Introduction
Caveolin-1 (CAV-1) is a member of the caveolin family of scaffold proteins that regulate the activity of many signaling molecules inside caveolae. Functionally, CAV-1 is implicated in various normal cell behaviors, including lipid transportation, cell growth, and death regulation. In transformed cells, alterations of CAV-1 and its loss are associated with reduced invagination and a loss of caveolae [1]. Expression of CAV-1 in cancer cells is cell-type dependent; it is more likely to be downregulated in ovarian, colon, and mesenchymal sarcomas and upregulated in lung, bladder, esophageal, and pancreatic cancers [2]. In breast cancer, CAV-1 was initially considered a tumor suppressor based on the findings of low gene expression related to the mechanisms underlying the inactivating point mutation, silencing, or downregulation of CAV-1 in invasive cancers [3]. However, a growing body of evidence has shown that high levels of CAV-1 expression are more associated with basaloid cancers than estrogen receptor–negative and/or progesterone receptor–negative, or HER-2–expressing cancers [4]. In addition, CAV-1 expression has also been associated with aggressive cancer subtypes, such as inflammatory breast cancer, due to promoter hypomethylation [5]. Recent studies of the biologic activities of CAV-1 have focused more on its roles in cancer-associated fibroblasts. The protective roles of stromal CAV-1 against mammary hyperplasia and tumorigenesis in breast cancer have been demonstrated in multiple studies [6,7], and the loss of CAV-1 in stromal cells has been shown to be an independent prognostic factor in patients with early breast cancer [2,8]. These findings imply that the biological functions of CAV-1 might be multidimensional and cell-type dependent in breast cancer. However, the biological roles of CAV-1 in the epithelial compartment, particularly in the triple-negative breast cancer subtype, are largely unknown.
Taxanes are potent anticancer agents that function by binding to the β subunits of tubulin, which results in stabilization of tubulin polymerization and repression of the dynamic instability of spindles. These activities lead to cell cycle arrest in the G2/M phase. Docetaxel (DTX) and paclitaxel (PTX) are commonly used in the treatment of metastatic breast, lung, ovarian, and digestive cancers; however, DTX treatment has shown better clinical efficacy compared to PTX in patients with breast cancer [9]. The better affinity of DTX for tubulin and the subsequent induction of both apoptotic and necrotic cell death have been suggested as potential mechanisms for the superior antitumor activity of DTX compared to PTX. Clinical data have indicated that the addition of taxane to conventional adjuvant chemotherapy improves the prognosis of patients with early breast cancer, and these benefits are mainly observed in the triple-negative subgroup [10]. However, the use of taxanes is limited due to a variety of toxicities, including bone marrow suppression, arrhythmia, peripheral neuropathy, skin and nail changes, and gastrointestinal problems. Thus, identification of biomarkers that can predict the therapeutic effects of taxanes in breast cancer is an important unmet clinical need. Significant efforts have been made to identify potential predictive markers, including single microtubule-associated protein, tau [11], or panels of gene signatures [12]. However, there is no biomarker for DTX that can guide physicians in selecting the best treatment regimen.
A recent in vitro study showed that the specific phosphorylation of CAV-1 enhances the PTX-mediated cytotoxicity in MCF-7 cells, which are a luminal type of breast cancer cells [13]. However, the potential association of CAV-1 and DTX response in various subtypes of breast cancer is not yet fully understood. Therefore, in this study, we attempted to determine whether CAV-1 functions as a modulator of cell growth and the cytotoxic activity of DTX in different subtypes of breast cancer cells in in vitro and in vivo models.
Materials and Methods
1. Cell culture
The ZR75-1, T47D, SKBR3, HCC1954, BT474, Hs578T, MDA-MB-231, MDA-MB-468, and MCF-7 cell lines were purchased from the American Type Culture Collection (ATCC, Manassas, VA). ZR75-1, T47D, SKBR-3, HCC1954, and MDA-MB-231 cells were maintained in RPMI-1640 medium (Thermo Fisher Scientific Inc., Waltham, MA). MCF-7 cells were maintained in RPMI-1640 medium supplemented with 4 mg/mL of insulin, human recombinant, zinc solution (Life Technologies, Grand Island, NY). BT474, Hs578T, and MDA-MB-468 cells were maintained in Dulbecco’s modified Eagle’s medium (Welgene Inc., Deagu, Korea). All of the culture media were supplemented with 10% fetal bovine serum, 100 units/mL of penicillin, 100 mg/mL of streptomycin, and 2 mM of L-glutamine.
2. Cell viability assay
Cells were seeded at a density of 2×105 cells in 6-well plates. After 24 hours, the cells were treated with various concentrations of DTX. Quantitative measures of cell viability were determined using a 3-[4,5-dimethylthiazol-2-yl]-2, 5-diphenyltetrazolium (MTT) assay. Briefly, MTT solution was added to the plates after treatment with DTX, and the plates were incubated for another 4 hours at 37°C. The absorbance of the converted MTT dye was measured at 540 nm using an iMARK microplate reader (Bio-Rad Laboratories Inc., Berkeley, CA). In all experiments the cell viability was expressed as a relative percentage of the untreated cells with error bars. All experiments were repeated at least twice. Statistical analyses were performed using Student’s t tests. p-values less than 0.05 were considered significant.
