Introduction
Radiotherapy is a commonly used cancer treatment that entails lethal doses of radiation against cancer cells [1]. However, exposure of normal tissue to radiation can cause both acute and chronic toxicity, including dermatitis, oral mucositis, altered taste, pain, dry mouth, decreased appetite, and even ulceration [2].
Oral mucositis is one of the most common complications of cancer therapy, chemotherapy, and radiation therapy. In patients with granulocytopenia, it often leads to systemic infections and nutritional deficiencies due to the intake of a restricted diet [3].
Despite technological advances, a successful method for the prevention of radiation-induced oral mucositis and normal cell toxicity has yet to be developed [4]. Although many recent studies have shown the potential of radiation in protection against chemicals and small molecules, most of them have yet to reach the preclinical stage due to their toxicity and side effects, and the unknown mechanisms involved in radiation protection. Radiation-induced oral mucositis is caused by a variety of mechanisms, including but not limited to the release of free radicals, modified proteins, and proinflammatory cytokines, including interleukin-1β, prostaglandins, and tumor necrosis factor by irradiated epithelial, endothelial, and connective tissue cells in the buccal mucosa [5].
Previous studies have reported an increase in intracellular reactive oxygen species (ROS) levels during radiation-induced oral mucositis. Scavengers such as vitamin E, amifostine, and N-acetylcysteine (NAC) are known to inhibit oral mucositis, indicating a role for ROS in radiation-induced oral mucositis [6].
Although the radioprotective effects of scavengers are unknown, ROS scavenger supplements are seen to partially protect against sublethal damage induced by ionizing radiations. Therefore, we elucidated the relationship between autophagy and the antioxidant signal transduction mechanism.
NAC is a free radical scavenging antioxidant [7]. Several studies have reported on its efficacy in reducing inflammation of the mucous membranes, improving the elimination and excretion of sputum in inflammatory diseases of the respiratory system, and inhibiting the secretion of cytokines [8].
Nuclear factor erythroid 2-related factor 2 (NRF2), an antioxidant, is regulated by upstream signal transduction factors such as mitogen-activated protein kinases, extracellular signal-regulated kinase, c-Jun N-terminal kinase, and phosphatidylinositol 3-kinase [9]. Under oxidative stress, NRF2 is not degraded and translocates to the nucleus where it binds to the promoter regions of antioxidant genes, such as glutathione transferases, UDP-glucuronosyltransferases, γ-glutamylcysteine synthetase, glutathione peroxidase, heme oxygenase-1, catalase, and NAD(P)H:quinone oxidoreductase-1, to upregulate their transcription [10].
The mechanism for the radioprotective effect of NRF2 is unknown. However, it is known to depend on radiation-induced ROS generation that leads to cell and DNA damage. Interestingly, recent reports have shown that the NRF2 pathway correlates with autophagic signaling and contributes to antioxidant-mediated protection of the cells by eliminating oxidatively damaged organelles and proteins [11].
In this study, we investigated the protective effect of NAC against radiation-induced oral mucositis in animal studies and keratinocytes. The associated signaling mechanisms, specifically those involving the autophagic signaling pathway, were also studied.
Materials and Methods
1. Animal study
Six-week-old female Sprague-Dawley rats were purchased from Orient Bio Co. Ltd. (Seongnam, Korea).
The animals were randomly assigned to either an irradiation group (n=20) or a non-irradiation group (n=20) for 3 weeks. Each group was divided into two groups. One group was treated with NAC (Mucomyst, Boryung Pharm, Ansan, Korea) (n=10), and the other group was treated with saline (n=10).
A single 30 Gy dose was delivered by opposing photon beams at a rate of 2 Gy/min bilaterally at a distance of 100 cm from the source to the axis using the 6 MV LINAC (21EX, Varian Medical Systems, Palo Alto, CA). Radiation dose and evaluation were previously described [12].
Rats were treated with NAC (Mucomyst, Boryung Pharm) from the day after irradiation. Rats were placed in an acryl box (30×20×20 cm), and a nebulizer was used to administer NAC (air flow, 10.01 L/min) for 5 minutes and stabilized for 5 minutes. The control groups were administered saline. Treatment was conducted twice every day for 3 weeks (9 am and 6 pm).
