AbstractPurposeThe aim of this study was to examine the clinical implications of a pathologically complete response after neoadjuvant chemoradiotherapy (CRT) followed by local excision for patients with cT2 rectal cancer who refused radical surgery.
Materials and MethodsSeventeen patients with cT2 primary rectal cancer within 6 cm from the anal verge who received neoadjuvant CRT and local excision because of patient refusal of radical surgery or poor performance status were included. Two patients had clinical involvement of a regional lymph node. Preoperative radiotherapy was delivered to the whole pelvis at a dose of 44 to 50.4 Gy in 22 to 28 fractions. All patients underwent transanal excision and eight patients (47%) received postoperative chemotherapy.
ResultsTen patients (59%) achieved ypT0. At a median follow-up period of 75 months (range, 22 to 126 months), four (24%) patients developed recurrence (two locoregional and two distant). The 5-year disease-free survival of all patients was 82%, and was higher in patients with ypT0 (90%) than in patients with ypT1-2 (69%, p=0.1643). Decreased disease-free survival was also observed in patients receiving capecitabine compared with 5-fluorouracil (54% vs. 100%, p=0.0298).
IntroductionNeoadjuvant chemoradiotherapy (CRT) followed by total mesorectal excision (TME) is a standard treatment in patients with locally advanced rectal cancer [1,2]. In addition to improved local control, reduced toxicity, increased sphincter preservation, and tumor downstaging have been demonstrated after neoadjuvant CRT [1]. Complete pathologic response (ypCR) of approximately 15% has been reported after CRT [3,4]. Patients with ypCR after CRT tend to have decreased local or distant recurrence and improved survival [4-6]. Some authors have reported that a wait-and-see policy without surgical resection could be possible with strict selection criteria [7,8].
Radical surgery is associated with significant morbidity, especially in cases of low rectal cancer [9,10], therefore, local excision might be an alternative treatment to radical surgery after neoadjuvant CRT in selected cT2-3 cases [11-13]. However, cautious and strict patient selection is crucial in this approach [14]. Higher risk of recurrence has limited the routine application of local excision alone in cT2 rectal cancer [15]. The incidence of local recurrence was 24% in T2 rectal cancer after local excision alone [15]. In addition, salvage treatment for failure after local excision is difficult and is often associated with treatment-related morbidity [16]. Under National Comprehensive Cancer Network (NCCN) guidelines, the standard treatment for cT2N0 rectal cancer is radical surgery, and adjuvant treatments are recommended according to pathological status [17]. For patients with cT2 distal rectal cancer whose sphincteric muscles cannot be preserved with abdominoperineal resection (APR), local excision after neoadjuvant CRT is an alternative approach to preserving the sphincter with equivalent oncologic outcomes [18,19]. A recent randomized clinical trial of local resection versus TME after neoadjuvant therapy showed equivalent disease-free survival (DFS) in patients with cT2N0, a diameter no larger than 3 cm, and histological grade G1-2 rectal cancer [20]. The probability of developing recurrence was 12% after local excision. Recurrence occurred only in low or non-responders to neoadjuvant CRT.
In this study, to elucidate the clinical implication of ypCR after neoadjuvant CRT in patients with cT2 distal rectal cancer, the Korean Radiation Oncology Group (KROG) conducted a multicenter retrospective study that examined the DFS according to ypT status.
Materials and MethodsSeventeen patients with cT2 primary rectal adenocarcinoma who were treated by neoadjuvant CRT and local excision between 2002 and 2009 were enrolled from four institutions. After approval by the KROG (KROG 12-06), the medical and radiotherapy (RT) records of the patients were reviewed retrospectively. The median age of patients was 63 years (range, 38 to 79 years). There were 11 (65%) men and six (35%) women. All tumors were located within 6 cm from the anal verge as measured by digital examination. For clinical staging, computed tomography (CT, n=15), magnetic resonance imaging (n=8), or transrectal ultrasound (n=11) was performed. Three patients were diagnosed by CT scan only. When positive lymph node involvement was defined as a lymph node ≥ 0.5 cm in the short-axis diameter, two patients had clinical involvement of a regional lymph node. The tumor characteristics at diagnosis are summarized in Table 1.
