INTRODUCTION
Numerous epidemiological studies have demonstrated that non-steroidal anti-inflammatory drugs (NSAIDs) can reduce the risk of several malignancies, most notably colon cancer (1). The major target of NSAIDs is cyclooxygenase (COX), a rate-limiting enzyme in the biosynthesis of prostaglandin (PG) and related eicosanoids. COX-2 expression has been observed in many tumor types, including lung, bladder, head and neck, gastric and breast cancers, in addition to colon cancer (2~4).
The association between enhanced expression of COX-2 and breast cancer was first suggested by reports of elevated PG levels in breast cancer tissues (5). Using a murine model of human breast cancer, higher PGE2 levels were observed in malignant mammary tissue than in normal and premalignant glands, which was positively correlated with increased metastatic potential (6). Various epidemiological studies have shown that the use of NSAIDs can reduce the risk of breast cancer (7). Indeed, dietary administration of celecoxib, a selective COX-2 inhibitor, produced striking reductions in the incidence, multiplicity and volume of mammary tumors, relative to the control group, in an animal model of breast cancer (8).
The relationship between COX-2 and carcinogenesis may involve several pathways, including conversion of procarcinogens into active carcinogens, inhibition of apoptosis, promotion of angiogenesis and increased tumor cell invasiveness (9~11). Recent evidence also supports a link between the COX-2 expression and mammary carcinogenesis, which may be partially dependent on the induction of aromatase by PGE2 (12). Thus, selective COX-2 inhibitors offer considerable promise in the prevention and treatment of human breast cancer. Although COX-2 expression may be involved in breast carcinogenesis, data on COX-2 expression rate in breast tumors has been conflicting, and there have been few studies on the possible role of COX-2 expression as a prognostic indicator in breast cancer patients that have undergone a curative surgical resection.
Therefore, the COX-2 expression in patients with operable breast cancer was assayed by immunohistochemistry. Moreover, the association between COX-2 expression and clinicopathological characteristics, including c-erb-B2 overexpression, angiogenesis, as assayed by the microvessel count (MVC), and the prognosis for disease-free survival in these patients were determined.
MATERIALS AND METHODS
1) Patients and tissues
Tissue samples were obtained from the surgically removed specimens of 205 patients with primary invasive breast cancer, who underwent curative surgical resection between March 1991 and June 1997 at the Asan Medical Center, and had been followed-up for at least 5 years. Pathological data, including tumor size, axillary nodal involvement status, histologic and nuclear grades, and estrogen and progesterone receptor expressions, were collected from previous pathology reports. Clinical data were also obtained from the hospital medical records. Of the 205 patients, 124 had no axillary node involvement, whereas 81 patients had, and of these 81 patients, 25, 19 and 38 had involvement of 1 to 3, 4 to 9 and more than 9 nodes, respectively. All patients with involved axillary nodes and 54.5% (67/124) of patients without axillary nodes received adjuvant chemotherapy.
2) Tissue microarray method
Tissue microarrays were constructed as described previously (13). Briefly, a total of 205 formalin-fixed, paraffin-embedded tissue blocks, containing breast carcinoma specimens, were retrieved from the archives of the Institute of Pathology at Asan Medical Center. Areas of invasive carcinoma were identified on corresponding hematoxylin and eosin (H & E)-stained slides. Tissue cylinders with a diameter of 0.6-mm were punched from 2 different tumor areas of each "donor" tissue block and brought into a "recipient" paraffin block (45×20 mm) using a Tissue Microarrayer (Beecher Instruments, Silver Springs, MD). Then, 2 recipient paraffin blocks, each containing 205 cores, were made. After cutting the recipient paraffin block and transferring with adhesive tape to coated slides, the slides were dipped in a layer of paraffin to prevent oxidation. Six replicate tissue microarrays were constructed, each containing a different tissue block from each of the 205 tumors. The tissue microarray sections were immunohistochemically stained for COX-2, CD34 and c-erb-B2.
3) Immunohistochemical staining
The tissue microarray slides made from paraffin-embedded tissue blocks were deparaffinized, and rehydrated for 5 minutes. After microwave pretreatment in citrate buffer (pH 6.0) for antigen retrieval, the slides were immersed in 0.3% hydrogen peroxide for 20 min to block the endogenous peroxidase activity. The slides were washed and incubated overnight at 4℃ with mouse monoclonal antibody against COX-2 (Cayman Chemical, Ann Arbor, MI) at a dilution of 1 : 100, mouse monoclonal antibody against CD34 (ImmunoTech, Cedex, France) at a dilution of 1 : 2500 and rabbit anti-human polyclonal antibody against c-erb-B2 (DAKO, LSAB+ kit; DAKO Corp., Carpinteria, CA) at a dilution of 1 : 500. After a second incubation with a biotinylated anti-goat antibody, the slides were incubated with peroxidase-conjugated streptavidin (DAKO). The reaction products were visualized by immersing the slides in diaminobenzidine tetrachloride and counterstaining with Mayer's hematoxylin.