2. Cell culture and radiation conditions
The human immortalized keratinocytes, HaCaT cells, were obtained from the American Type Culture Collection (ATCC, Manassas, VA). HaCaT cells were maintained in high glucose Dulbecco’s modified Eagle’s medium (Gibco, Grand Island, NY), supplemented with 10% fetal bovine serum and 100 U/mL penicillin-streptomycin (Gibco, Paisley, PA) at 37°C with 5% CO2 under humidified conditions.
3. Terminal deoxynucleotidyl transferase dUTP nick end labeling assay
Apoptotic cells in the buccal mucosa were assessed by DNA fragmentation within cells using the In Situ Cell Death Detection Kit, POD (Roche Molecular Biochemicals, Indianapolis, IN), according to the manufacturer’s protocol. Nuclei were counterstained with Hoechst 33342.
4. Cell cycle analysis and measurement of ROS production
HaCaT cells were harvested by trypsinization and washed with phosphate buffered saline (PBS). Cold 70% ethanol was slowly added to the cells while vortexing, and were fixed overnight at –20°C. The cells were washed with PBS twice, centrifuged at 1,300 rpm for 3 minutes, and resuspended in 200 μL PBS. Subsequently, they were incubated with 300 μg/mL RNase (Intron Biotechnology, Seongnam, Korea) for 30 minutes at 37°C, and 500 μL propidium iodide (10 μg/mL, Invitrogen, Carlsbad, CA) for another 30 minutes at 4°C in a dark room. Cell cycle distribution was calculated in 10,000 cells using a BD FACS Aria III instrument (BD Biosciences, Bedford, MA).
The cellular ROS production was measured by treating HaCaT cells with 10 μM hydroethidine (Molecular Probes, Eugene, OR) for 30 minutes at 37°C. Fluorescence-stained cells were then analyzed with BD FACS Aria III (BD Biosciences).
5. Western blot analysis
Cells were lysed in RIPA buffer (Sigma-Aldrich, St. Louis, MO) containing 50mM Tris (pH 8.0), complete EDTA-free protease inhibitor, and PhoSTOP (Roche Molecular Biochemicals, Basel, Switzerland), as described previously. The cell lysates were mixed with 5× sodium dodecyl sulfate sample buffer and run on a 10%-12% sodium dodecyl sulfate polyacrylamide gel electrophoresis gel, followed by electrophoretic transfer to PVDF membrane. Targeted proteins were immunoblotted with specific antibodies. The following primary antibodies were used: p21, p27, phospho-p53 (Ser15), p53, cyclin B1, γH2AX, mammalian target of rapamycin (mTOR), phospho-mTOR, ATG3, ATG5, P62, LC3B, and glyceraldehyde 3-phosphate dehydrogenase (1:1,000, Cell Signaling Technology, Danvers, MA). Secondary antibodies (1:4,000, anti-rabbit IgG or anti-mouse IgG) were purchased from Cell Signaling Technology.
6. Cell proliferation assay (BrdU assay)
Cell proliferation was measured using a BrdU assay kit (Roche Diagnostics, Penzberg, Germany), according to the manufacturer’s protocol (BD Biosciences) as described previously [12]. Absorbance was measured at a wavelength of 370 nm using an enzyme-linked immunosorbent assay reader (Bio-Tek, Winooski, VT). The rate of cell proliferation was expressed as a percentage of untreated cells.
7. Transmission electron microscopy
The cells were fixed in 2% glutaraldehyde after treatment with vehicle or NAC (10 mM, Sigma-Aldrich) only, radiation alone (20 Gy), or radiation (20 Gy) plus NAC (10 mM), as previously described [14]. All thin sections were observed with an electron microscope (JEM-1011, Jeol, Tokyo, Japan) at an acceleration voltage of 80 kV, and the images were analyzed with the Camera-Megaview III Soft imaging system.