3. Western blot analyses
The cells were harvested in lysis buffer (20 mM Tris-HCl [pH 7.5], 0.5% Triton X-100, 150 mM sodium chloride, 10% glycerol, 1 mM sodium orthovanadate, 20 mM sodium fluoride, and 100 mM phenylmethylsulfonyl fluoride) containing Xpert protease inhibitor cocktail solution (genDEPOT Inc., Barker, TX) and incubated on ice for 50 min. Proteins (30 μg) were separated on 8% or 12% sodium dodecyl sulfate-polyacrylamide gel and transferred to polyvinylidene fluoride membranes. The membranes were blocked in 5% nonfat milk for 1 hour at room temperature and incubated with the appropriate primary antibodies at 4°C overnight, followed by washing and incubation with horseradish peroxidase-conjugated secondary antibodies. The protein bands were visualized using the ECL system (GE Healthcare, Piscataway, NJ), and the images were developed on X-ray film (Agfa HealthCare NV, Mortsel, Belgium). Antibodies for the estrogen receptor, ErbB2, p53, Bcl-2, and Bax were obtained from Santa Cruz Biotechnology Inc. (Santa Cruz, CA). Antibodies for CAV-1, poly-ADP-ribose polymerase, and pBcl-2 were purchased from Cell Signaling Technology Inc. (Danvers, MA). β-Actin was obtained from Sigma-Aldrich Co. LLC (St. Louis, MO).
4. [3H]thymidine incorporation assay
DNA synthesis was measured using a [3H]thymidine uptake assay. The cells were plated in 6-well plates, serum starved for 48 hours, and then treated with DTX for 48 hours. In the last 4 hours, the cells were pulsed with 1 μCi/mL [3H]thymidine (GE Healthcare, Milano, Italy). The cells were washed twice with serum-free medium and precipitated with 5% trichloroacetic acid for 15 minutes at 4°C. The precipitates were washed twice with 95% ethanol, dissolved in 1 mL of NaOH, and then analyzed by liquid scintillation counting.
5. CAV-1 stable transfection
A pcDNA3.1 expression plasmid containing CAV-1 cDNA was obtained from Dr. Hong-Guang Zhu (Fudong University, Shanghai, China). MCF-7 and ZR75-1 cells were stably transfected with a CAV-1 expression plasmid (CAV-1) or empty vector (EV) with Lipofectamine 2000 reagent (Life Technologies). Forty-eight hours after the transfection, the media was replaced with G418-containing media (400 μg/mL). Individual colonies were selected during the subsequent 2 weeks of selection, and positive transfectants were screened by western blot analyses.
6. siRNA transfection
siRNA targeting CAV-1 (sense, 5ʹ-AGACGAGCUGAGCGAGAAGCA-3ʹ; antisense, 5ʹ-UGCUUCUCGCUCAGCUCG UCU-3ʹ; Lot No. S20121127-020) was purchased from Bioneer Corporation (Daejeon, Korea). MDA-MB-231 and Hs578T cells were seeded and transiently transfected with the CAV-1 expression plasmid or EV with Lipofectamine 2000 reagent (Life Technologies). After transfection, the downregulation of CAV-1 expression was confirmed by western blot analyses.
7. Cell cycle analysis
Harvested cells were fixed in 70% ice-cold ethanol and incubated with 4 μg/mL of propidium iodide and 100 μg/mL of RNase A for 45 minutes at 37°C. Stained samples were analyzed using FACScalibur and Cell Quest Pro (BD, Oxford, UK).
8. Apoptosis assay
Apoptosis was evaluated using annexin V–fluorescein isothiocyanate (annexin V–FITC; BD Biosciences, San Jose, CA). Briefly, cells were seeded and treated with DTX (10 nM) for 24 hours. The cells were then harvested and stained with 5 μL of annexin V–FITC and 5 μL of propidium iodide at room temperature for 15 minutes. The stained samples were analyzed by FACScalibur and Cell Quest Pro (BD).
9. In vivo breast cancer xenograft assays
Female nude mice (BALB/c, nu/nu), aged 6 weeks and weighing 18-22 g, were purchased from Orient Bio Inc. (Seongnam, Korea). All mice were implanted with 0.72 mg of 60-day release 17β-estradiol pellets (2.5 mg/p, Innovative Research of America, Sarasota, FL). MCF-7 (5×106) cells were implanted subcutaneously into mice (n=5 per group). Seven days later, EV/MCF-7 and CAV-1/MCF-7 cells (5×106 cells) in 100 μL of phosphate buffered saline (PBS) were injected subcutaneously into the right flank of the mice. When the tumor reached a size greater than 200 mm3, the mice were treated with PBS or DTX at 10 mg/kg twice a week in the four following randomly divided groups (5 mice per group): EV/MCF-7 (PBS control and DTX) and CAV-1/MCF-7 (PBS control and DTX). The tumor diameters were measured using calipers twice per week for more than 3 weeks. The tumor volumes were calculated using the following formula: Volume (mm3)=Width2×Length/2. The mice were then sacrificed, and the tumor tissues were collected for histological analyses. Differences in tumor growth between the animal treatment groups were analyzed using two-way repeated measures analysis of variance (ver. 10.0, SPSS Inc., Chicago, IL). In all cases, p-values less than 0.05 were considered significant. All animal procedures were performed according to the Institutional Animal Care and Use and Institutional Review Board committee (KIRAMS2011-20) at the Korea Institute of Radiological and Medical Science (KIRAMS, Seoul, Korea).