Patient refusal of radical surgery (n=16) and poor performance status (n=1) were the reasons for undergoing local excision following neoadjuvant CRT. Patients who chose local excision were fully informed about the tumor response to neoadjuvant CRT and the options of surgical modalities between radical resection and local excision. RT was delivered to the whole pelvis at a dose of 45 Gy in 25 fractions (n=8), 50.4 Gy in 28 fractions (n=5), or 44 Gy in 22 fractions (n=4) by 6-15 MV photon beams. The regimens of concurrent chemotherapy included 5-fluorouracil (5-FU; n=9, 53%), capecitabine (n=7, 41%), and S-1 plus irinotecan (n=1, 6%). There was no incidence of grade 3 or higher toxicity associated with neoadjuvant CRT. The median interval between completion of CRT and surgery was 53 days (range, 40 to 75 days). All patients underwent transanal tumor excision. Postoperative chemotherapy was delivered to eight (47%) patients, and six received four cycles of 5-FU.
The median follow-up duration calculated from the initiation of CRT was 75 months (range, 24 to 126 months). The primary endpoint of this study was DFS according to ypTclassification, which was divided into ypT0 or not. DFS was defined as the time from the initiation of the CRT to rectal cancer relapse or death, while second primary cancers were not included. DFS was estimated using the Kaplan-Meier test. DFS according to ypT-classification was compared using the log-rank test. The secondary endpoint was pattern of disease recurrence. SAS ver. 9.1.3 (SAS Institute Inc., Cary, NC) was used for statistical analysis. A p < 0.05 was chosen for statistical significance.
Results1. Surgical pathology after CRTThe primary tumor included ypT0 (n=10, 59%), ypT1 (n=6, 35%), and ypT2 (n=1, 6%). All of the ypT1-2 tumors were adenocarcinoma. Histologic grade was well-differentiated in one tumor, and moderately-differentiated in six tumors. Lymphovascular invasion, perineural invasion, and involvement of the resection margin were not observed. Resection margins were not involved in pathologically residual tumors, although the distances of resection margins were not described, except two patients. There was no pathologic involvement of regional lymph node (n=4). Among the seven patients with ypT1-2 disease, five received postoperative chemotherapy, while three of the 10 patients with ypT0 received adjuvant chemotherapy. The clinicopathologic characteristics according to ypT-classification are summarized in Table 2.
2. Survival rates according to tumor responseDuring the follow-up period, four patients (24%) experienced recurrence (Table 3). Locoregional and distance recurrences each occurred in two patients, respectively. Among the patients who achieved ypT0 after neoadjuvant CRT, one patient (10%) experienced regional recurrence after 14 months. Three (43%) out of the seven ypT1-2 patients experienced a recurrence. One patient had locoregional recurrence and two had distant metastasis. The 5-year DFS of all patients was 82% (Fig. 1A). DFS in patients with ypT0 tumors was higher than in patients with ypT1-2 tumors (90% vs. 69%) (Fig. 1B), however, the difference was not significant (p=0.1643). Lower DFS was observed in patients who received capecitabine as neoadjuvant CRT, compared with patients treated with 5-FU (54% vs. 100%, p=0.0298). Among the seven patients with tumors close to the anal verge (< 3 cm), three patients had disease relapse (p=0.1434) (Table 4). No significant difference in DFS was observed for other clinicopathologic characteristics (Table 4). None of the patients died during the follow-up period.
DiscussionypCR after neoadjuvant CRT is known to be associated with favorable long-term oncologic outcomes [4-6]. According to results of a pooled analysis, the ypCR rate is 16% [4]. Local control, distant metastasis-free survival, DFS, and overall survival were favored in patients with ypCR. The adjusted hazard ratio by Cox proportional hazards model was 0.41 (95% confidence interval [CI], 0.21 to 0.81), 0.49 (95% CI, 0.34 to 0.71), 0.54 (95% CI, 0.40 to 0.73), and 0.65 (95% CI, 0.47 to 0.89) for the outcomes, respectively. However, most studies included patients with cT3 rectal cancer who underwent radical surgery. Similar findings were observed in patients who underwent local excision following neoadjuvant CRT for cT2-3 rectal cancer [12]. The prevalence of ypT0 was 22% (53 of 237). After local excision, none of the patients with ypT0 experienced local recurrence, while 2%, 7%, and 21% of patients with ypT1, ypT2, and ypT3, respectively, experienced local recurrence. Although 34% of the patients had cT2 rectal cancer, the relationship between ypT-classification and clinical outcome in cT2 disease was not evaluated.