8. Quantitative real-time polymerase chain reaction
Total RNAs from HaCaT cells treated with vehicle or NAC (10 mM, Sigma-Aldrich) only, radiation only (20 Gy), or radiation (20 Gy) plus NAC were isolated using TRIzol reagent (Gibco-BRL, Grand Island, NY). The cDNA synthesis was performed as described previously [15]. We quantified the targeted gene expression via one-step real-time PCR using Step One Plus TM (Applied Biosystems, Foster City, CA). All primers were purchased from Qiagen (Hilden, Germany) and resuspended in 100 µM stock solutions in TE buffer (pH 8.0, Teknova, Hollister, CA).
9. Immunohistochemistry
Immunohistochemistry was performed using paraffin-embedded tissue sections collected on poly L-lysine–coated slides. The specimens were briefly incubated in a blocking solution with anti-LC3B (1:200), NRF2 (1:200) antibody overnight at 4°C. The sections were thoroughly rinsed in PBS and incubated for 2 hours at room temperature with SPlink HRP Detection Kit (GBI Labs, Mukilteo, WA). Immunolabeling was performed after three washes in PBS and stained with Liquid DAB+ Substrate Kit (GBI Labs).
10. Immunocytochemistry
HaCaT cells were cultured on microscope coverslips (Thermo Fisher Scientific, Rochester, NY) and treated with vehicle or NAC (10 mM, Sigma-Aldrich) only, radiation only (20 Gy), or radiation (20 Gy) plus NAC. After 24 hours, the slides were washed with PBS, fixed for 20 minutes in 3.7% formaldehyde, and rehydrated in PBS. Immunocytochemistry assays were performed as described previously [16]. The slides were washed and mounted with Vectashield (Vector Laboratories, Inc., Burlingame, CA). Cells were imaged using a fluorescence microscope (EVOS, Seattle, WA) [17].
11. Isolation of nuclear and cellular extracts
Nuclear and cellular extracts were isolated from cells treated with vehicle or NAC (10 mM, Sigma-Aldrich) only, radiation only (20 Gy), or radiation (20 Gy) plus NAC (10 mM) for 24 hours using the NE-PER Nuclear and Cytoplasmic Extraction Reagent kit (Pierce Biotechnology, Rockford, IL), following the manufacturer’s protocol.
12. Statistical analysis
Data from at least three independent experiments were expressed as mean±SD. Comparisons of the means of different groups were performed using one-way analysis of variance (ANOVA). We conducted one-way ANOVA based on the Mann-Whitney U test using SPSS ver. 20.0 statistical software (IBM Corp., Armonk, NY). p-values < 0.05 were considered statistically significant.
Results
1. Pre-treatment with NAC protects irradiated HaCaT cells against DNA damage
Radiation induces HaCaT cell death by inducing DNA damage [18]. Therefore, DNA damage markers were measured, and cell cycle analysis was performed to determine whether 10 mM NAC pre-treatment could prevent DNA damage.
We analyzed protein-related DNA damage for p-ATM, ATM, p21, p53, cyclin B1, and γH2AX to determine whether NAC could block the ATM pathway and DNA damage mediating the cell cycle arrest observed. Increased protein levels or phosphorylation of p-ATM, p-p53 (Ser15), p21, cyclin B1, and γH2AX after irradiation and treatment with NAC reduced the phosphorylation of p-ATM, p-p53 (Ser15), and the protein levels of p21, cyclin B1, and γH2AX, which were increased by radiation treatment (Fig. 1A).
Interestingly, the length of the S phase of the cell cycle increased during radiation treatment and decreased after NAC treatment (Fig. 1B).
As shown in Fig. 1C, the mRNA levels of cyclin A and B were significantly lower in the NAC-treated group than in the radiation only group. These results were consistent with those of the western blot analysis.
In addition, to analyze DNA damage related to cell cycle arrest, we evaluated the expression of γH2AX, which plays an essential role in the cellular DNA damage response. NAC significantly inhibited the expression of γH2AX induced by irradiation (Fig. 1D). These results suggested that the protective mechanism of action of NAC and decreased DNA damage correlate with reduced phosphorylation of ATM and p53.
