelines of the JGCA, was performed. Additional organ resection was performed to facilitate a more extensive lymph node dissection. The residual tumor classification (R) and pathological staging was performed according to the UICC classification (6).
All patients were followed up regularly according to a standardized follow-up program which consisted of physical examination, routine blood chemistry, tumor markers, chest X-ray, abdominopelvic ultrasonography and computed tomography. Endoscopy was performed every 6 months or yearly, and other radiological studied were performed only on suspicion of recurrence. The main patterns of recurrence were recorded as the first site of detectable failure at the time of diagnosis. Any metastatic involvement observed after identification of the first recurrence was not considered. The type of recurrence was classified as follows: (i) local if tumor was detected in the remnant stomach, anastomosis, or in the perigastric lymph nodes; (ii) distant if tumor recurred as peritoneal and hematogenous pattern, or recurred at the distant lymph nodes. The follow-up was closed on July 2003; the median length of follow-up was 34 months (range 1~131 months). At the time of the last follow-up, 139 patients (53.7%) were still alive, 15 (5.8%) were lost to follow-up and 105 (40.5%) had died from recurrence or other causes.
Reflux esophagitis and anstomotic stricture were confirmed by endoscopic examination and biopsy along with associated symptoms, such as heartburn, regurgitation, and dysphagia.
1) Statistical analysis
Cumulative survival rates were calculated by the Kaplan-Meier method, and log-rank test was used to compare the survival curves of the two groups of patients. The lost cases and deaths from any other causes than gastric cancer were treated as censored data for the analysis of survival rates. The Cox's proportional hazard model was used to evaluate various factors simultaneously. Other statistical analyses were performed by the t-test and χ2 test. A P value less than 0.05 was considered to be statistically significant. All statistical analyses were carried out with the Statistical Package for the Social Sciences (SPSS®, Chicago, IL) program.
RESULTS
1) Patient characteristics
Detailed clinicopathological characteristics of the study group are presented in Table 1. With the exception of tumor size, both groups were comparable with regard to age, sex, gross and histological type of tumors. There was a higher splenectomy rate in the total gastrectomy group (45.9%) than in the proximal gastrectomy group (18.9%). The mean number of lymph nodes retrieved in the surgical specimen was significantly higher for total gastrectomy (30.1±12.9 nodes) than for proximal gastrectomy (24.7±12.8; p=0.001), whereas the number of histopathologically positive lymph nodes were not different between the two groups (6.1±9.1 versus 4.5±7.8 respectively).
Curative resection (R0) was performed in 232 patients (89.6%).The distributions of depth of tumor invasion, lymph node and distant metastasis (pT, pN and pM stages) as well as the residual tumor classification were also similar in the two groups (Table 2).
2) Morbidity and mortality
There were no differences in major complications, including anastomotic leakage, intraabdominal abscess, wound infection, pancreatitis/fistula, and other surgery-related complications between the two groups. However, the incidences of reflux esophagitis and anastomotic stricture were more common in the proximal gastrectomy group compared with the total gastrectomy group (Table 3).
The overall postoperative 30-day death rate was 1.9%; three patients (1.6%) in the total gastrectomy group and two patients (2.7%) in the proximal gastrectomy group, with no significant difference between the groups.
3) Recurrence and survival
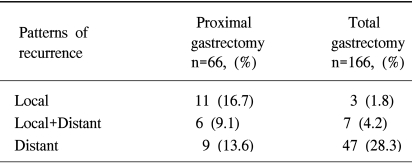
The median follow-up period for patients undergoing an R0 resection was 36 months. Of these patients, recurrence was confirmed by clinical and radiological examination or by reoperation in 26 patients (46.1%) of the proximal gastrectomy group, and 57 (39.6%) of the total gastrectomy group, respectively. Regarding the main patterns of recurrence, local recurrence was dominant in the proximal gastrectomy group, whereas distant recurrence was dominant in the total gastrectomy group (Table 4). The median time to recurrence was 16 months in the total gastrectomy group, compared to 26 months in the proximal gastrectomy group.