10. Histopathologic analysis
Expression of CAV-1 (Santa Cruz Biotechnology Inc.), Ki67 (Dako North America Inc., Carpinteria, CA), and p53 (DO-7, Leica Biosystems Newcastle Ltd., Newcastle upon Tyne, UK) was determined using an immunohistochemical approach on formalin-fixed paraffin-embedded 5-μm-thick tissue sections. For each case, one representative section was stained with hematoxylin and eosin.
Results
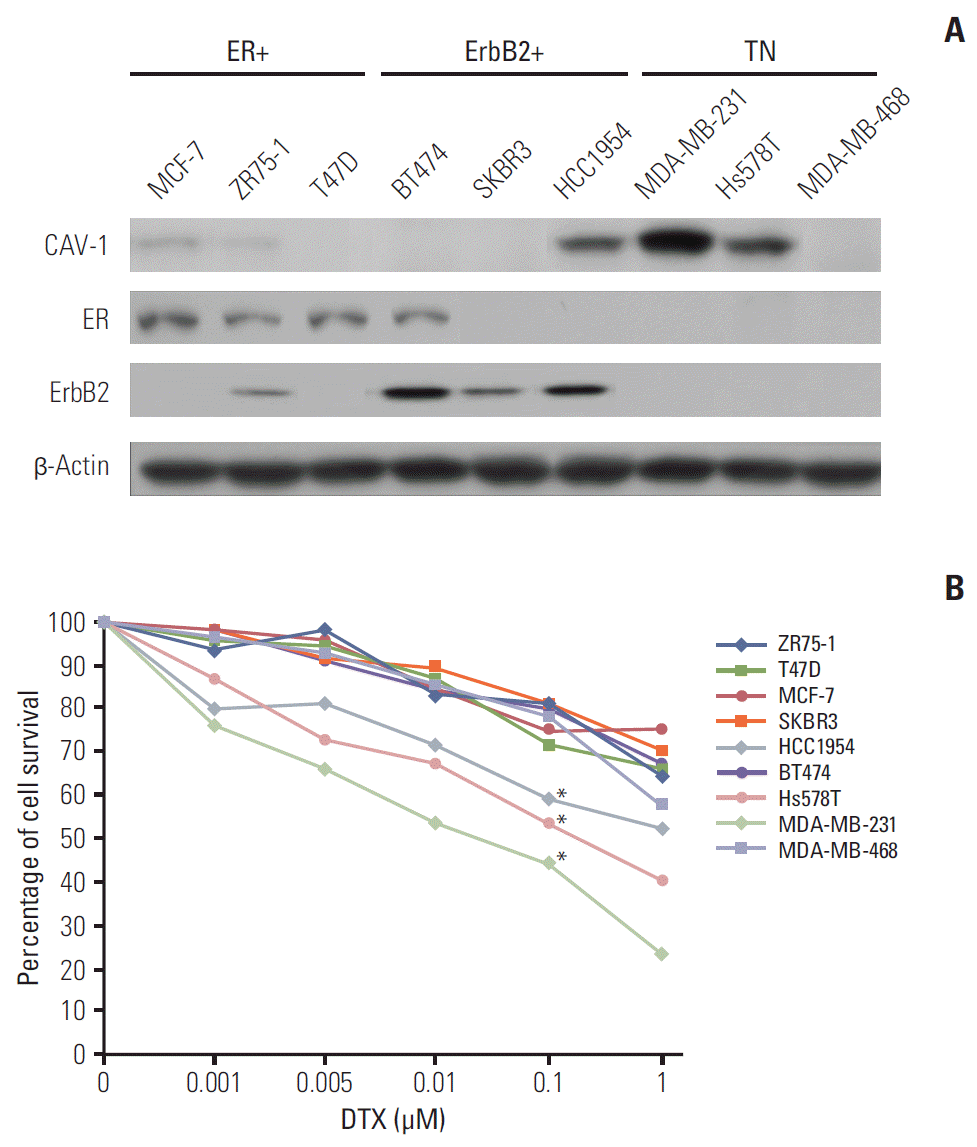
1. Correlation of CAV-1 expression and DTX sensitivity in three subtypes of breast cancer cells
To investigate the potential relationship between CAV-1 and DTX sensitivity in three representative subtypes (estrogen receptor–positive, ErbB2-positive, triple negative) of breast cancer cells, we first profiled the levels of expression of CAV-1 in a panel of nine different breast cancer cell lines (ZR75-1, T47D, MCF-7, SKBR3, HCC1954, BT474, Hs578T, MDA-MB-231, and MDA-MB-468). As shown in Fig. 1A, CAV-1 expression was detected in two of the triple-negative cell lines (MDA-MB-231 and Hs578T) and one of the ErbB2-positive cell lines (HCC1954), while it was downregulated in other breast cancer cell lines. Next, to determine whether DTX sensitivity differed according to the levels of CAV-1 expression, the cell survival rate after DTX treatment was analyzed in these cell lines. As shown in Fig. 1B, the percentage of viable cells in the CAV-1-expressing cell lines (MDA-MB-231, Hs578T, and HCC1954) was significantly decreased after DTX treatment in a dose-dependent manner compared to that in other breast cancer cell lines (p < 0.05). These results suggested that CAV-1 might have a modulating role in DTX-induced cell death in breast cancer cells.