A recent randomized trial reported equivalent DFS to TME after local excision following neoadjuvant CRT [20]. The trial included patients with cT2N0, histologic grades 1-2 with tumors < 3 cm in diameter and within 6 cm of the anal verge, similar to the current study. After local resection, ypT0 was observed in 28% of patients, comparable to that of patients who received TME (26%). The probability of developing disease recurrence at the end of follow-up was 12% after local resection, and the corresponding overall survival rate was 72%. Although the duration of follow-up was shorter and the number of patients was smaller, the rate of ypT0 (59%) and overall survival rate were higher in the current study. In addition, the randomized trial did not evaluate the clinical implication of ypT0 after local resection. In the current study, ypCR after neoadjuvant CRT for cT2 rectal cancer showed an association with favorable outcomes in patients who underwent local excision. After neoadjuvant CRT, ypT0 showed an association with improved DFS compared with ypT1-2 in cT2 distal rectal cancer. The small number of patients limited the power of the study, and higher rate of ypT0 might be influenced by potential selection bias, which could be caused by patient’s choice of local excision after they were informed about the clinical tumor response and the option of avoidance of radical resection. However, local excision could be a feasible alternative to radical surgery in properly selected patients with ypCR after neoadjuvant CRT for cT2 distal rectal cancer, which is located within 6 cm of the anal verge.
Of particular interest, most cases of disease recurrence occurred in patients who had received capecitabine as neoadjuvant therapy, regardless of postoperative chemotherapy. However, no difference in the proportion of patients with ypCR after neoadjuvant CRT was observed between the two chemotherapeutic regimens (67% vs. 57%, p=1). Compared to 5-FU, oral capecitabine demonstrated comparable or improved tumor response, local control, DFS, and overall survival in patients who received neoadjuvant CRT for locally advanced rectal cancer [21,22]. In addition to the convenience of oral administration and lesser toxicities, capecitabine has several advantages, including preferential activation in tumor tissue and a synergistic effect with X-ray [23]. Oral capecitabine is currently a valid option in neoadjuvant CRT for rectal cancer, as investigated in previous studies [21,22]. However, most previous studies regarding local excision following neoadjuvant CRT used 5-FU as a chemotherapeutic regimen [12,13,20]. Efficacy of oral capecitabine in this setting has not been well established. Whether capecitabine is still as effective as 5-FU in patients receiving neoadjuvant CRT followed by local excision should be clarified. However, due to the small sample size, different DFS according to neoadjuvant chemotherapeutic regimen in the current study is not sufficient to answer this question.