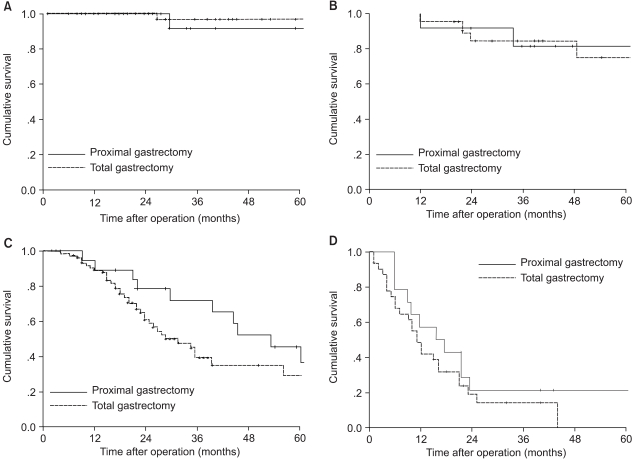
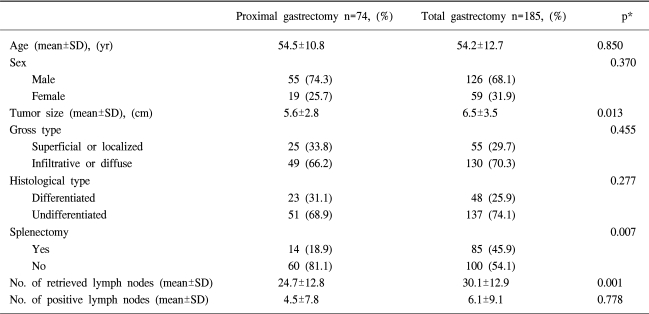
The estimated 5-year overall survival rate for the entire group was 51.8%. There was no significant difference in overall 5-year survival rates between the proximal gastrectomy group (54%) and the total gastrectomy group (47.8%), (Fig. 1). When survival was analyzed within each stage (stage I to IV), there were no differences in the 5-year survival rates between the two groups (Fig. 2).
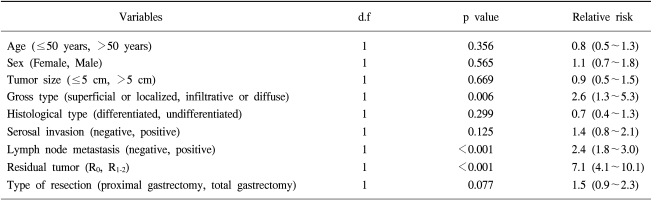
Multivariate analysis showed that residual tumor, lymph node metastasis and gross type of tumor were independent prognostic factors in patients with upper third gastric cancer. The extent of gastric resection was not an independent prognostic factor (Table 5).
DISCUSSION
Apart from its incidence, a striking difference in the characteristics of proximally located gastric cancer is observed between the east and the west. Type I tumor, or adenocarcinoma of the lower esophagus often associated with Barrett's esophagus, according to Siewert's classification (7) was reported to be the most prevalent type in the European institutions (8,9). However, Type III subcardial carcinoma was by far the most common type in Japan (10) as well as in the current study. Thus, most patients enrolled in this study had Type III tumors that required treatment according to the well established gastric cancer guidelines.
There has been controversy about the extent of gastric resection for the treatment of the upper third gastric cancer. Advocates of total gastrectomy stress the possible benefit of a more radical lymph node dissection (11). It is apparent from the number of lymph nodes retrieved in the current study that total gastrectomy does result in a more complete lymph node dissection. However, several studies have reported that the incidence of metastasis to the lymph nodes along the lower stomach, especially to the supra- and infrapyloric lymph nodes, was very low in cases of upper third gastric cancer, and that resection of theses nodes had little impact on survival (12~16). Therefore, proximal gastrectomy could be an alternative to total gastrectomy provided that the tumor size and location permit preservation of an adequately large remnant of stomach without compromising tumor clearance distally.