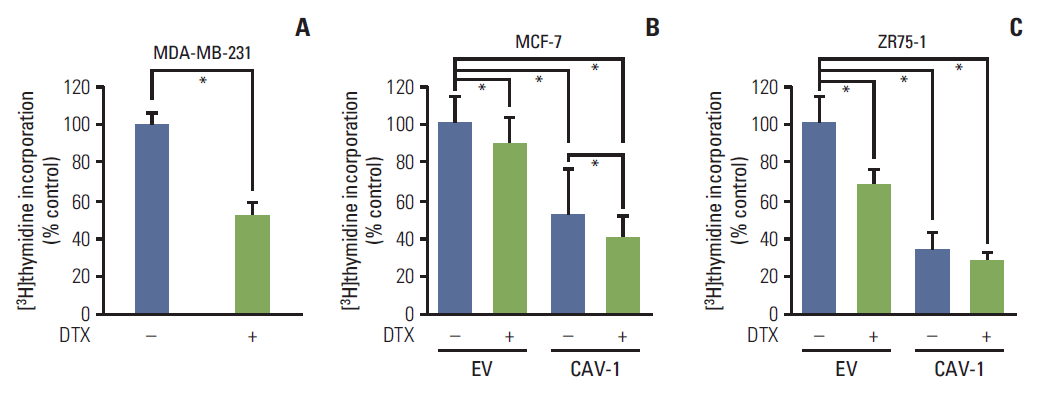
2. Effects of CAV-1 on the antiproliferative activity of DTX
To elucidate the biological effects of CAV-1 in the antiproliferative activities of DTX, we examined the changes in thymidine incorporation after DTX treatment in breast cancer cell lines with or without CAV-1 expression. As shown in Fig. 2A, CAV-1–expressing MDA-MB-231 cells showed significantly decreased thymidine incorporation after DTX treatment (p < 0.05). When the low CAV-1–expressing MCF-7 and ZR75-1 cells were transfected by either the CAV-1 gene or a control vector, CAV-1–transfected cells showed an obvious decrease in thymidine incorporation compared to control vector-transfected cells (Fig. 2B and C). In addition, the antiproliferative effects of DTX were greatly enhanced by CAV-1 overexpression, and this finding was consistent in both MCF-7 and ZR75-1 cells. These findings indicated that CAV-1 might act as an antiproliferative factor and a modulator of DTX activity in breast cancer cells.
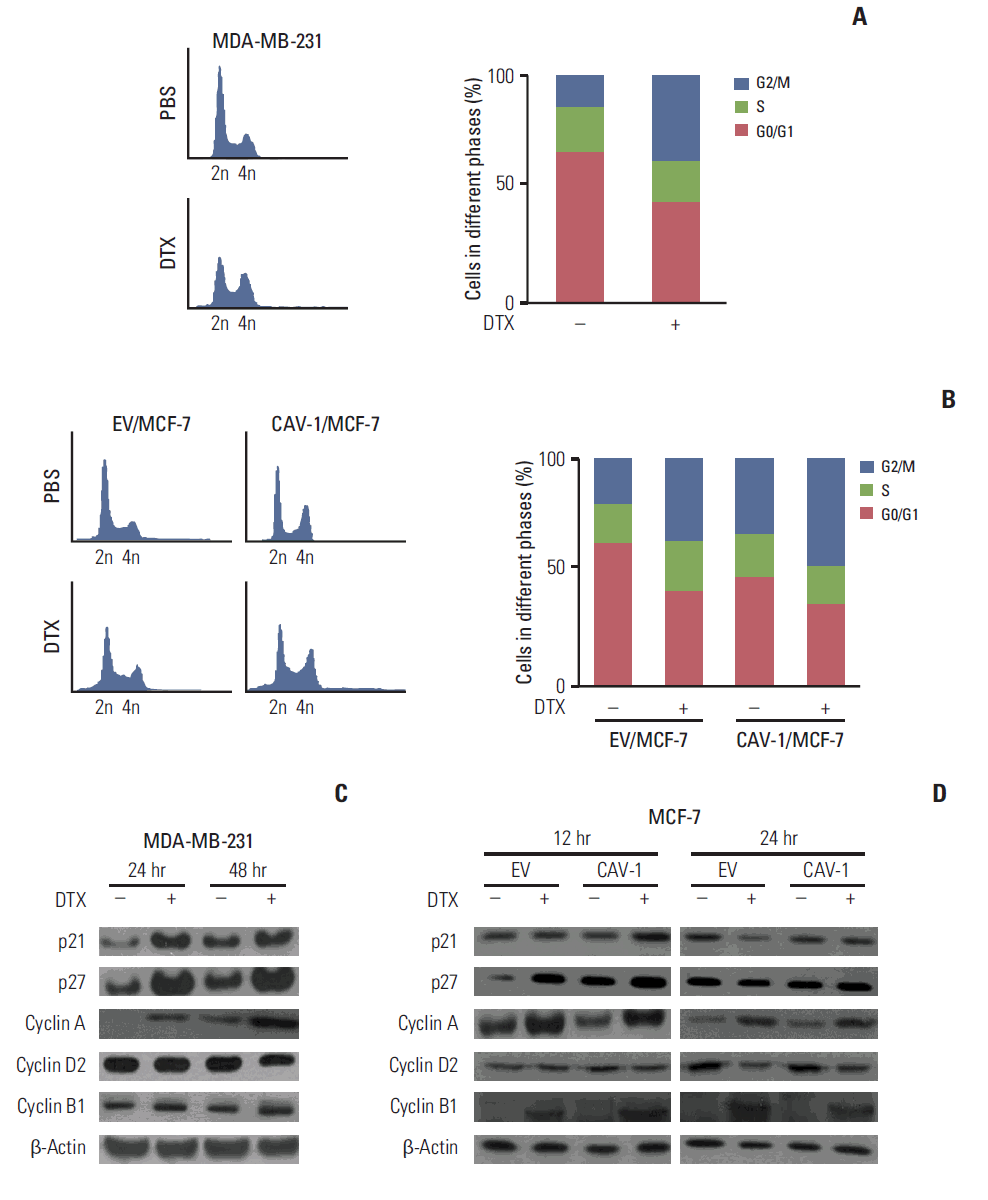
3. CAV-1 increased DTX-induced G2/M cell cycle arrest
The antiproliferative activities of DTX mainly result from its inhibitory effects on mitotic cell division between metaphase and anaphase, which stabilizes microtubule assembly and prevents physiological microtubule depolymerization/disassembly [14]. To determine whether CAV-1 altered DTX-induced cell cycle arrest, we examined the changes in the cell cycle that occurred after DTX treatment in MDA-MB-231 and MCF-7 cells with or without CAV-1 expression. As shown in Fig. 3A and B, G2/M arrest was expectedly induced after DTX treatment in both MDA-MB-231 and MCF-7 cells. In addition, significantly higher percentages of cells arrested in the G2/M phases after DTX treatment were observed in CAV-1–transfected MCF-7 cells compared to EV-transfected MCF-7 cells (35.7% and 47.2%, respectively, p < 