In the current study, most of the enrolled patients refused radical surgery such as APR because the tumors were located in the distal rectum. Although a meta-analysis showed no difference in general quality of life (QoL) following APR or anterior resection [24], a sphincter-preserving procedure is preferred in terms of QoL for patients [25]. In this regard, local excision combined with neoadjuvant CRT could be the first treatment option in patients with cT2 distal rectal cancer for whom the anal sphincter cannot be preserved. After local excision, patients with residual tumors in the specimen should consider radical surgery. Because local excision alone is associated with increased risk of disease recurrence, it should be applied cautiously to patients with residual disease after neoadjuvant CRT.
Fig. 1.(A) Disease-free survival of all patients. (B) Disease-free survival according to ypT-classification. 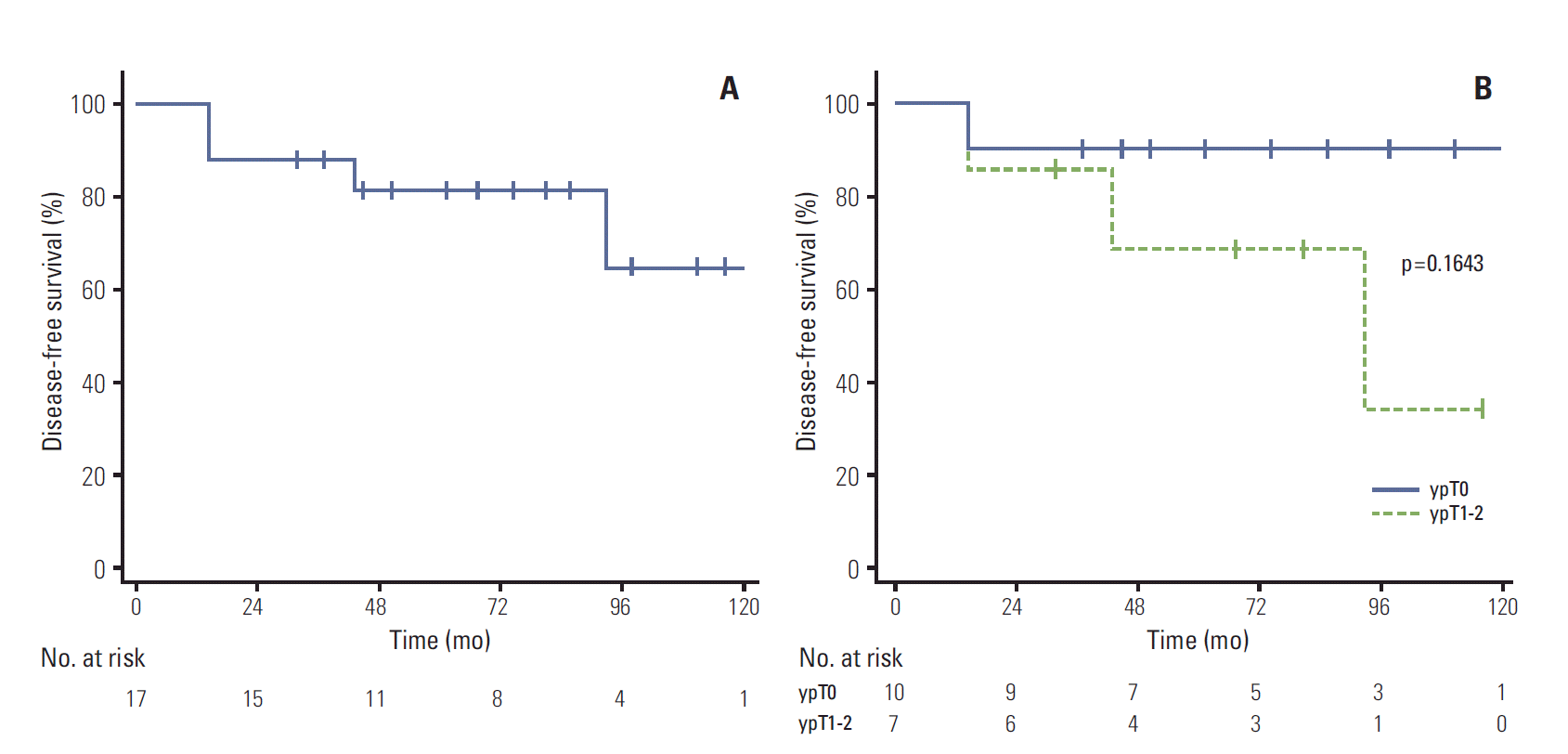
Table 1.Tumor characteristics at diagnosis Table 2.Clinicopathologic characteristics according to ypT-classification Table 3.Characteristics of patients who experienced disease recurrence Table 4.Disease-free survival according to clinicopathologic characteristics
References1. Sauer R, Becker H, Hohenberger W, Rodel C, Wittekind C, Fietkau R, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004;351:1731–40.
2. van Gijn W, Marijnen CA, Nagtegaal ID, Kranenbarg EM, Putter H, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol. 2011;12:575–82.
3. Sanghera P, Wong DW, McConkey CC, Geh JI, Hartley A. Chemoradiotherapy for rectal cancer: an updated analysis of factors affecting pathological response. Clin Oncol (R Coll Radiol). 2008;20:176–83.
4. Maas M, Nelemans PJ, Valentini V, Das P, Rodel C, Kuo LJ, et al. Long-term outcome in patients with a pathological complete response after chemoradiation for rectal cancer: a pooled analysis of individual patient data. Lancet Oncol. 2010;11:835–44.
5. Belluco C, De Paoli A, Canzonieri V, Sigon R, Fornasarig M, Buonadonna A, et al. Long-term outcome of patients with complete pathologic response after neoadjuvant chemoradiation for cT3 rectal cancer: implications for local excision surgical strategies. Ann Surg Oncol. 2011;18:3686–93.
6. de Campos-Lobato LF, Stocchi L, da Luz Moreira A, Geisler D, Dietz DW, Lavery IC, et al. Pathologic complete response after neoadjuvant treatment for rectal cancer decreases distant recurrence and could eradicate local recurrence. Ann Surg Oncol. 2011;18:1590–8.