2. NAC inhibits radiation-induced intracellular ROS production via regulation of NRF2 expression in HaCaT cells
Radiation has been shown to increase ROS-induced oxidative stress in cells [19]. Therefore, we investigated ROS levels after irradiation to identify the mechanism by which NAC inhibits ROS production. First, the generation of ROS was quantified using dihydroethidium (DHE). As shown in Fig. 2A, the significant increase in ROS generation induced by irradiation was inhibited significantly by NAC. To confirm these results, we used DHE fluorescence staining. As shown in Fig. 2B, the radiation-induced increase in fluorescence intensity was inhibited significantly by NAC.
To elucidate the mechanism underlying the activity of radiation and NAC, we evaluated the effects of radiation and NAC-induced changes on gene expression. Radiation has been reported to increase NRF2 expression in human lymphocytes. Given the role of NRF2-Keap1 signaling in stress response, the NRF2 signaling mechanism protects cells by activating antioxidant-related genes [20].
To identify the relationship between radiation-induced ROS and NRF2 expression, HaCaT cells were irradiated, and the protein levels of NRF2 were measured by western blotting. A representative western blot is shown in Fig. 2C. The results confirmed the increased expression of NRF2 after radiation treatment and showed that NAC treatment reduced the expression of NRF2.
As shown in Fig. 2D, NAC treatment attenuated the protein level of NRF2 increased by radiation treatment in both the nuclear and cytoplasmic fractions.
To confirm our findings regarding NRF2 expression, the nuclear translocation of each molecule was analyzed using immunocytochemistry. These results were consistent with those of western blot analysis.
3. NAC inhibits radiation-induced autophagy in HaCaT cells
Recent reports have shown that autophagy is closely related to radiation [21]. Therefore, to identify the relationship between radiation-induced ROS generation and DNA damage and the inhibitory effect of NAC, autophagy-related proteins were quantified by western blotting. As shown in Fig. 3A, radiation treatment increased the expression of autophagy-related proteins such as Beclin-1, LC3B, and p62. However, NAC treatment reduced the expression of Beclin-1, LC3B, and p62, which had increased after radiation treatment. Furthermore, LC3 puncta were visualized using a laser scanning fluorescent confocal microscope. As shown in Fig. 3B, radiation treatment enhanced the accumulation of LC3 puncta in HaCaT cells, which was effectively suppressed by NAC.