0.05). We then analyzed the levels of expression of cell cycle-related proteins in these cell lines after DTX treatment. In MDA-MB-231 cells, p21, p27, and cyclin A were increased, but the expression of cyclin D2 was decreased 48 hours after DTX treatment (Fig. 3C). CAV-1–transfected MCF-7 cells showed relatively enhanced expression levels of p21 and p27 and expression levels of cyclin D2 were downregulated compared to EV-transfected MCF-7 cells after DTX treatment (Fig. 3D). In addition, the expression level of cyclin B1 showed a greater increase after DTX treatment in CAV-1–transfected cells compared to that in EV-transfected cells. These results suggested that CAV-1 expression contributed to the DTX-induced G2/M cell cycle arrest in breast cancer cells.
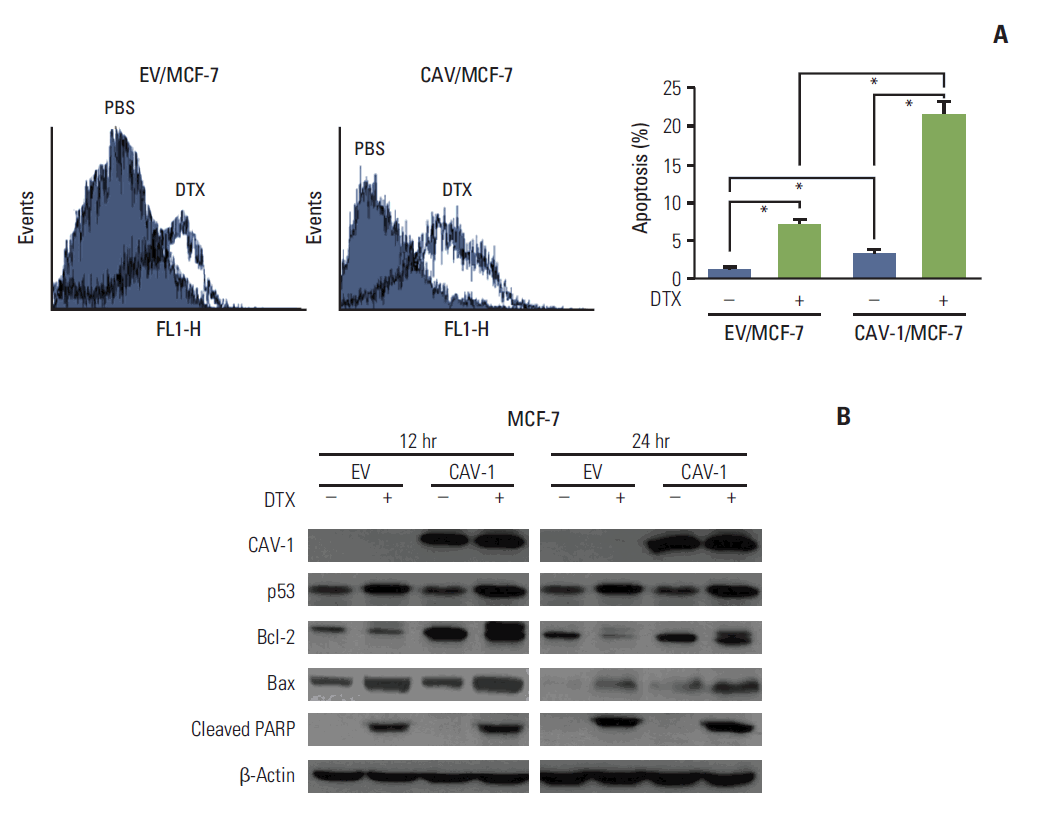
4. CAV-1 modulated DTX-induced apoptotic cell death
To investigate the potential effects of CAV-1 in DTX-induced apoptotic cell death in breast cancer cells, the percentage change in apoptotic cell death was analyzed with annexin V-positive cells after DTX treatment. As shown in Fig. 4A, the calculated percentage of apoptotic cells induced by DTX was highly increased in CAV-1–transfected MCF-7 cells compared to EV-transfected MCF-7 cells (17.8% vs. 4.2%, respectively; p < 0.05). In addition, the levels of p53, Bax, and cleaved poly-ADP-ribose polymerase showed a greater increase after DTX treatment in CAV-1–transfected MCF-7 cells compared to EV-transfected MCF-7 cells (Fig. 4B). Unexpectedly, we found that the levels of Bcl-2, an antiapoptotic protein, were highly upregulated in CAV-1-transfected MCF-7 cells compared to EV-transfected cells. In addition, the increased levels of Bcl-2 expression observed in CAV-1–transfected MCF-7 cells after DTX treatment were detected as two separate bands, which represented the phosphorylated and inactivated form of Bcl-2 protein, as previously reported [15].