7. Lambregts DM, Maas M, Bakers FC, Cappendijk VC, Lammering G, Beets GL, et al. Long-term follow-up features on rectal MRI during a wait-and-see approach after a clinical complete response in patients with rectal cancer treated with chemoradiotherapy. Dis Colon Rectum. 2011;54:1521–8.
8. Maas M, Beets-Tan RG, Lambregts DM, Lammering G, Nelemans PJ, Engelen SM, et al. Wait-and-see policy for clinical complete responders after chemoradiation for rectal cancer. J Clin Oncol. 2011;29:4633–40.
9. Chessin DB, Enker W, Cohen AM, Paty PB, Weiser MR, Saltz L, et al. Complications after preoperative combined modality therapy and radical resection of locally advanced rectal cancer: a 14-year experience from a specialty service. J Am Coll Surg. 2005;200:876–82.
10. Hendren SK, O'Connor BI, Liu M, Asano T, Cohen Z, Swallow CJ, et al. Prevalence of male and female sexual dysfunction is high following surgery for rectal cancer. Ann Surg. 2005;242:212–23.
11. Bonnen M, Crane C, Vauthey JN, Skibber J, Delclos ME, Rodriguez-Bigas M, et al. Long-term results using local excision after preoperative chemoradiation among selected T3 rectal cancer patients. Int J Radiat Oncol Biol Phys. 2004;60:1098–105.
12. Borschitz T, Wachtlin D, Mohler M, Schmidberger H, Junginger T. Neoadjuvant chemoradiation and local excision for T2-3 rectal cancer. Ann Surg Oncol. 2008;15:712–20.
13. Nair RM, Siegel EM, Chen DT, Fulp WJ, Yeatman TJ, Malafa MP, et al. Long-term results of transanal excision after neoadjuvant chemoradiation for T2 and T3 adenocarcinomas of the rectum. J Gastrointest Surg. 2008;12:1797–805.
14. Perez RO, Habr-Gama A, Lynn PB, Sao Juliao GP, Bianchi R, Proscurshim I, et al. Transanal endoscopic microsurgery for residual rectal cancer (ypT0-2) following neoadjuvant chemoradiation therapy: another word of caution. Dis Colon Rectum. 2013;56:6–13.
15. Gopaul D, Belliveau P, Vuong T, Trudel J, Vasilevsky CA, Corns R, et al. Outcome of local excision of rectal carcinoma. Dis Colon Rectum. 2004;47:1780–8.
16. You YN, Roses RE, Chang GJ, Rodriguez-Bigas MA, Feig BW, Slack R, et al. Multimodality salvage of recurrent disease after local excision for rectal cancer. Dis Colon Rectum. 2012;55:1213–9.
17. National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: rectal cancer. v.4, 2013 [Internet]. Fort Washington: National Comprehensive Cancer Network; 2013. [cited 2013 Apr 13]. Available from: http://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf.
18. Rengan R, Paty P, Wong WD, Guillem J, Weiser M, Temple L, et al. Distal cT2N0 rectal cancer: is there an alternative to abdominoperineal resection? J Clin Oncol. 2005;23:4905–12.
19. Hingorani M, Hartley JE, Greenman J, Macfie J. Avoiding radical surgery after pre-operative chemoradiotherapy: a possible therapeutic option in rectal cancer? Acta Oncol. 2012;51:275–84.
20. Lezoche E, Baldarelli M, Lezoche G, Paganini AM, Gesuita R, Guerrieri M. Randomized clinical trial of endoluminal locoregional resection versus laparoscopic total mesorectal excision for T2 rectal cancer after neoadjuvant therapy. Br J Surg. 2012;99:1211–8.
21. Chan AK, Wong AO, Jenken DA. Preoperative capecitabine and pelvic radiation in locally advanced rectal cancer--is it equivalent to 5-FU infusion plus leucovorin and radiotherapy? Int J Radiat Oncol Biol Phys. 2010;76:1413–9.
22. Hofheinz RD, Wenz F, Post S, Matzdorff A, Laechelt S, Hartmann JT, et al. Chemoradiotherapy with capecitabine versus fluorouracil for locally advanced rectal cancer: a randomised, multicentre, non-inferiority, phase 3 trial. Lancet Oncol. 2012;13:579–88.
23. Sawada N, Ishikawa T, Sekiguchi F, Tanaka Y, Ishitsuka H. X-ray irradiation induces thymidine phosphorylase and enhances the efficacy of capecitabine (Xeloda) in human cancer xenografts. Clin Cancer Res. 1999;5:2948–53.
|
|