4) Evaluation of staining
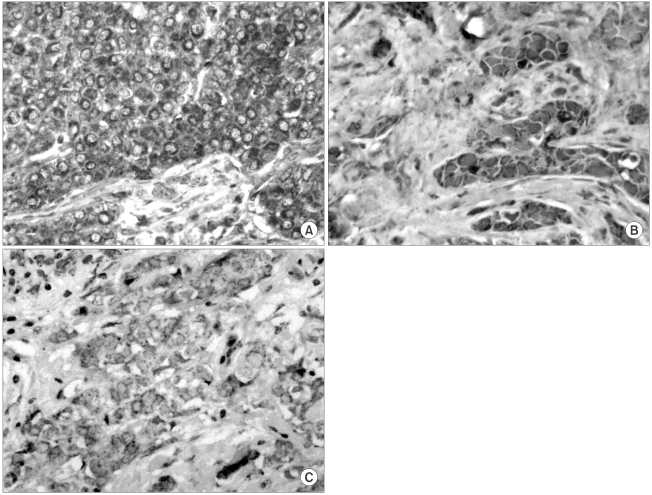
COX-2 immunohistochemical staining was scored independently by two investigators blinded to the patient information. Positive staining of smooth muscle cells provided an internal positive control for COX-2 staining. Positive tumor cells were cytoplasmically stained, and the percentage of positive tumor cells in each sample semi-quantitatively graded (quantity score) as: 0 (<10%), 1 (10~40%), 2 (40~70%) or 3 (≥70%). The intensity of immunostaining was determined on a scale of 0 to 3, with 0 for negative, 1 for weak, 2 for moderate and 3 for strong. Both a quantity score and immunostaining intensity of 2 or more (>40% staining with moderate or strong intensity) was defined as positive COX-2 expression (14).
As a parameter of angiogenesis, microvessels were counted using the same two slides made by the tissue microarray method, at a ×200 magnification. The higher number of microvessels on the 2 slides was defined as the MVC values (15), which were expressed as the mean±SD.
Immunostaining for c-erb-B2 was scored on a scale of 0 to 3, according to the staining of the cytoplasmic membrane, with 0 for staining of ≤10%, 1 for staining of >10% with a weak staining intensity, 2 for staining of >10% with moderate staining intensity and 3 for staining of >10% with strong staining intensity of the tumor cells. A staining score of 2 or 3 was defined as c-erb-B2 overexpression (16).
5) Statistical analysis
The relationships between COX-2 expression and other clinicopathological parameters, such as estrogen and progesterone receptors, axillary lymph nodal status and stage, were compared using χ2 or Fisher's exact probability tests where appropriate. The strength of association between the COX-2 score and MVC was assessed by Student's t-tests. The Kaplan-Meier method was used to estimate the survival, and differences were analyzed using log-rank tests. Disease-free survival (DFS) was defined as the time from the day of the operation to a documented recurrence, or death from any other cause. The Cox proportional hazards model was used for a multivariate analysis of the prognostic factors. The SPSS program (version 11.0) was used, and a p<0.05 was defined as being statistically significant.
RESULTS
1) COX-2 expression in human breast carcinoma
Immunoreactivity of COX-2 protein showed diffuse staining in the cytoplasm of tumor cells, but COX-2 expression was not observed in normal breast tissue (Fig. 1). The stroma was either negative or weakly positive. Immunoreactivity of COX-2 was evaluated in 205 breast cancer tissues, of which 38 (18.5%) were negative, 49 (23.9%) weakly positive, 84 (41.0%) moderately positive and 34 (16.6%) strongly positive. Thus, grade 2 or 3 COX-2 positive tumor cells were observed in 118/205 (57.6%) of the tumors samples assayed.