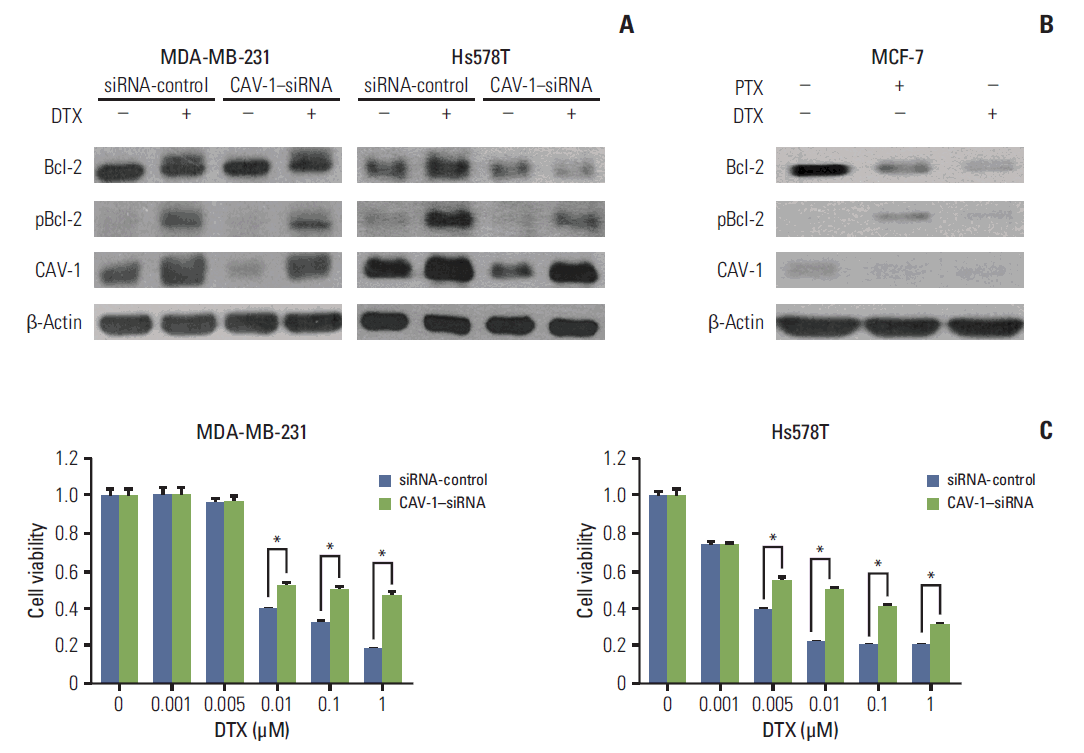
Next, determine whether the mechanisms for this DTX-induced apoptotic cell death were affected by the endogenous expression levels of CAV-1, we assessed the total and phosphorylated forms of Bcl-2 protein after DTX treatment in MDA-MB-231 and MCF-7 cells. As shown in Fig. 5A, two triple-negative breast cancer cell types (MDA-MB-231 and Hs578T) with high levels of endogenous CAV-1 expression showed increased CAV-1 expression and the induction of Bcl-2 phosphorylation after DTX treatment. However, the changes in the levels of CAV-1 expression and phosphorylated Bcl-2 were diminished after CAV-1 siRNA transfection. In contrast, MCF-7 cells with very low levels of CAV-1 showed decreased Bcl-2 expression after DTX treatment, and CAV-1 expression was not induced (Fig. 5B). In addition, the silencing of CAV-1 resulted in significantly decreased DTX-induced cytotoxicity in two triple-negative breast cancer cells (Fig. 5C). Therefore, these results suggested that the endogenous expression of the CAV-1 gene plays a pivotal role in DTX-induced apoptosis in breast cancer cells.