To confirm these results, transmission electron microscopy was performed to visualize autophagic structures in HaCaT cells. As shown in Fig. 3C, untreated cells showed normal nuclei, mitochondria, and other organelles and no autophagic vacuoles were detected. In contrast, autophagic vacuoles were observed in irradiated cells, suggesting that radiation treatment induced autophagy in HaCaT cells. In addition, NAC treatment significantly decreased the number of autophagic vacuoles compared with radiation treatment.
In summary, NAC can effectively inhibit the transcription of LC3B, ATG5, and p62 in HaCaT cells (Fig. 3D).
4. NAC protected against radiation-induced histopathological changes in injured rat buccal mucosa
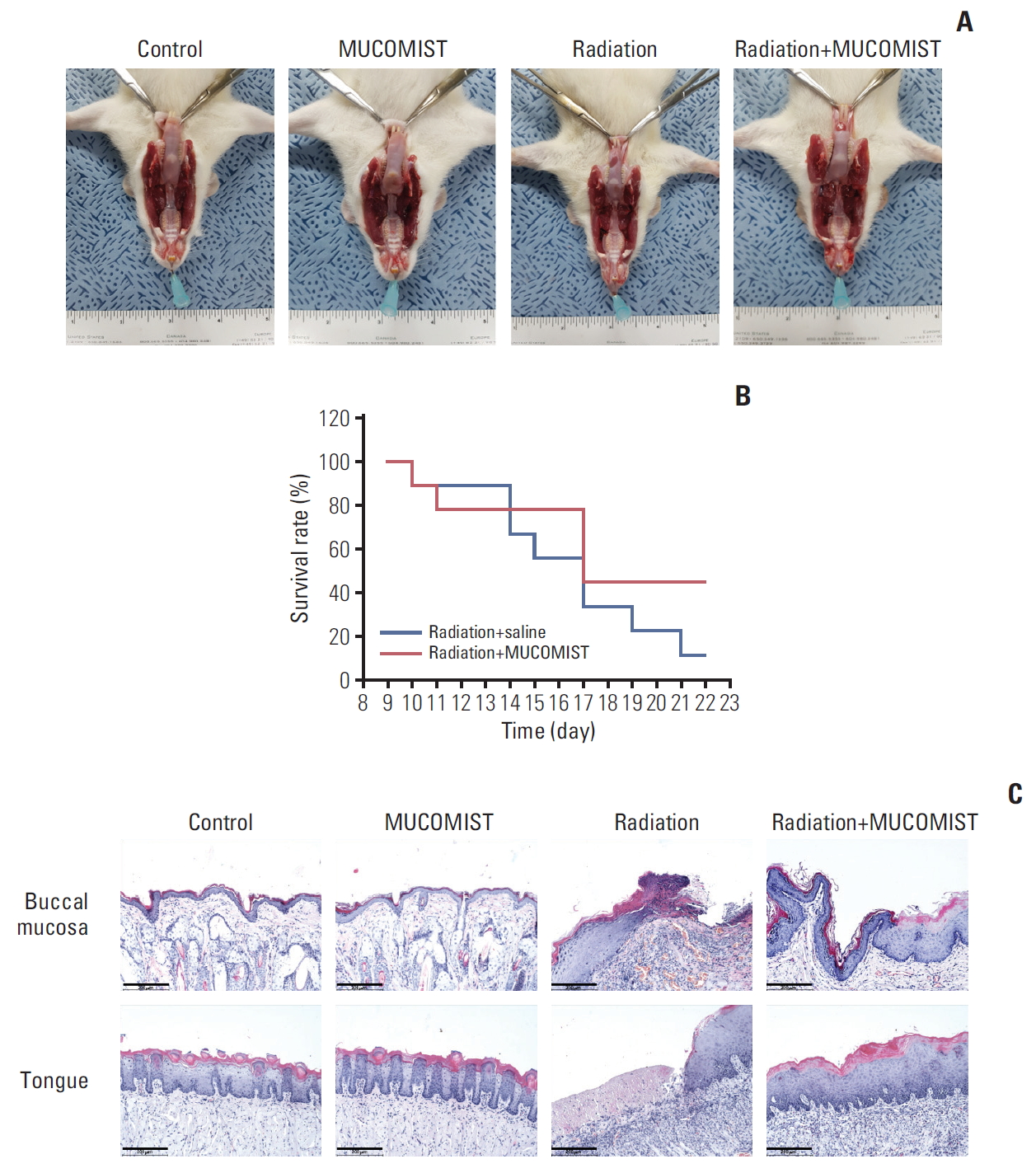
To determine the protective effects of NAC against radiation in vivo, we exposed rats to 30 Gy of radiation plus NAC. Thereafter, we investigated the morphological changes caused by the treatment and the effect on survival up to 3 weeks of development. All animals were examined after death during the experiment or euthanized after 3 weeks.
The food intake between the experimental groups was not significantly different until day 5, when the irradiation group and the radiation+NAC-treated group showed a significant decrease in food intake. Therefore, there was no statistically significant difference in the average food intake of the radiation+NAC group and the irradiation group (S1A Fig.). The weights of the irradiated rats were also not significantly different from those of the radiation+NAC group (S1B Fig.).
Inflammation and survival rate, however, were inhibited in the radiation+NAC group compared with that in the radiation treatment group (Fig. 4B).
Histopathologically, the buccal mucosa of the irradiation group was severely ulcerated with necrotic inflammatory exudates. However, after 3 weeks of irradiation, the tongue and buccal mucosa in the radiation+NAC group recovered similar to those in the control group (Fig. 4A). Mucosal recovery was also observed in hematoxylin-eosin stained tissue (Fig. 4C).