2) Association of COX-2 expression with clinicopathological parameters
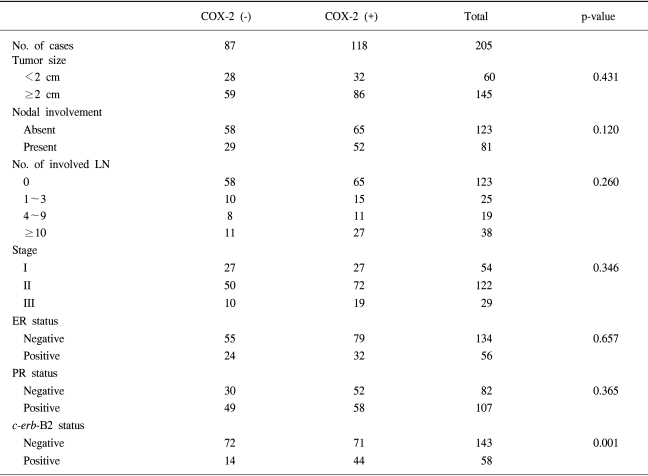
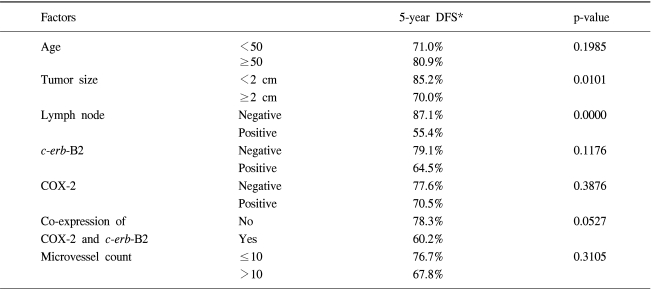
The association of COX-2 expression with various clinicopathological parameters of breast cancer patients is shown in Table 1. No statistically significant relationships were observed between COX-2 expression and tumor size (p-value 0.431), axillary nodal involvement (p-value 0.120), estrogen receptor status (p-value 0.657) or progesterone receptor status (p-value 0.365).
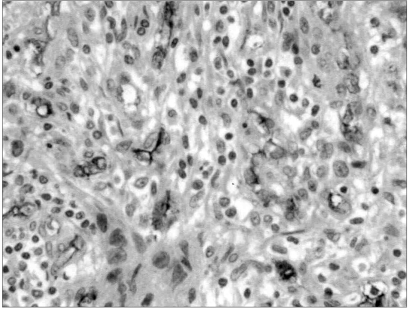
The mean MVC value was 10.3±8.2, ranging from 0 to 45. The immunohistochemical staining for CD34 in the microvessels of the breast cancer tissue are shown in Fig. 2. The MVC was significantly higher in specimens with than without COX-2 expression (13.3±8.0 vs. 6.6±7.0) (p-value 0.050).
Overexpression of c-erb-B2 was detected in 58/205 (28.3%) breast cancer tissue specimens. The COX-2 expression rate was significantly higher in c-erb-B2 positive than in c-erb-B2 negative breast cancers (44/58, 75.9% vs. 71/143, 49.7%) (p-value 0.001).
3) Association of COX-2 expression with disease-free survival
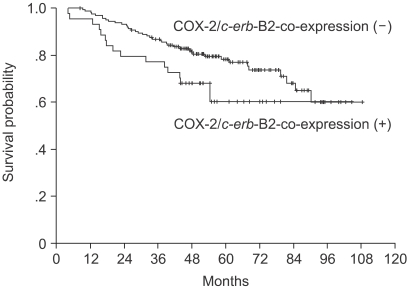
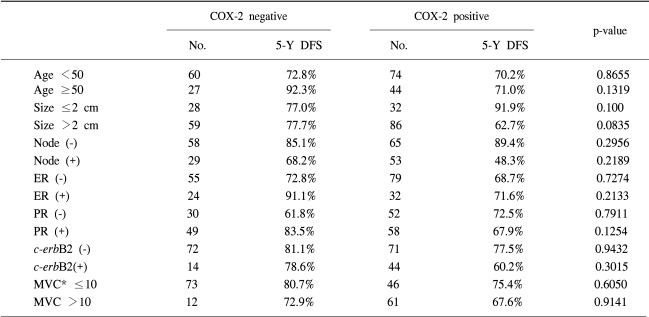
A univariate analysis showed statistically significant differences in the 5-year DFS rate according to the axillary nodal status (negative vs. positive) and tumor size (<2 cm vs. ≥2 cm) (Table 2). The 5 year DFS rate was 77.6% in the COX-2 negative group and 70.5% in the COX-2 positive group, showing that COX-2 expression alone was not a statistically significant prognostic factor affecting the 5-year DFS rate (p-value 0.3876). Both the MVC value and c-erb-B2 expression were not statistically significant prognostic factors of the 5-year DFS rate. However, patients co-expressing both COX-2 and c-erb-B2 showed a tendency towards poorer 5-year DFS rates than those that did not (60.2% vs. 78.3%, p-value 0.0527) (Fig. 3). The five-year DFS rates for several groups of patients, according to the COX-2 expression, are summarized in Table 3.