5. CAV-1 enhanced the antitumor effects of DTX in a breast cancer in vivo model
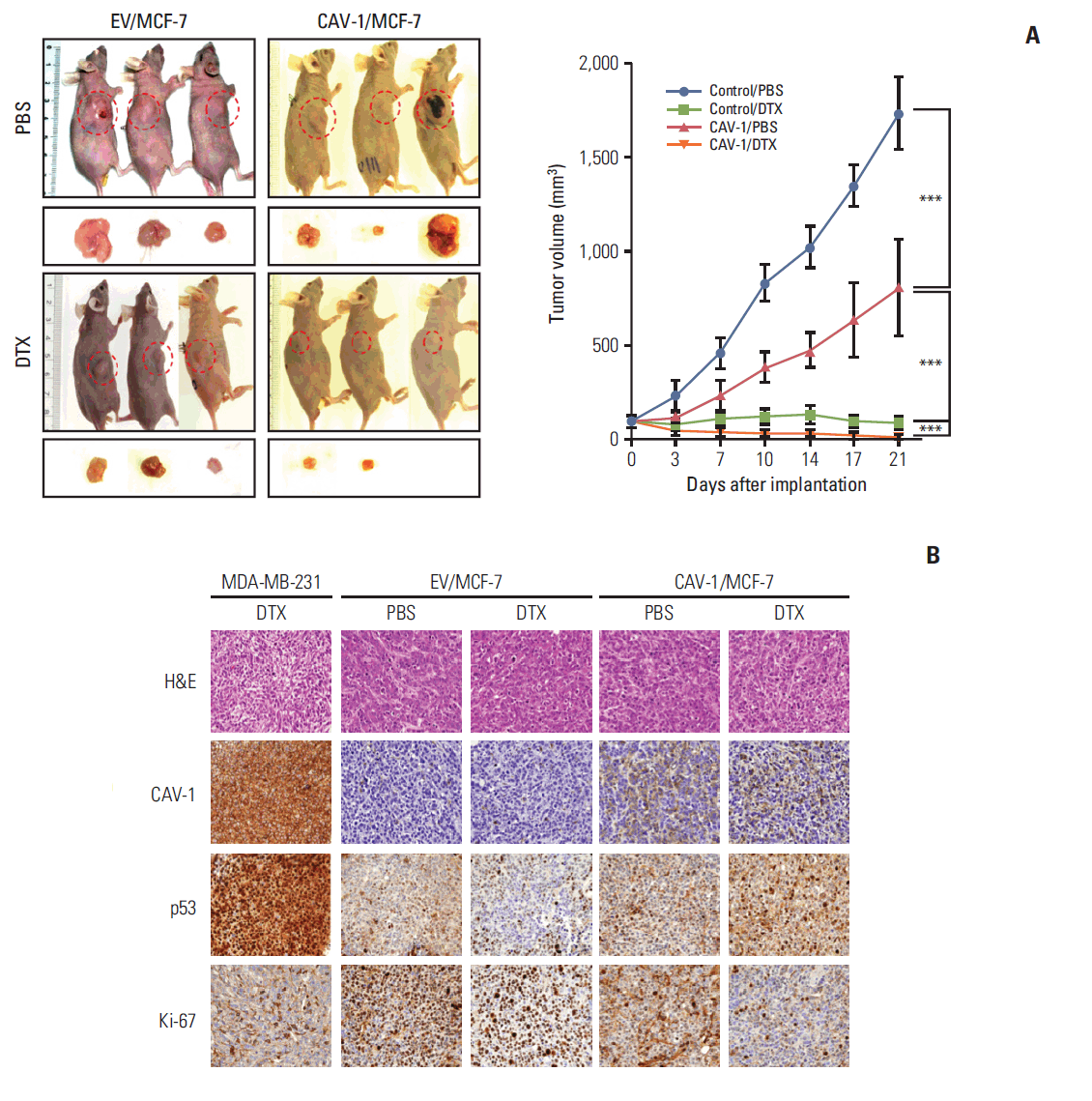
The findings observed in the in vitro study were further evaluated in nude mice. The mean tumor size in each group of mice showed that the tumors of EV/MCF-7 cells treated with PBS showed the most rapid growth, followed by tumors of EV/MCF-7 cells treated with DTX. Notably, CAV-1–overexpressing MCF-7 cells (CAV-1/MCF-7) showed greatly attenuated tumor growth compared to that in EV/MCF-7 cells, and these growth-inhibitory effects were further enhanced by DTX treatment (p < 0.001) (Fig. 6A). Immunohistochemical analyses were performed to examine the biological changes in the in vivo tumors (Fig. 6B). The protein expression of p53 was induced both by DTX treatment and by the ectopic expression of CAV-1 in MCF-7 cells, and its expression was further enhanced by the combinatorial effects of DTX and CAV-1. Consequently, the immunostaining of Ki-67 was more decreased in DTX-treated and CAV-1–transfected MCF-7 tumors compared to DTX-treated EV/MCF-7 tumors. Further antiproliferative effects of DTX in the presence of CAV-1 expression in MCF-7 cells were confirmed by Ki-67 staining.
Discussion
The current study addressed the question of whether CAV-1 had tumor-suppressive functions and activity in DTX-mediated cytotoxicity in different subtypes of breast cancer cells. The main findings of this study were that CAV-1 had antiproliferative and cell cycle arrest functions and modulated the DTX-induced apoptosis of MDA-MB-231 and MCF-7 breast cancer cells, which were confirmed in vivo.