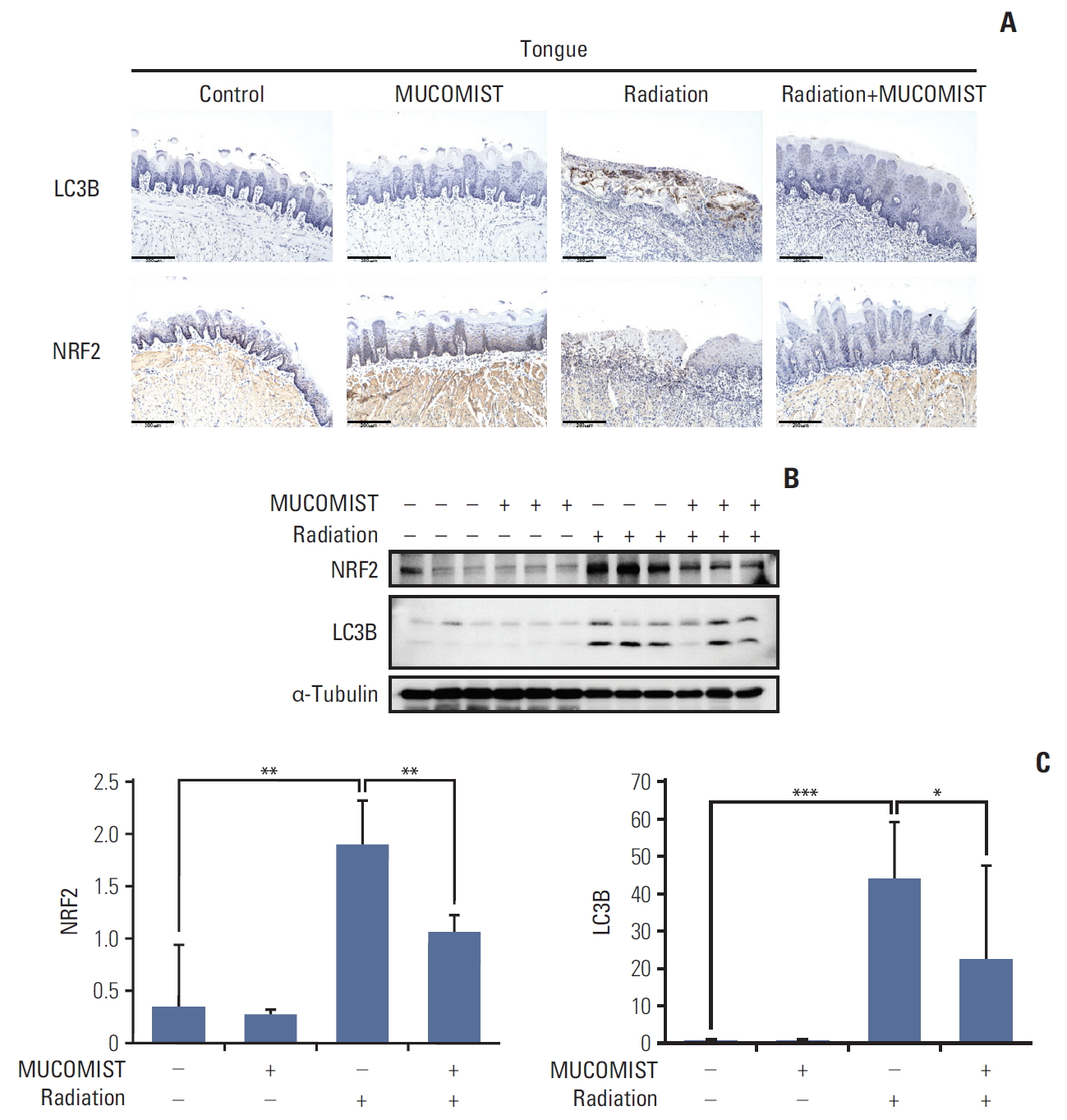
5. NAC prevents radiation-induced autophagy and NRF2 expression in rat buccal mucosa
To determine whether NAC treatment had similar effects in vivo, we confirmed the expression of NRF2 and LC3B in the rat tongue. The expression levels of LC3B and NRF2 were elevated in the group exposed to radiation, compared with those in the group treated with radiation+NAC (Fig. 5A).
We also conducted western blotting for NRF2 and LC3B in the rat tongue. NRF2 and LC3B protein levels increased in the radiation treatment group. In the radiation+NAC group, however, there was a marked decrease in NRF2 and LC3B expression (Fig. 5B and C), indicating that NAC treatment was effective in vivo as well as in vitro.