After the multivariate analysis with the Cox proportional hazards model, only axillary nodal involvement and co-expression of both COX-2 and c-erb-B2 were found to be independent prognostic factors of the 5-year DFS rate (Table 4).
DISCUSSION
The COX-2 was found to be expressed in 57.6% (118/205) of breast cancer tissues, and was localized exclusively to the neoplastic cells. COX-2 is not abundantly overexpressed in the majority of human breast cancers, with conflicting data regarding the frequency of its expression. Although one study showed COX-2 expression in 13/13 human breast tumors by reverse transcriptase-polymerase chain reaction (RT-PCR) (4), another reported COX-2 protein in only 2 of 44 samples by Western blotting (17). COX-2 was expressed in 43% of 57 invasive breast carcinomas (18), 36% of 221 breast carcinoma samples (19) and 37.4% of 1576 invasive breast cancers in a recent large, multi-centered study (14). A possible explanation for these different COX-2 expression rates is that this protein may be expressed predominantly in certain subsets of human breast cancers. For example, it was recently reported that COX-2 was expressed in 14/15 c-erb-B2-overexpressing breast cancers, but in only 4 of 14 c-erb-B2-negative breast cancers (20). Our study also indicated that the COX-2 expression rate was significantly higher in c-erb-B2 positive than in c-erb-B2 negative tumors (75.9 vs. 49.7%, p-value 0.001).
Both the COX-2 and c-erb-B2 pathways have been shown to modulate the growth of epithelial cancers. For example, activation of c-erb-B2 has been reported to stimulate the proliferation of colon cancer cells via the induction of COX-2 (21). In addition, c-erb-B2 levels were reduced in the rectal mucosa of patients treated with low doses of the COX-2 inhibitor, sulindac (22). Although the underlying mechanism is still unclear, COX-2 expression may be a "downstream target" for c-erb-B2-mediated tumorigenesis (20). Since COX-2 inhibitors may be useful in cancer prevention and treatment, inhibition of one of these pathways may increase the effects of inhibitors of the other pathway. For example, combined treatment with COX-2 and c-erb-B2 inhibitors more effectively reduced the growth of colorectal carcinoma cells in vitro than either agent alone (23), and the combination of a COX-2 inhibitor and an HER-2/neu tyrosine kinase inhibitor had an enhanced antiproliferative effect on the growth of human breast cancer cell lines (24).
COX-2 has been also reported to induce angiogenesis, which may be essential for tumor growth, and its involvement in angiogenesis has been reported in other tumor types, including colorectal and gastric cancers (10). We have shown here that the microvessel count was significantly higher in breast cancer tissues expressing COX-2 than in those that did not (p-value 0.050). Although little is known about its mechanism, COX-2 positive tumors are thought to produce angiogenic factors, and COX-2 inhibitors have been shown to suppress angiogenesis and tumor growth of the COX-2 overexpressing cancer xenografts by inhibiting the expression of these angiogenic factors (11). Although these angiogenic factors were not assayed for in this study, our results suggest that COX-2 may play a role in the angiogenesis of breast cancer.
Human breast cancer cell lines expressing COX-2 were observed to have more aggressive and metastatic phenotypes than those not expressing COX-2 (7). In addition, COX-2 expression has been associated with the aggressive traits of breast cancers, including larger tumor size, positive lymph node status, poor differentiation, negative hormone receptor status, c-erb-B2 overexpression and decreased survival (14,18). In our study, however, COX-2 expression in breast cancers did not correlate with tumor size, number of involved axillary lymph nodes, tumor histology, or estrogen and progesterone receptor status. Moreover, a recent study reported a lack of correlation between COX-2 expression and tumor size, grade, lymph node status and vascular invasion (25). These findings raise the possibility that COX-2 expression may be more closely involved in the initial development of breast cancer than in its progression, but further work is needed to determine whether COX-2 expression is involved in tumor growth and/or metastasis.
In the current study, COX-2 expression alone was unable to predict prognosis in terms of the DFS. However, breast cancer patients with tumors co-expressing both COX-2 and c-erb-B2 had a poorer 5-year DFS than those that did not (60.2% vs. 78.3%, p-value 0.0527). As mentioned above, there is growing evidence that the COX-2 and c-erb-B2 pathways may be interconnected. Although c-erb-B2 overexpression may be associated with a poor prognosis in breast cancer patients, these findings are still not conclusive. Our data suggest that COX-2 co-expression may have a role as an additional marker of a poor prognosis in patients with c-erb-B2 positive breast cancers. It would be of interest to determine whether combination therapy with a COX-2 inhibitor and trastuzumab, a monoclonal antibody against c-erb-B2, has any clinical relevance, and a phase II study is currently ongoing to evaluate this question.