CAV-1 has been suggested to function either as a tumor suppressor or as an oncogene depending on the tumor type and context of tumor progression. In breast cancer, oncogenic functions of CAV-1 have been proposed based on the findings of its upregulation in highly aggressive drug-resistant subsets of basal-like, metaplastic, or inflammatory cancers [16]. While its prognostic role is unclear, in a comprehensive meta-analysis, the epithelial expression of CAV-1 showed significant positive correlations with the clinical characteristics of triple negativity and positivity for cytokeratin 5/6 [17]. More recently, overexpression of CAV-1 was observed in the claudin-low mesenchymal subtype of breast cancer cells in murine models, implicating it as a possible biomarker for this challenging subgroup [18]. However, these observations have not been clearly supported by biologic studies in any triple-negative breast cancer model. In the current study, CAV-1 was highly expressed in two triple-negative and one ErbB2 subtype of breast cancer cell lines in our panel (Fig. 1A). In addition, ectopic expression of CAV-1 decreased cell proliferation by inducing cell cycle arrest in the G2/M phase in MCF-7 cells (Fig. 2). Of note, CAV-1 per se had efficacy similar to that of DTX in MCF-7 cells, which supported the findings of earlier studies reporting that CAV-1 is a tumor suppressor in human breast cancer cells [19]. Therefore, determine the clinical relevance of CAV-1 in the triple-negative mesenchymal subtype of breast cancer is warranted.
The phosphorylated form of CAV-1 is known to enhance PTX-mediated cytotoxicity in MCF-7 cells. Shajahan et al. suggested that CAV-1 sensitizes apoptosis by the induction of p21 in a p53-dependent manner and by suppression of Bcl-2. In reality, however, CAV-1 expression is observed mainly in the basal type of tumors, which contain nonfunctional p53 protein. Indeed, in this study, the CAV-1–expressing cell lines that showed high sensitivity to DTX (HCC1954, MDA-MB-231, and Hs578T) are known to have mutant p53 [20]. Thus, in tumor cells with nonfunctional p53, different mechanisms might contribute to CAV-1 in taxane-mediated cytotoxicity. More recently, data have emerged suggesting that Bcl-2 plays an important role in PTX-mediated cytotoxicity. Although the precise molecular mechanism for regulating the expression levels of Bcl-2 family proteins is not clear, a study by Shajahan et al. [15] expanded prior understanding of the activity of CAV-1 by suggesting that the phosphorylated form of CAV-1α facilitates PTX-induced apoptosis by inactivating Bcl-2 and Bcl-xL. In the current study, dramatically increased levels of the phosphorylated form of Bcl-2 were observed in CAV-1 transfected MCF-7 cells compared to MCF-7 cells, which showed an increase in p53 protein rather than phosphorylated Bcl-2 after DTX treatment (Fig. 4). Conversely, the knock down of CAV-1 in MDA-MB-231 and Hs578T cells resulted in a significant decrease in the phosphorylated form of Bcl-2 after DTX treatment (Fig. 5A). These data suggested that the modulating effects of CAV-1 in DTX-induced apoptosis resulted mainly from the inhibitory activity on Bcl-2 in p53 nonfunctional MDA-MB-231 and Hs578T cells.
DTX is known to have a higher affinity for microtubules than PTX due to its more potent induction of Bcl-2 phosphorylation [14]. While DTX is one of the most commonly used chemotherapeutics in patients with breast cancer, predictive biomarkers of DTX sensitivity are still inconclusive [21]. Based on in vitro data, CAV-1 has been suggested as a sensitizer of PTX in its induction of apoptosis in estrogen receptor-positive breast cancer cells [13]. In the current study, unlike other triple-negative cell line, CAV-1–negative MDA-MB-468 cells showed very low sensitivity to DTX, and CAV-1–positive HCC1954 cells were significantly more sensitive to DTX than the other ErbB2 type of cells. These results suggested that CAV-1 might be an important determinant of DTX sensitivity in breast cancer. In our in vitro study, transfection of CAV-1 significantly dampened the proliferation of tumor cells, and its effects were enhanced by DTX treatment (Fig. 2). This was further confirmed by the knock down of CAV-1 in MDA-MB-231 and Hs578T cells, which resulted in significantly decreased DTX-induced cytotoxicity (Fig. 5C). Consistent with these results, in the in vivo study, immunohistochemical staining of the tumors showed a further increase in p53 expression and a decrease in the proliferative marker, Ki-67, in CAV-1–transfected MCF-7 cells treated with DTX (Fig. 6B). The data presented here confirmed that CAV-1, a highly versatile and context-dependent gene, had tumor-suppressive and chemomodulating effects in breast cancer cells in vivo. Given that CAV-1 is more preferentially expressed in the clinically challenging subtypes of breast cancers, including the basal, inflammatory, and metaplastic subtypes, it should be evaluated as a biomarker for prediction of response to DTX.
Our data suggesting the chemomodulating effects of CAV-1 in breast cancer cells were in contrast to the findings of previous reports that CAV-1 is a mediator of multidrug resistance [22-24]. Data addressing the relationship of CAV-1 and drug resistance was first reported from studies of adriamycin-resistant MCF-7 cells [22] and taxol-resistant A549 cells [25]. However clinical data from studies of patients with breast cancer, did not support the in vitro data. CAV-1 expression was not a prognostic factor in patients with breast cancer treated with anthracycline-based adjuvant treatment [4]. In a small series of patients with advanced non-small-cell lung cancer, positive expression of CAV-1 was a predictive factor of poor response to gemcitabine-based chemotherapy and worse clinical outcomes [24]. Data on the clinical significance of CAV-1 relevant to responses to chemotherapy and prognoses are still limited, therefore our data should be confirmed in a clinical study of taxane-treated patients with other subtypes of cancer.