Recently, demonstrated that the time to recurrence and 5-year survival rate were not different between proximal and total gastrectomies, and concluded that total gastrectomy was not necessary for proximal gastric cancer (15). Showed that although three tumors invading the serosa had metastatic lymph nodes along the lower part of the stomach (16), the survival rate was not different between proximal and total gastrectomies; they advocated that proximal gastrectomy should be performed when the tumor invasion was not beyond the muscularis propria. Similarly, we found no difference in the overall survival rates between the two groups, irrespective of tumor stage. Moreover, our multivariate analysis showed that significant prognostic factors were residual tumor, lymph node metastasis, and infiltrative or diffuse type tumor; the extent of resection was not an independent prognostic factor for the upper third gastric cancer.
Despite these findings, scepticism about proximal gastrectomy lingers among the surgeons for several reasons. Previous reports concerning operative morbidity and mortality related to proximal gastrectomy are conflicting, some showing no difference (3,15) and others showing high morbidity and mortality rates as compared with total gastrectomy (17). Similar morbidity and mortality rates observed in the present study, regardless of the extent of resection, suggests that both procedures can be performed safely at an acceptable level. However, the incidences of reflux esophagitis (16.2%) and anastomotic stricture (35.1%) were more common in the proximal gastrectomy group compared with the total gastrectomy group (0.5 and 8.1% respectively). Like the present authors, several investigators have postulated that direct anastomosis between the esophagus and the remnant stomach result in significant gastroesophageal reflux, and subsequently, to various degrees of esophagitis and stricture (18,19). To avoid this, esophagogastrostomy such as valvuloplasty plus fundoplasty (20), gastric tube reconstruction (21), jejunal interposition (22), and jejunal pouch interposition (23) were devised. Although these procedures are complicated and time-consuming, we have used the jejunal pouch interposition as the usual reconstruction method following proximal gastrectomy since 2000, and a prospective study currently is ongoing as part of our continuing efforts to assure the efficacy of proximal gasrectomy and jejunal pouch interposition for the treatment of early cancer in the upper third of the stomach.
The next important finding of this study is the relatively high incidence of local recurrence in the proximal gastrectomy group. Local recurrence following proximal gastrectomy is a cause for concern on the assumption that it might be prevented if total gastrectomy had been performed. In this study, local recurrence including the combined pattern occurred in 17 out of 66 patients (25.7%), with most of them occurring at the remnant stomach or anastomosis. Among them, 4 patients underwent completion total gastrectomy at 13, 38, 47, and 78 months after initial proximal gastrectomy, respectively. The causes of local recurrence following a curative resection of gastric cancer may be an inadequate resection margin, either proximally or distally, along with the possibilities of remnant cancer, or a missed multiple gastric cancer (24). Considering the wide variance of time to recurrence in our series (8 to 78 months), we could partially explain the causes of a local recurrence, following proximal gastrectomy, by all of these possibilities. In our previous study (25), the risk factors for local recurrence following proximal gastrectomy were infiltrative or the diffuse type tumor, greater than 5 cm in tumor size and serosal invasion. Therefore, we believe that proximal gastrectomy should be avoided in these patients with these risk factors of local recurrence, whenever possible. Finally, as this study was non-randomized, there may be an element of bias. Further prospective studies are needed to confirm the adequacy of long-term results between these surgical approaches.
CONCLUSIONS
The extent of resection for the upper third gastric cancer does not affect long-term outcomes in terms of survival and mortality rate. However, proximal gastrectomy not only contributes to significant reflux esophagitis and anastomotic stricture, but also increase the likelihood of local recurrence. Therefore, further studies on these problems as well as patient quality of life are necessary to reappraise the proximal gastrectomy as the surgical procedure of choice for the upper third gastric cancer.