Discussion
The mechanism of radiation-induced oral mucositis is not fully understood. However, radiation therapy is essential for tumor treatment and is commonly used despite its side effects, such as skin damage and oral mucositis [22].
Radiotherapy and chemotherapy are known to induce ROS generation that activates various signaling pathways that have recently been reported to interact via autophagy-inducing factors [23]. These activated pathways inhibit cell regeneration and induce apoptosis and ulceration. DNA damage is also known to be induced directly by radiation; indirectly, S-phase arrest is induced by ROS [19]. Our results showed that NAC secured the progression of the S-phase by suppressing ROS-mediated DNA damage. In addition, another previous report demonstrated that NAC protected the cell cycle against UV damage; our results also showed its protective effect on the cell cycle against radiation damage [24]. Thus, we hypothesized that the antioxidant NAC is a possible protective agent against radiation-induced oral mucositis.
Previous studies have identified the preventive effect of NAC on radiation through 3D cell culture [25]. However, our experiments were performed in vivo to confirm the effect of NAC. In addition, the report did not observe the effect of NAC single treatment. Apoptosis was reduced when QYD (Qingre Liyan decoction) was used with NAC. In our study, cell damage was caused by ROS, which is increased upon irradiation, thereby resulting in increased autophagy. However, NAC treatment inhibited ROS production, and consequently, prevented cell death.
In our previous report, we demonstrated that the protective effect of NAC against radiation and cisplatin is mediated via the reduction of ROS generation [26]. Natural antioxidants play a role in autophagy and cell death induction. Therefore, we investigated the protective effect of NAC against radiation-induced cellular damage in HaCaT cells and in a rat model [27]. We concluded that NAC treatment prevents radiation-induced mucositis by decreasing NRF2-dependent ROS synthesis. Recent studies have shown that a low-dose radiation of 2.5 Gy increased the nuclear accumulation of NFR2 in the mouse macrophage cell line RAW 264.7 [28]. In our study, we also found nuclear accumulation of NRF2 induced by exposure to radiation.
The activation mechanism of NRF2 is mediated via direct interaction between p62 and SQSTM1, as well as the antioxidant response. Given that the accumulation of p62 and SQSTM1 is a hallmark of autophagy response, we investigated the association between autophagy and the NRF2 pathway.
Autophagy is activated by ROS. Under oxidative stress, autophagy has a protective effect against cardiovascular, renal, and neurological disorders and obesity [29,30]. Therefore, it is an important regulator of cell survival following damage and exposure to increased ROS levels or starvation. Autophagy eliminates ROS within the cell, which is similar to the role of the antioxidant-signaling pathway.
In our study, radiation-induced autophagy promoted the expression of NRF2 and induced the expression of p62. These results suggested that autophagy may play a regulatory or synergistic role in antioxidant-signaling pathways. However, the role of autophagy in radiation-induced early and delayed toxicity is unclear, and studies investigating the toxicity and death in autophagy are still ongoing.
However, regarding the mechanism involved, autophagy was activated and NRF2 production was promoted in irradiated cells by ROS. In our study, we found that NAC prevented both autophagy and NRF2 production by inhibiting the synthesis of radiation-induced ROS in cells and an animal model, thus demonstrating the ability of NAC to protect against radiation-induced DNA damage, including oral mucositis. We observed the protective effect of NAC against radiation-induced cellular damage via its function in inhibiting ROS production.
Although the relevance of autophagy requires further elucidation to confirm the effectiveness of NAC, our results suggest that NAC may inhibit oral mucositis, which is the most common complication of chemotherapy.
Even though our animal experiments showed that treatment with NAC is not effective with respect to oral intake and body weight, histological analysis results confirmed that NAC treatment improved tissue healing rate and quality. Therefore, it seems that NAC can treat radiation-induced oral mucositis, although NAC treatment did not affect oral intake or body weight. Therefore, more experiments are required to find a more effective way of NAC treatment, such as oral intake and body weight, to improve clinical parameters.
In this study, we demonstrated that NAC significantly inhibited radiation-induced autophagy in keratinocytes and rat buccal mucosa. These results suggest that NAC may be a safe and effective therapeutic candidate for the inhibition of radiation-induced buccal mucosa damage.